Back
BackMatter and Energy: Classification, Properties, and Changes
Study Guide - Smart Notes

Matter and Its Classification
Definition and Types of Matter
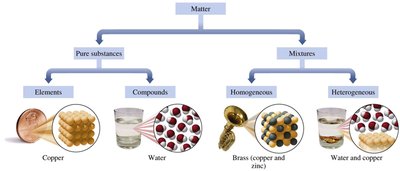
Matter is anything that has mass and occupies space. All physical objects are composed of matter, which can be classified based on its composition into pure substances and mixtures.
Pure Substances: Have a fixed or definite composition. They can be further classified as elements or compounds.
Mixtures: Contain two or more different substances that are physically mixed but not chemically combined. The composition can vary and components can be separated by physical methods.
Pure Substances: Elements and Compounds
Elements: Pure substances composed of only one type of atom (e.g., copper, lead, aluminum).
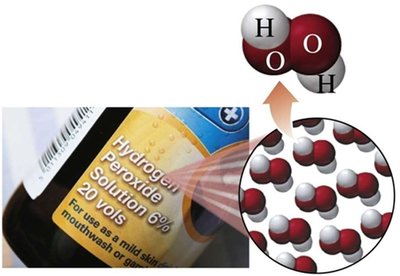
Compounds: Pure substances composed of two or more elements chemically combined in a fixed ratio (e.g., water, table salt, sugar, hydrogen peroxide).

Mixtures: Homogeneous and Heterogeneous
Homogeneous Mixtures (Solutions): Have a uniform composition throughout; different parts are not visible (e.g., brass, air, salt water).
Heterogeneous Mixtures: Composition varies from one part to another; different parts are visible (e.g., salad, sand and water, oil and water).
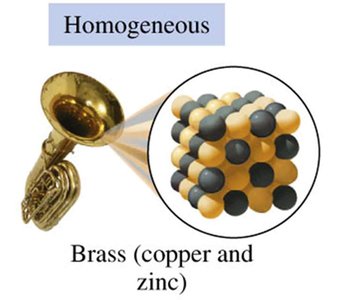
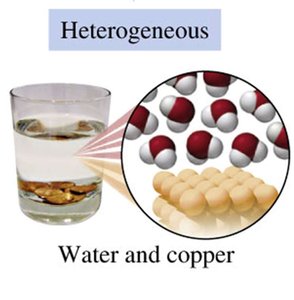
States and Properties of Matter
Physical States of Matter
Matter exists in three physical states: solid, liquid, and gas. Each state is characterized by the arrangement and movement of its particles.
Solids: Definite shape and volume; particles are closely packed in a fixed, rigid pattern and vibrate in place.
Liquids: Definite volume but no definite shape; particles are close but can move past each other, allowing liquids to flow and take the shape of their container.
Gases: No definite shape or volume; particles are far apart and move rapidly, filling the shape and volume of their container.
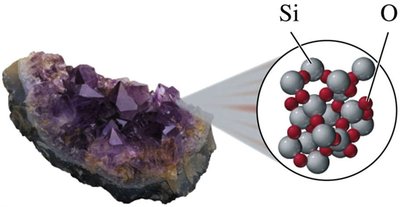
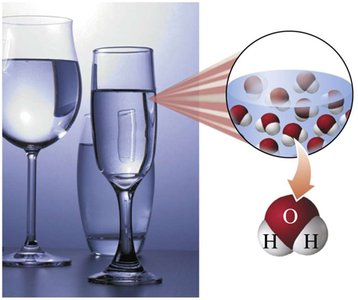

Physical and Chemical Properties
Physical Properties: Characteristics observed or measured without changing the substance's identity (e.g., color, melting point, boiling point, density, conductivity).
Chemical Properties: Describe the ability of a substance to interact with other substances and change into a new substance (e.g., flammability, reactivity).

Physical and Chemical Changes
Physical Changes: Changes in state or appearance without altering composition (e.g., melting, boiling, dissolving, cutting).
Chemical Changes: Substances are transformed into new substances with different properties (e.g., rusting, burning, caramelizing sugar).

Temperature and Energy
Temperature Scales
Temperature measures how hot or cold an object is. The three main temperature scales are Fahrenheit (°F), Celsius (°C), and Kelvin (K).
Celsius (°C): Used in science; water freezes at 0°C and boils at 100°C.
Fahrenheit (°F): Common in the United States; water freezes at 32°F and boils at 212°F.
Kelvin (K): The SI unit; absolute zero (0 K) is the lowest possible temperature, equivalent to -273°C.

