 Back
BackMetabolic Pathways for Carbohydrates: Structure, Energy, and Coenzymes (Ch. 22 Study Notes)
Study Guide - Smart Notes

Ch. 22: Metabolic Pathways for Carbohydrates
Concept Map of Carbohydrate Metabolism
This chapter explores the major metabolic pathways for carbohydrates, focusing on the digestion of carbohydrates, the fate of glucose, and the interconnections between glycolysis, glycogenesis, glycogenolysis, and the pentose phosphate pathway.
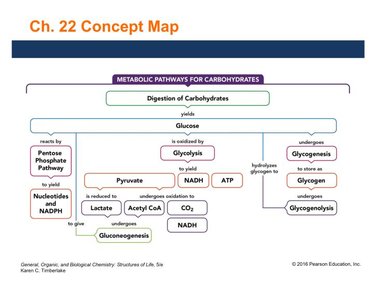
Metabolic Reactions of Glucose
Glucose is a central molecule in metabolism, serving as a substrate for multiple pathways. Key processes include glycolysis (breakdown of glucose), gluconeogenesis (synthesis of glucose), glycogenesis (formation of glycogen), and the pentose phosphate pathway (production of ribose and NADPH).
Glycolysis: Converts glucose to pyruvate, producing ATP and NADH.
Gluconeogenesis: Synthesizes glucose from noncarbohydrate sources.
Pentose Phosphate Pathway: Produces ribose (for nucleic acids) and NADPH (for biosynthetic reactions).
Glycogenesis: Converts glucose to glycogen for storage.
Glycogenolysis: Breaks down glycogen to release glucose.
Cori Cycle: Converts lactate (from anaerobic glycolysis) back to glucose in the liver.

Ch. 22.1 Metabolism and Energy
Overview of Metabolism
Metabolism encompasses all chemical reactions in living organisms, divided into two main types:
Catabolic reactions (catabolism): Break down large, complex molecules into smaller ones, releasing energy (exothermic).
Anabolic reactions (anabolism): Use ATP energy to build larger molecules from smaller ones (endothermic).
Example: The overall catabolic reaction for food metabolism is:

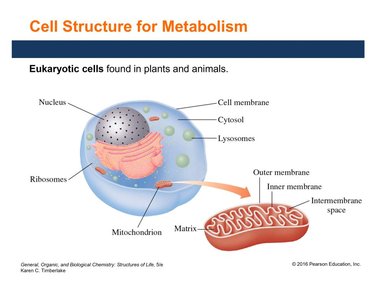
Stages of Catabolism
Catabolism occurs in three main stages:
Stage 1: Digestion – Enzymes hydrolyze large molecules into smaller units that enter the bloodstream.
Stage 2: Cellular Breakdown – Small molecules are further broken down into two- and three-carbon compounds within cells.
Stage 3: Oxidation – Acetyl CoA is oxidized to CO2 and H2O, producing ATP via the citric acid cycle and electron transport chain.
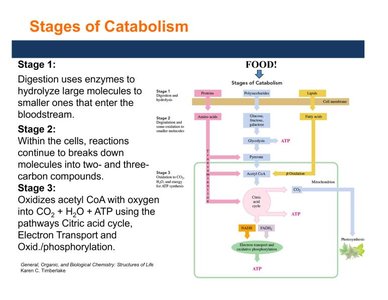
Cell Structure for Metabolism
Metabolic processes occur within eukaryotic cells, which contain specialized organelles:
Nucleus: Contains genetic material (DNA).
Mitochondria: Site of ATP synthesis and energy production.
Ribosomes: Protein synthesis.
Lysosomes: Digest and recycle cell structures.
Cell membrane: Separates cell contents from the environment.
Cytosol: Fluid part of cytoplasm where many reactions occur.
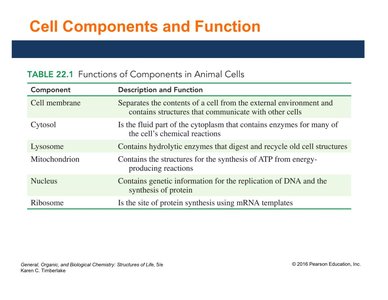
Cell Components and Their Functions
The following table summarizes the main components of animal cells and their functions:
Component | Description and Function |
|---|---|
Cell membrane | Separates cell contents from the environment; communication |
Cytosol | Fluid part of cytoplasm; site of many chemical reactions |
Lysosome | Contains enzymes for digestion and recycling |
Mitochondrion | Site of ATP synthesis from energy-producing reactions |
Nucleus | Contains genetic information for DNA replication and protein synthesis |
Ribosome | Site of protein synthesis using mRNA |
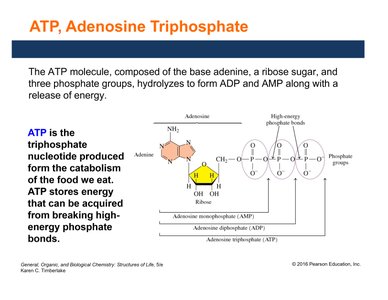
ATP: Adenosine Triphosphate
ATP (adenosine triphosphate) is the primary energy carrier in cells. It consists of adenine, ribose, and three phosphate groups. Hydrolysis of ATP to ADP (adenosine diphosphate) or AMP (adenosine monophosphate) releases energy used for cellular work.
ATP stores energy in high-energy phosphate bonds.
Energy is released when these bonds are broken.
Hydrolysis of ATP Yields Energy
The hydrolysis of ATP to ADP and inorganic phosphate (Pi) releases 7.3 kcal (31 kJ) per mole:
Hydrolysis of ADP to AMP also releases 7.3 kcal/mol:

