 Back
BackMini-Textbook Study Guide: Molecular Compounds, Lewis Structures, Electronegativity, and Molecular Shapes
Study Guide - Smart Notes

Molecular Compounds: Sharing Electrons
Covalent Bonds and Molecular Compounds
Molecular compounds are formed when atoms of two or more nonmetals share electrons, resulting in covalent bonds. The shared electrons are valence electrons, and the atoms achieve stability by completing their valence shells. A molecule is a discrete group of atoms in a definite proportion.
Covalent bond: A chemical bond formed by the sharing of electron pairs between atoms.
Valence electrons: Electrons in the outermost shell involved in bonding.
Molecule: A group of atoms bonded together in a fixed ratio.
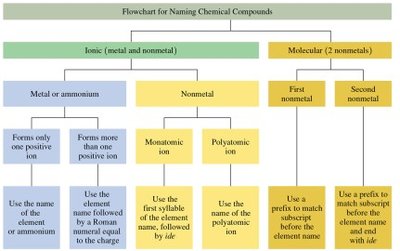
Naming Molecular Compounds
The names of molecular compounds require prefixes because several different compounds can be formed from the same two nonmetals.
The first nonmetal is named by its element name.
The second nonmetal is named using the first syllable of its name followed by ide.
Prefixes indicate the number of atoms: mono, di, tri, tetra, penta, hexa, hepta, octa, nona, deca.
Mono is never used with the first atom in the formula.
Examples of Molecular Compound Names and Formulas
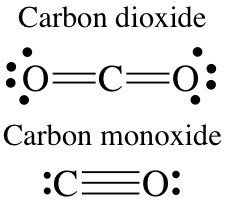
CO2: Carbon dioxide
CO: Carbon monoxide
NO: Nitrogen monoxide
NCl3: Nitrogen trichloride
B2O3: Diboron trioxide
P2O5: Diphosphorus pentoxide
Classifying Compounds: Ionic vs. Covalent
Identifying Compound Types
A compound is ionic if the first element is a metal or a polyatomic ion.
A compound is covalent if the first element is a nonmetal.
Examples: K2O (ionic), N2O (covalent)
Lewis Structures for Molecules and Polyatomic Ions
Lewis Structures and the Octet Rule
Lewis structures represent molecules by showing the arrangement of valence electrons to give octets. Shared electrons (bonding pairs) are shown as two dots or a single line between atoms, while lone pairs are placed on the outside.
Octet rule: Atoms tend to lose, gain, or share electrons until they are surrounded by eight valence electrons.
Exceptions: Hydrogen requires only two electrons; elements like P, S, Cl, Br, and I can have expanded octets.
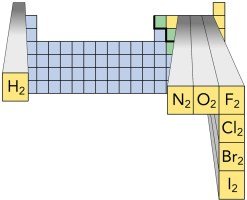
Elements That Exist as Diatomic Molecules
Certain elements exist naturally as diatomic molecules, meaning two atoms are bonded together.
Examples: H2, N2, O2, F2, Cl2, Br2, I2
Drawing Lewis Structures: Steps
Sum up the valence electrons, accounting for ions.
Write the symbols for each atom and draw single bonds between connected atoms (most metallic atom in center; H always outside).
Complete octets around all atoms using unshared pairs.
Place leftover electrons on the central atom.
If extra electrons are needed, try multiple bonds (double or triple).
Double and Triple Bonds
Double bond: Two pairs of electrons shared.
Triple bond: Three pairs of electrons shared.
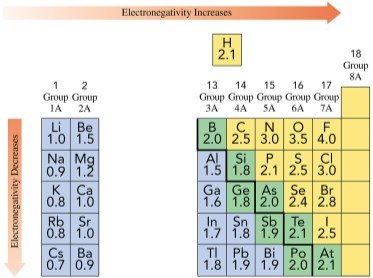
Electronegativity and Bond Polarity
Electronegativity Trends
Electronegativity is the ability of an atom to attract shared electrons in a chemical bond.
Increases from left to right across a period.
Decreases down a group.
Highest for nonmetals (fluorine is the highest).
Lowest for metals.
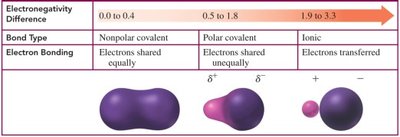
Bond Polarity and Electronegativity Difference
The difference in electronegativity between two atoms determines the bond type:
0.0–0.4: Nonpolar covalent (electrons shared equally)
0.5–1.8: Polar covalent (electrons shared unequally)
1.9–3.3: Ionic (electrons transferred)
Shapes and Polarity of Molecules
VSEPR Theory and Molecular Geometry
Valence Shell Electron-Pair Repulsion (VSEPR) theory describes the orientation of electron groups around the central atom to minimize repulsion. The shape is determined by the number of atoms and lone pairs attached to the central atom.