Temperature Conversion Equations
From Celsius to Fahrenheit:
From Celsius to Kelvin:
Energy: Kinetic and Potential
Kinetic Energy: Energy of motion (e.g., moving objects, flowing water).
Potential Energy: Stored energy due to position or chemical composition (e.g., energy in food, gasoline).
Heat and Units of Energy
Joule (J): The SI unit of energy; 1 kilojoule (kJ) = 1000 J.
Calorie (cal): The amount of energy needed to raise the temperature of 1 gram of water by 1°C; (exact).
kilocalorie (kcal): 1000 calories; used in food energy (1 Calorie = 1 kcal).
Energy and Nutrition
Carbohydrates are the body's primary energy source, followed by fats and proteins when carbohydrate reserves are depleted. Food energy is measured in Calories (kcal) or kilojoules (kJ).

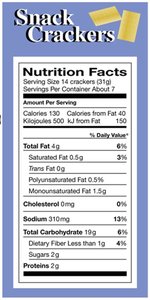
Food Type | kJ/g | kcal/g |
|---|---|---|
Carbohydrate | 17 | 4 |
Fat | 38 | 9 |
Protein | 17 | 4 |
Heat, Specific Heat, and Phase Changes
Specific Heat
Specific heat is the amount of heat required to raise the temperature of 1 gram of a substance by 1°C. It is substance-specific and measured in J/g°C or cal/g°C.
Equation:
Where = heat (J or cal), = mass (g), = specific heat, = temperature change (°C)

Substance | J/g°C | cal/g°C |
|---|---|---|
Water (liquid) | 4.184 | 1.00 |
Aluminum | 0.897 | 0.214 |
Copper | 0.385 | 0.0920 |
Iron | 0.452 | 0.108 |
Gold | 0.129 | 0.0308 |
Silver | 0.235 | 0.0562 |
Titanium | 0.523 | 0.125 |
Ethanol | 2.46 | 0.588 |
Ammonia | 2.04 | 0.488 |
Sodium chloride | 0.864 | 0.207 |
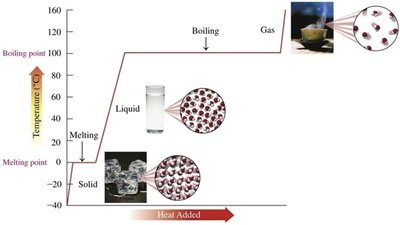
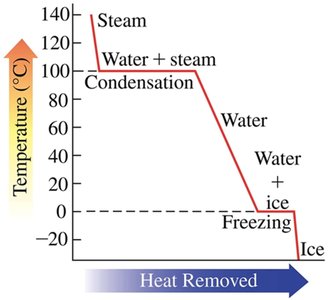
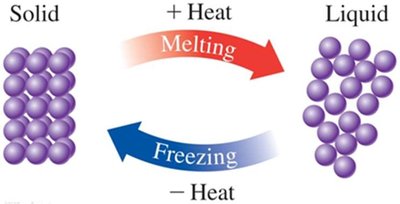
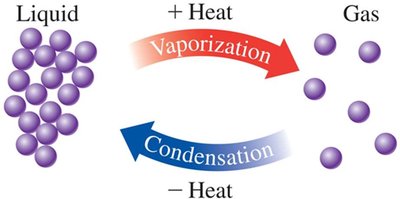
Phase Changes: Melting, Freezing, Boiling, and Condensation
Melting: Solid to liquid at the melting point; requires heat input.
Freezing: Liquid to solid at the freezing point; releases heat.
Boiling (Vaporization): Liquid to gas at the boiling point; requires heat input.
Condensation: Gas to liquid at the boiling point; releases heat.

Heats of Fusion and Vaporization
Heat of Fusion (water): or (energy to melt 1 g of ice at 0°C)
Heat of Vaporization (water): or (energy to vaporize 1 g of water at 100°C)
Equations:
Heat for melting/freezing:
Heat for vaporization/condensation:
Heating and Cooling Curves
Heating and cooling curves graphically represent temperature changes as heat is added or removed. Diagonal lines indicate temperature changes within a phase, while horizontal lines (plateaus) indicate phase changes where temperature remains constant.