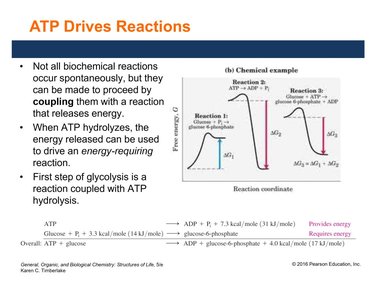
ATP Drives Reactions
Many biochemical reactions are not spontaneous and require energy input. These reactions are driven by coupling them to ATP hydrolysis. For example, the first step of glycolysis is coupled with ATP hydrolysis to provide the necessary energy.
ATP hydrolysis provides energy for endergonic (energy-requiring) reactions.
Coupling reactions is essential for metabolism.
22.2 Important Coenzymes in Metabolic Pathways
Redox Reactions and Coenzymes
Metabolic pathways often involve oxidation-reduction (redox) reactions. Oxidation is the loss of electrons or hydrogen, while reduction is the gain of electrons or hydrogen. Coenzymes such as NAD+, NADP+, FAD, and Coenzyme A are essential for carrying electrons, hydrogen ions, or functional groups during these reactions.
Oxidation: Loss of hydrogen/electrons or gain of oxygen; produces energy.
Reduction: Gain of hydrogen/electrons or loss of oxygen; requires energy input.
Coenzymes are required to carry electrons and H+ to or from the substrate.

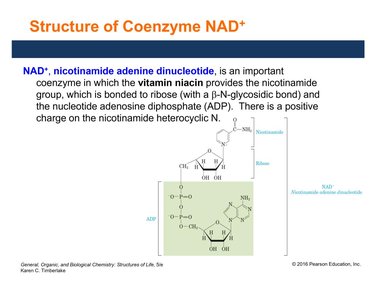
Structure and Function of Coenzyme NAD+
NAD+ (nicotinamide adenine dinucleotide) is derived from niacin (vitamin B3). It acts as an electron carrier in redox reactions, especially those forming carbonyl (C=O) bonds.
Composed of a nicotinamide group, ribose, and ADP.
Accepts electrons and hydrogen ions to become NADH.
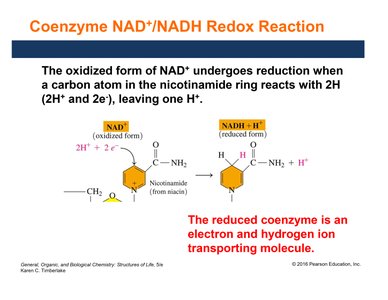
NAD+/NADH Redox Reaction
NAD+ is reduced to NADH by accepting two electrons and one hydrogen ion. NADH acts as an electron and hydrogen ion transporter in metabolism.
Function of NAD+
NAD+ is required in dehydrogenation reactions that produce carbon–oxygen double bonds, such as the oxidation of alcohols to aldehydes or ketones.

