 Back
BackMatter and Measurements: Foundations of Chemistry for Healthcare Professionals
Study Guide - Smart Notes

Matter and Measurements
Chemistry: The Science of Everyday Experience
Chemistry is the study of matter—its composition, properties, and transformations. Matter is anything that has mass and occupies space. Chemistry is fundamental to understanding the materials and processes encountered in healthcare and daily life.

States of Matter
Matter exists in three primary states: solid, liquid, and gas. Each state has distinct physical properties based on the arrangement and movement of particles.
Solid State
Definite volume and shape
Particles are closely packed in a regular pattern
Liquid State
Definite volume but no definite shape
Takes the shape of its container
Particles are close together but can move past one another
Gas State
No definite shape or volume
Assumes the shape and volume of its container
Particles are far apart and move randomly
Chemical and Physical Properties of Matter
Properties of matter are classified as chemical or physical:
Chemical properties: Observed only when matter is changed into a new substance (e.g., flammability, reactivity).
Physical properties: Observed without changing the composition (e.g., color, shape, odor, boiling point, melting point, solubility).


Chemical and Physical Changes
Chemical changes: Accompanied by a change in composition (e.g., burning paper, fizzing of vinegar and baking soda).
Physical changes: Occur without a change in composition (e.g., freezing, melting, evaporation).

Atoms and Molecules
Dalton’s Atomic Theory (1808)
All matter is made up of tiny particles called atoms.
Elements are made up of identical atoms.
Compounds are combinations of atoms of two or more elements.
Atoms are rearranged in chemical reactions but are never created nor destroyed.
Molecules vs. Atoms
Molecule: Smallest particle of a pure substance capable of stable independent existence.
Atom: Basic particle that makes up molecules.
Classification of Matter
Matter is classified based on its chemical and physical properties:
Pure Substances
Have constant composition and fixed properties.
Classified as elements or compounds.
Example: Pure water always contains the same proportions of hydrogen and oxygen.
Elements
Made up of homoatomic molecules or individual atoms of the same kind.
Examples: Oxygen gas (O2), copper metal (Cu).
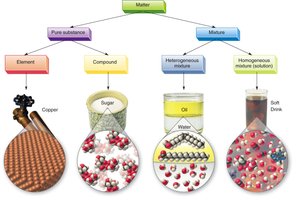

Compounds
Made up of heteroatomic molecules or ions of two or more different kinds.
Examples: Water (H2O), table salt (NaCl).

Mixtures
Composition and properties can vary.
Two types: homogeneous (solutions) and heterogeneous.
Heterogeneous Mixtures
Properties depend on the location of the sample.
Examples: Blood, oil and vinegar, smog, soda.
Homogeneous Mixtures
Also called solutions.
Properties are uniform throughout the mixture.
Example: Sugar dissolved in water.
Summary Table: Classification of Matter
Type | Definition | Example |
|---|---|---|
Element | Pure substance, one kind of atom | Copper, Oxygen |
Compound | Pure substance, two or more kinds of atoms | Water, Table salt |
Homogeneous Mixture | Uniform composition | Sugar water, air |
Heterogeneous Mixture | Non-uniform composition | Blood, oil and vinegar |
Measurement
Measurements are fundamental in chemistry and consist of a number and a unit. Units are standardized and measurements are made using devices such as rulers, balances, and graduated cylinders.
Metric System
Decimal system; units related by factors of 10.
Base units: meter (length), gram (mass), liter (volume).
Prefixes indicate multiples or fractions of base units (e.g., kilo-, centi-, milli-).
Mass vs. Weight
Mass: Amount of matter in an object; independent of location.
Weight: Gravitational force acting on an object; depends on location.
Volume
Base unit: liter (L).
1 mL = 1 cm3 = 1 cc.
Volume formula:
Significant Figures
Significant figures reflect the precision of a measurement. Exact numbers result from counting or definitions; inexact numbers result from measurements.
All non-zero digits are significant.
Zeros between non-zero digits are significant.
Leading zeros are not significant.
Zeros at the end of a decimal are significant.
Zeros at the end of a non-decimal are not significant.
Rules for Calculations
Multiplication/Division: Answer has the same number of significant figures as the original number with the fewest significant figures.
Addition/Subtraction: Answer has the same number of decimal places as the original number with the fewest decimal places.
Scientific Notation
Scientific notation expresses numbers as a coefficient (between 1 and 10) multiplied by a power of 10. Useful for very large or small numbers.
Example:
Example:
Problem Solving Using Conversion Factors
Conversion factors are used to convert quantities from one unit to another. They are written as equalities and used as fractions to cancel unwanted units.
Example:
To convert 130 lb to kg:
General Steps
Identify the original quantity and unit.
Choose the appropriate conversion factor.
Set up the calculation so unwanted units cancel.
Perform the calculation and round to the correct number of significant figures.
Summary
This chapter introduces the foundational concepts of matter, its classification, measurement, and the importance of precision in scientific work. Understanding these basics is essential for further study in chemistry and its applications in healthcare.