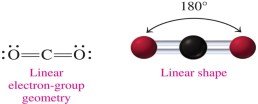
Linear: 2 electron groups, bond angle 180° (e.g., CO2)
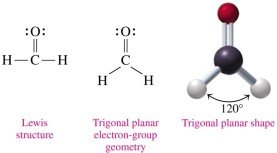
Trigonal planar: 3 electron groups, bond angle 120° (e.g., H2CO)
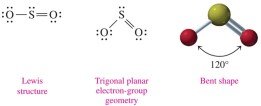
Bent: 3 electron groups (2 bonds, 1 lone pair), bond angle 120° (e.g., SO2)
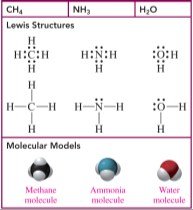
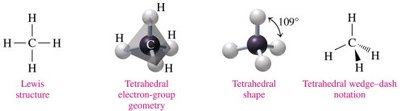
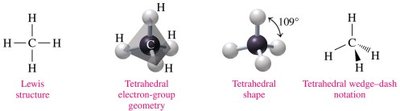
Tetrahedral: 4 electron groups, bond angle 109° (e.g., CH4)
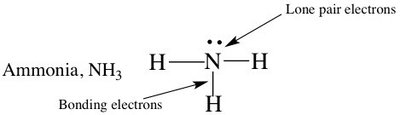
Trigonal pyramidal: 4 electron groups (3 bonds, 1 lone pair), bond angle 109° (e.g., NH3)
Bent: 4 electron groups (2 bonds, 2 lone pairs), bond angle 109° (e.g., H2O)
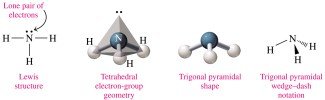
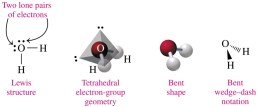
Polarity of Molecules
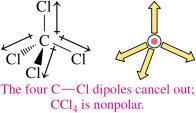
Nonpolar molecules: Contain nonpolar bonds or polar bonds that cancel in a symmetrical arrangement (e.g., CO2, CCl4).
Polar molecules: Contain polar bonds that do not cancel, resulting in a partial negative charge on one end (e.g., H2O, NH3, HCl).
If the central atom is symmetrically surrounded by atoms of the same element, the molecule is nonpolar.
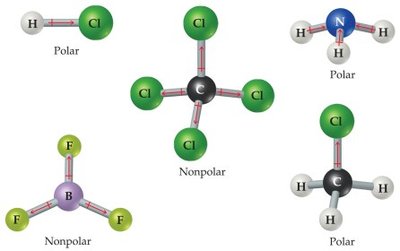
Summary Table: Molecular Shapes and Polarity
Linear: CO2 (nonpolar)
Trigonal planar: H2CO (polar), BF3 (nonpolar)
Bent: SO2 (polar), H2O (polar)
Tetrahedral: CH4 (nonpolar), CCl4 (nonpolar)
Trigonal pyramidal: NH3 (polar)
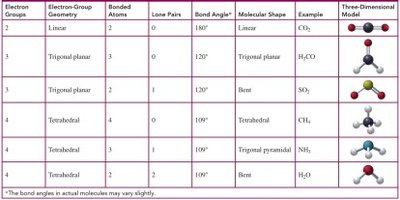
Electron Groups | Geometry | Bonded Atoms | Lone Pairs | Bond Angle | Shape | Example |
|---|---|---|---|---|---|---|
2 | Linear | 2 | 0 | 180° | Linear | CO2 |
3 | Trigonal planar | 3 | 0 | 120° | Trigonal planar | H2CO |
3 | Trigonal planar | 2 | 1 | 120° | Bent | SO2 |
4 | Tetrahedral | 4 | 0 | 109° | Tetrahedral | CH4 |
4 | Tetrahedral | 3 | 1 | 109° | Trigonal pyramidal | NH3 |
4 | Tetrahedral | 2 | 2 | 109° | Bent | H2O |
Key Takeaways
Molecular compounds are formed by sharing electrons between nonmetals.
Lewis structures help visualize electron arrangement and bonding.
Electronegativity differences determine bond type and polarity.
VSEPR theory predicts molecular shapes based on electron group repulsion.
Molecular polarity depends on bond polarity and molecular geometry.
Additional info: Expanded explanations and context were added to clarify naming conventions, Lewis structure steps, and VSEPR theory for self-contained study notes.
