 Back
BackMeasurements, Matter, and Energy: Study Notes for College Chemistry
Study Guide - Smart Notes

Measurements in Chemistry
Standard International (SI) Units and Metric System
Accurate measurement is fundamental in chemistry. The SI system is the global standard for scientific measurements, ensuring consistency and clarity across disciplines.
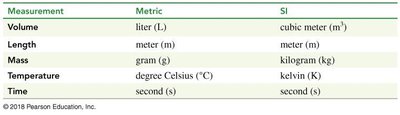
Volume: SI unit is cubic meter (m3), metric unit is liter (L).
Length: Both SI and metric use meter (m).
Mass: SI unit is kilogram (kg), metric unit is gram (g).
Temperature: SI unit is kelvin (K), metric unit is degree Celsius (°C).
Time: Both SI and metric use second (s).
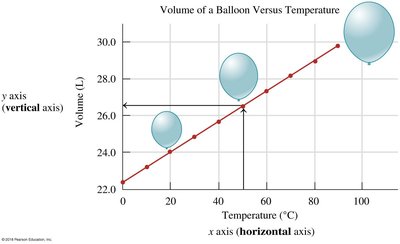
Graphing Data
Graphs are essential for visualizing relationships between variables in chemistry. The x-axis represents the independent variable, while the y-axis shows the dependent variable.
Example: Volume of a balloon versus temperature demonstrates how volume changes with temperature.
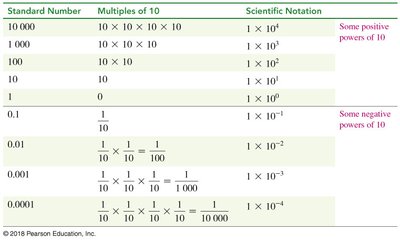
Scientific Notation
Scientific notation simplifies the expression of very large or small numbers, using a coefficient and a power of ten.
Format: where a is the coefficient (1 ≤ a < 10), n is the exponent.
Moving decimal left: Negative exponent.
Moving decimal right: Positive exponent.


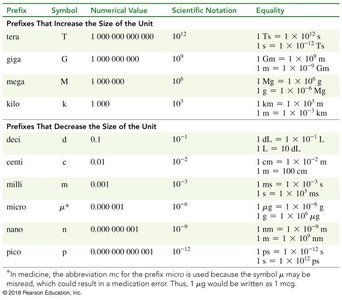
Metric Prefixes and Orders of Magnitude
Prefixes indicate the size of units in the metric system, allowing for easy conversion between scales.
Kilo (k):
Centi (c):
Milli (m):
Micro (μ):
Nano (n):
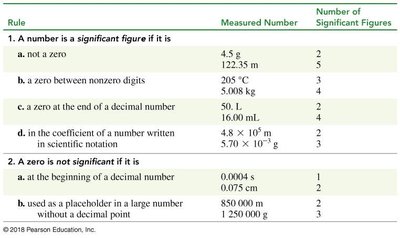
Significant Figures
Significant figures reflect the precision of a measurement. Rules determine which digits are significant.
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Zeros at the end of a decimal number are significant.
Zeros at the beginning or as placeholders are not significant.
Scientific notation: All digits in the coefficient are significant.
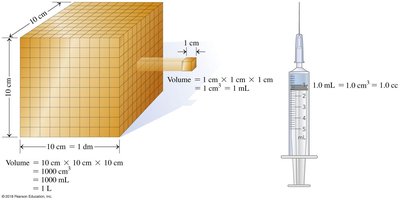
Volume and Conversion Factors
Volume is commonly measured in liters and milliliters. Conversion factors are used to switch between units.
1 mL = 1 cm3
1 L = 1000 mL
1 in = 2.54 cm (exact)

Unit Cancellation and Conversion Examples
Unit cancellation is a method for converting between units using conversion factors.
Example: Converting pounds to kilograms using .
Formula:



Matter and Its Classification
Types of Matter
Matter is classified as pure substances or mixtures. Pure substances include elements and compounds, while mixtures can be homogeneous or heterogeneous.
Elements: Single type of atom (e.g., copper).
Compounds: Two or more elements chemically combined (e.g., water).
Homogeneous mixtures: Uniform composition (e.g., brass).
Heterogeneous mixtures: Non-uniform composition (e.g., water and copper).
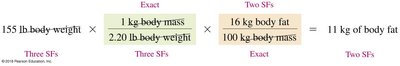
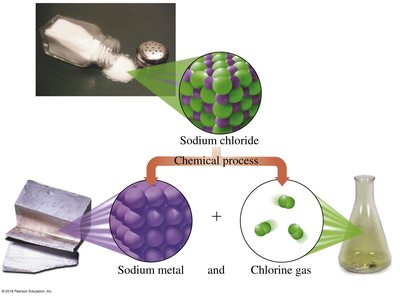
Physical vs Chemical Changes
Physical changes alter the form of a substance without changing its identity, while chemical changes produce new substances.
Physical changes: Phase changes (melting, freezing, boiling).
Chemical changes: Formation of new compounds (e.g., sodium chloride from sodium and chlorine).
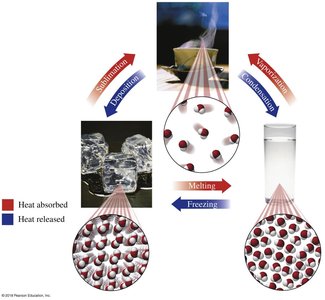
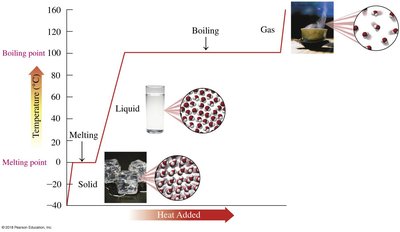
States of Matter and Phase Change Diagrams
Matter exists in solid, liquid, and gas phases. Phase changes occur when energy is added or removed.
Melting point: Temperature at which a solid becomes a liquid.
Boiling point: Temperature at which a liquid becomes a gas.
Phase change diagrams: Show temperature changes and energy input during transitions.
Physical Properties: Density and Temperature
Density
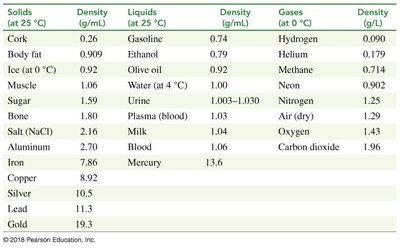
Density is a physical property defined as mass per unit volume. It is unique for each substance and can be used for identification.
Formula:
Units: g/mL for liquids and solids, g/L for gases.
Measurement: Volume displacement method for solids.

