 Back
BackMatter and Energy: Physical and Chemical Properties, Changes, and Energy in Nutrition
Study Guide - Smart Notes

Matter and Energy
Classification of Matter
Matter is defined as anything that has mass and occupies space. It makes up all substances and materials encountered in daily life, such as water, wood, and plastics. Matter can be classified based on its state (solid, liquid, gas) and its composition (pure substances or mixtures).
States of Matter: Solid, liquid, gas
Composition: Pure substances (elements and compounds), mixtures (homogeneous and heterogeneous)
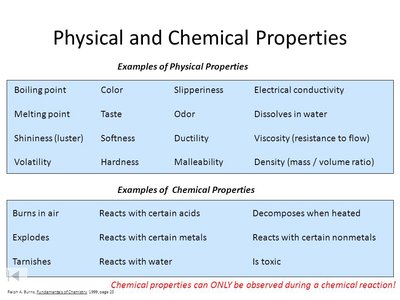
Physical and Chemical Properties
Properties of matter are categorized as physical or chemical. Physical properties can be observed or measured without changing the substance's identity, while chemical properties describe how a substance reacts or changes into other substances.
Physical Properties: Color, melting point, density, hardness, odor, solubility, electrical conductivity
Chemical Properties: Flammability, reactivity with acids, toxicity, decomposition, tarnishing
Physical and Chemical Changes
Changes in matter are classified as physical or chemical. Physical changes alter the form or appearance of matter without changing its composition, while chemical changes result in the formation of new substances with different properties.
Physical Change: Melting, boiling, dissolving, cutting
Chemical Change: Burning, rusting, decomposition, reacting with acids
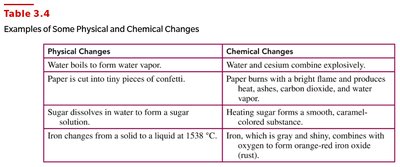
Physical Changes | Chemical Changes |
|---|---|
Water boils to form water vapor. | Water and cesium combine explosively. |
Paper is cut into tiny pieces of confetti. | Paper burns with a bright flame and produces heat, ashes, carbon dioxide, and water vapor. |
Sugar dissolves in water to form a sugar solution. | Heating sugar forms a smooth, caramel-colored substance. |
Iron changes from a solid to a liquid at 1538 °C. | Iron, which is gray and shiny, combines with oxygen to form orange-red iron oxide (rust). |
Examples of Physical and Chemical Changes
Physical Change Example: A gold ingot hammered to form gold leaf.
Chemical Change Example: Iron nails corrode in the presence of water, forming rust (Fe2O3).


Energy
Definition and Types of Energy
Energy is the ability to do work. It is classified as kinetic energy (energy of motion) or potential energy (energy stored due to position or chemical composition).
Kinetic Energy: Energy of moving objects (e.g., rollerblading, mowing the lawn)
Potential Energy: Energy stored in objects or substances (e.g., water behind a dam, food, gasoline)
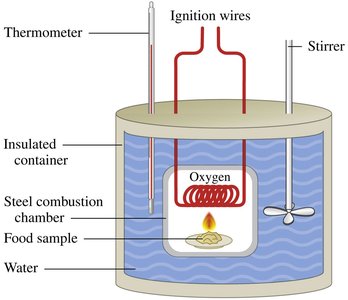
Units of Energy
The SI unit for energy is the joule (J). In nutrition, energy is often measured in calories (cal) and kilocalories (kcal).
1 calorie (cal): Energy needed to raise the temperature of 1 g of water by 1°C
1 kilocalorie (kcal): 1000 calories
1 nutritional Calorie (Cal): 1 kcal

Energy and Nutrition
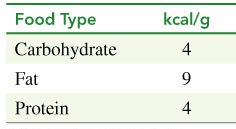
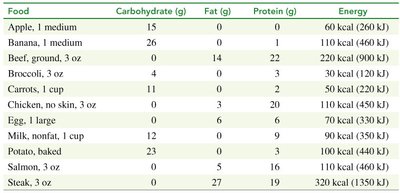
Food provides energy for the body. Carbohydrates are the primary source, followed by fats and proteins when carbohydrate reserves are depleted. The energy value of food is measured in kcal/g.
Carbohydrates: 4 kcal/g
Fats: 9 kcal/g
Proteins: 4 kcal/g

Calculating Energy Content in Food
The total energy in a food item is calculated by multiplying the mass of each macronutrient by its energy value and summing the results.
Formula:
Example: 1 apple contains 15 g carbohydrates: kcal
Example: 1 egg contains 6 g fat and 6 g protein: kcal (fat), kcal (protein), total kcal
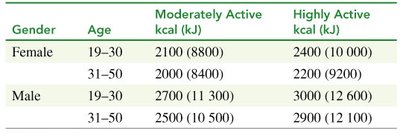
Daily Energy Requirements
The number of kilocalories needed daily depends on gender, age, and physical activity. Weight loss occurs when food intake is less than energy output.
Gender | Age | Moderately Active (kcal) | Highly Active (kcal) |
|---|---|---|---|
Female | 19–30 | 2100 | 2400 |
Female | 31–50 | 2000 | 2200 |
Male | 19–30 | 2700 | 3000 |
Male | 31–50 | 2500 | 2900 |
Summary Table: Physical vs. Chemical Properties and Changes
Physical Properties | Chemical Properties |
|---|---|
Color, melting point, density, hardness, odor, solubility | Flammability, reactivity with acids, toxicity, decomposition |
Physical Changes | Chemical Changes |
|---|---|
Melting, boiling, dissolving, cutting | Burning, rusting, decomposition |
Key Point: Physical properties and changes do not alter the chemical identity of a substance, while chemical properties and changes result in new substances.
