 Back
BackMatter and Energy: Classification, Properties, and Energy in Nutrition
Study Guide - Smart Notes

Classification of Matter
Definition and Categories
Matter is defined as anything that has mass and occupies space. It constitutes all substances and objects in the universe, including water, wood, and plastics. Matter can be categorized based on its state and composition.
States of Matter: Solid, liquid, and gas.
Composition: Pure substances (elements and compounds) and mixtures (homogeneous and heterogeneous).

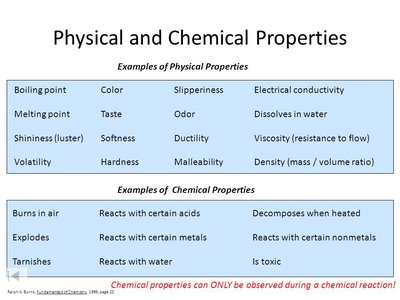
Physical and Chemical Properties
Definitions and Examples
Properties of matter are divided into physical and chemical properties:
Physical Properties: Characteristics observed or measured without changing the substance's identity (e.g., color, density, melting point).
Chemical Properties: Characteristics that describe how a substance reacts or changes into other substances (e.g., flammability, reactivity with acids).
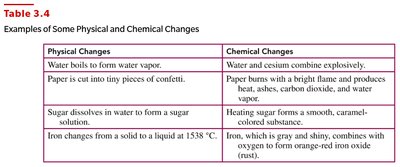
Physical and Chemical Changes
Distinguishing Changes
Changes in matter are classified as physical or chemical:
Physical Change: Alters the form or appearance but not the composition (e.g., melting ice, cutting paper).
Chemical Change: Produces one or more new substances with different properties (e.g., rusting iron, burning wood).
Physical Changes | Chemical Changes |
|---|---|
Water boils to form vapor | Paper burns with a bright flame |
Paper is cut into confetti | Iron rusts to form iron oxide |
Sugar dissolves in water | Heating sugar forms caramel |
Iron melts at 1538°C | Water and cesium combine explosively |
Density
Definition and Calculation
Density is a physical property defined as mass per unit volume. It is calculated using the formula:
Example: If a bone sample has a mass of 2.15 g and a volume of 1.40 cm3, its density is .
Energy
Forms and Units
Energy is the ability to do work. It exists in two main forms:
Kinetic Energy: Energy of motion (e.g., moving water, rollerblading).
Potential Energy: Stored energy due to position or composition (e.g., water behind a dam, chemical bonds in food).
The SI unit for energy is the joule (J), but the calorie (cal) is also commonly used, especially in nutrition.
Energy in Nutrition
Calories and Food Types
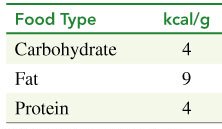
Food provides energy, measured in kilocalories (kcal) or nutritional Calories (Cal). The energy content of macronutrients is:
Food Type | kcal/g |
|---|---|
Carbohydrate | 4 |
Fat | 9 |
Protein | 4 |
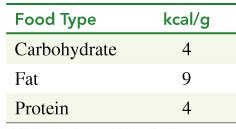
Example Calculation: 1 cup of whole milk contains 12 g carbohydrate, 9 g fat, and 9 g protein. Total energy = (12 × 4) + (9 × 9) + (9 × 4) = 48 + 81 + 36 = 165 kcal
The number of kilocalories needed daily depends on age, gender, and activity level. Carbohydrates are the primary energy source, followed by fats and proteins when carbohydrate reserves are depleted.

Practice and Application
Classifying Properties and Changes
Physical Properties: Color, hardness, odor, density, melting point, solubility.
Chemical Properties: Flammability, reactivity with acids, toxicity.
Physical Changes: Ice melting, cutting pizza, boiling water.
Chemical Changes: Burning wood, rusting iron, toasting marshmallow.