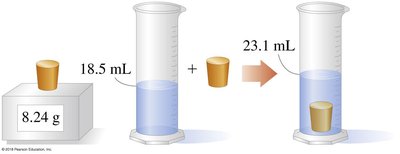
Temperature
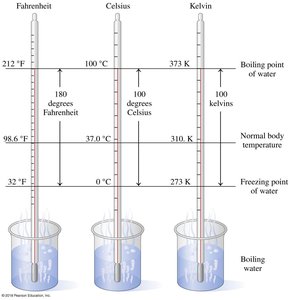
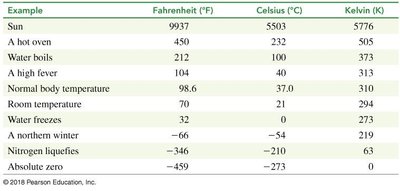
Temperature measures the average kinetic energy of molecules. Common scales include Celsius, Fahrenheit, and Kelvin.
Celsius: Based on water's melting and boiling points.
Kelvin: Absolute scale, zero is absolute zero.
Fahrenheit: Used in the U.S., less common in science.
Conversion formulas:
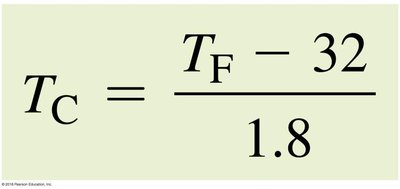
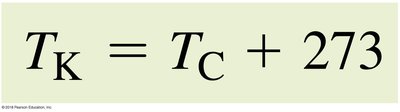
Energy in Chemistry
Forms of Energy and Units
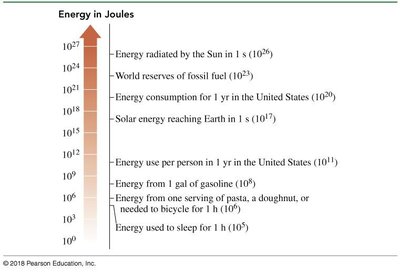
Energy is the capacity to do work or produce heat. The SI unit is the joule (J), while the calorie (cal) is commonly used in nutrition.
1 cal = 4.184 J
Energy conversions: and
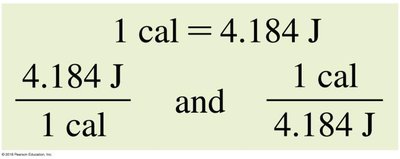
Energy in Nutrition
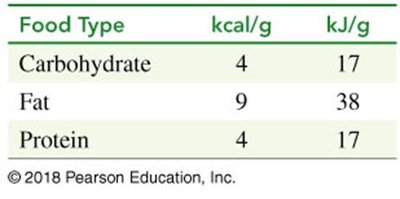
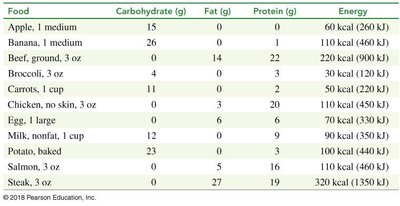
Food provides energy in the form of carbohydrates, fats, and proteins. Energy content is measured in kilocalories (kcal) or kilojoules (kJ).
Carbohydrate: 4 kcal/g (17 kJ/g)
Fat: 9 kcal/g (38 kJ/g)
Protein: 4 kcal/g (17 kJ/g)
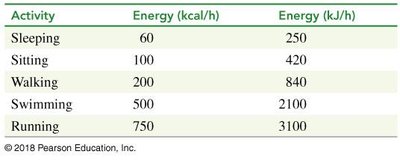
Energy Expenditure by Activity
Different activities require varying amounts of energy, measured in kcal/h or kJ/h.
Sleeping: 60 kcal/h
Sitting: 100 kcal/h
Walking: 200 kcal/h
Swimming: 500 kcal/h
Running: 750 kcal/h
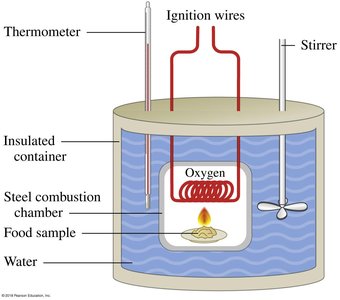
Specific Heat
Specific heat is the amount of heat required to raise the temperature of 1 gram of a substance by 1°C. It is a unique physical property for each substance.
Formula:
Units: cal/g°C or J/g°C
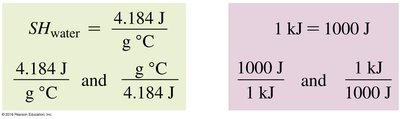
Water: SH = 1.00 cal/g°C or 4.184 J/g°C

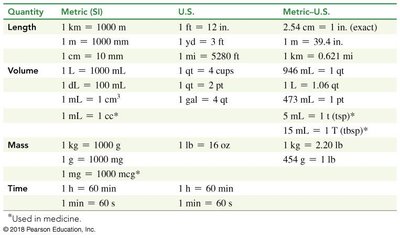
Summary Table: Key Conversion Factors
Quantity | Metric (SI) | U.S. | Metric-U.S. |
|---|---|---|---|
Length | 1 km = 1000 m | 1 yd = 3 ft | 1 m = 39.4 in.; 1 in. = 2.54 cm (exact) |
Volume | 1 L = 1000 mL | 1 qt = 4 cups | 946 mL = 1 qt |
Mass | 1 kg = 1000 g | 1 lb = 16 oz | 1 kg = 2.20 lb; 454 g = 1 lb |
Time | 1 h = 60 min | 1 h = 60 min | 1 min = 60 s |
*Additional info: This summary table consolidates key conversion factors for quick reference, supporting accurate calculations in chemistry.*