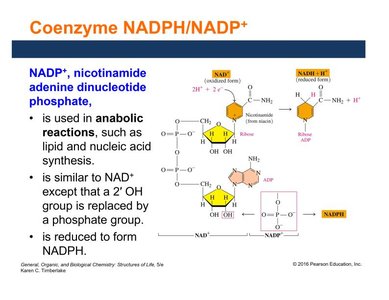
Coenzyme NADP+/NADPH
NADP+ (nicotinamide adenine dinucleotide phosphate) is similar to NAD+ but contains a phosphate group. It is used in anabolic reactions, such as lipid and nucleic acid synthesis, and is reduced to NADPH.
Participates in biosynthetic (anabolic) pathways.
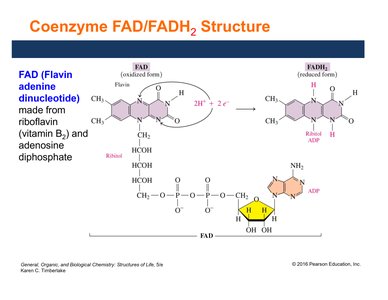
Coenzyme FAD/FADH2 Structure and Function
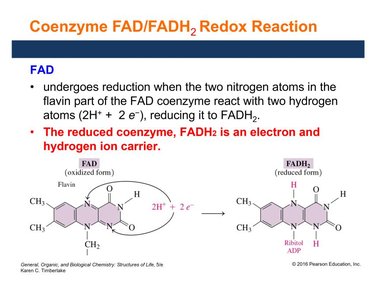
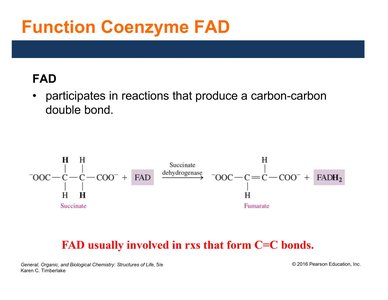
FAD (flavin adenine dinucleotide) is derived from riboflavin (vitamin B2). It acts as an electron and hydrogen carrier, especially in reactions forming carbon–carbon double bonds.
FAD is reduced to FADH2 by accepting two electrons and two hydrogen ions.
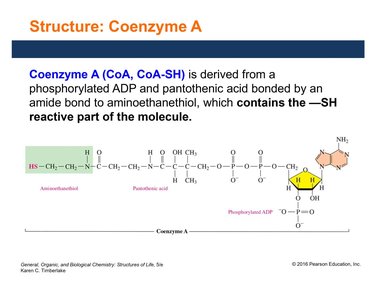
Structure and Function of Coenzyme A (CoA)
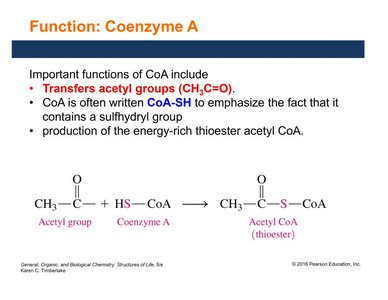
Coenzyme A (CoA, CoA-SH) contains a reactive sulfhydryl (–SH) group and is derived from pantothenic acid and ADP. It is essential for transferring acetyl groups (CH3CO–) and forming energy-rich thioester bonds, such as in acetyl CoA.
Transfers acetyl groups in metabolic pathways.
Key for the citric acid cycle and fatty acid metabolism.
Types of Metabolic Reactions
Metabolic reactions require enzymes and often coenzymes. The main types of reactions include oxidation, reduction, hydration, dehydration, rearrangement, transfer of phosphate or acetyl groups, decarboxylation, and hydrolysis. The following table summarizes the enzymes and coenzymes involved:
Reaction | Enzyme | Coenzyme |
|---|---|---|
Oxidation | Dehydrogenase | NAD+, NADP+, FAD |
Reduction | Dehydrogenase | NADH + H+, NADPH + H+, FADH2 |
Hydration | Hydratase | — |
Dehydration | Dehydratase | — |
Rearrangement | Isomerase, epimerase | — |
Transfer of phosphate group | Phosphatase, kinase | ATP, GDP, ADP |
Transfer of acetyl group | Acetyl CoA transferase | CoA |
Decarboxylation | Decarboxylase | ADP, GDP |
Hydrolysis | Protease, lipase, synthetase | ADP, GDP |

Summary
This chapter provides a comprehensive overview of the metabolic pathways for carbohydrates, the role of ATP as the energy currency of the cell, and the importance of coenzymes in facilitating redox and group transfer reactions. Understanding these concepts is essential for further study of metabolism and energy production in biological systems.
