 Back
BackMolecular Compounds: Structure, Bonding, and Polarity
Study Guide - Smart Notes

6.5 Molecular Compounds: Sharing Electrons
Introduction to Molecular Compounds
Molecular compounds are formed when two or more nonmetal atoms share electrons, resulting in covalent bonds. These compounds are distinct from ionic compounds, which involve the transfer of electrons between metals and nonmetals. The sharing of valence electrons allows each atom to achieve a stable electron configuration, often resembling that of the nearest noble gas.
Covalent bond: A chemical bond formed by the sharing of one or more pairs of electrons between atoms.
Molecule: A discrete group of atoms held together by covalent bonds in a definite proportion.
Naming Molecular Compounds
The names of molecular compounds require prefixes to indicate the number of atoms of each element present, as several different compounds can be formed from the same two nonmetals. The first nonmetal is named by its element name, and the second nonmetal uses the first syllable of its name followed by ide. Prefixes are used to indicate the number of atoms, except that mono is never used for the first element.
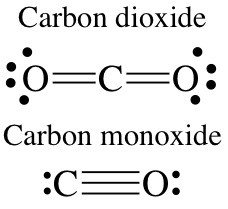
Examples: CO2 is carbon dioxide; CO is carbon monoxide; NCl3 is nitrogen trichloride.
When vowels "o" and "o" or "a" and "o" appear together, the first vowel is omitted (e.g., monoxide, not monooxide).
Prefix | Number |
|---|---|
mono- | 1 |
di- | 2 |
tri- | 3 |
tetra- | 4 |
penta- | 5 |
hexa- | 6 |
hepta- | 7 |
octa- | 8 |
nona- | 9 |
deca- | 10 |
Writing Formulas from Names
To write the formula of a molecular compound from its name, use the symbol for each element and a subscript if a prefix indicates two or more atoms. For example, diphosphorus pentoxide is P2O5.
Identifying Ionic vs. Covalent Compounds
A compound is usually ionic if the first element is a metal or a polyatomic ion, and covalent if the first element is a nonmetal. For example, K2O (potassium oxide) is ionic, while N2O (dinitrogen oxide) is covalent.
6.6 Lewis Structures for Molecules and Polyatomic Ions
Lewis Structures and the Octet Rule
A Lewis structure represents the arrangement of valence electrons in a molecule. Atoms tend to gain, lose, or share electrons to achieve an octet, which is the stable electron configuration of noble gases. Shared electrons (bonding pairs) are shown as lines or pairs of dots between atoms, while nonbonding pairs (lone pairs) are placed on the outside.
Diatomic Molecules
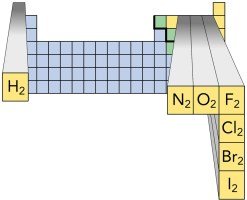
Certain elements exist naturally as diatomic molecules, meaning they are found as pairs of atoms bonded together. These include hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and iodine.
Drawing Lewis Structures
Sum the valence electrons, accounting for ions if present.
Write the symbols for each atom and connect them with single bonds. The most metallic atom is usually central (except H, which is always terminal).
Complete octets around outer atoms first, then the central atom.
Place any leftover electrons on the central atom.
If the central atom lacks an octet, form double or triple bonds as needed.
Lewis structures help visualize the arrangement of atoms and electrons in a molecule.
Exceptions to the Octet Rule
Hydrogen requires only two electrons (a duet).
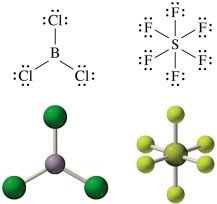
Elements such as P, S, Cl, Br, and I can have expanded octets (10 or 12 electrons).
Example: In SF6, sulfur has 12 valence electrons.
6.7 Electronegativity and Bond Polarity
Electronegativity
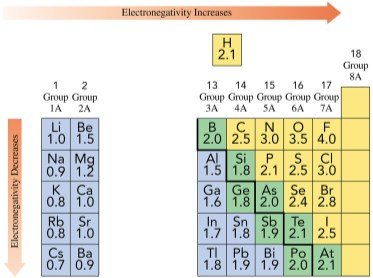
Electronegativity is the ability of an atom to attract shared electrons in a chemical bond. It increases across a period (left to right) and decreases down a group (top to bottom) in the periodic table. Nonmetals have high electronegativity, with fluorine being the highest, while metals have low electronegativity.
Bond Polarity
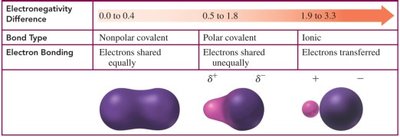
The difference in electronegativity between two atoms determines the polarity of the bond:
Nonpolar covalent bond: Electrons are shared equally (difference 0.0–0.4).
Polar covalent bond: Electrons are shared unequally (difference 0.5–1.8).
Ionic bond: Electrons are transferred (difference >1.8).
6.8 Shapes and Polarity of Molecules
VSEPR Theory and Molecular Geometry
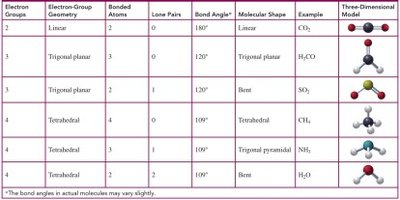
Valence Shell Electron-Pair Repulsion (VSEPR) theory states that electron groups around a central atom are arranged as far apart as possible to minimize repulsion. The shape of a molecule is determined by the number of electron groups and bonded atoms around the central atom.
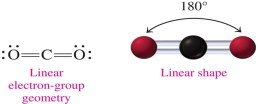
Linear: 2 electron groups, 180° bond angle (e.g., CO2).
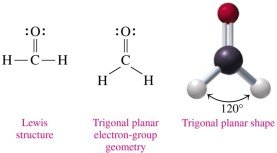
Trigonal planar: 3 electron groups, 120° bond angle (e.g., H2CO).
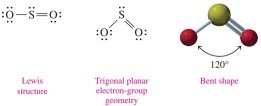
Bent: 3 electron groups (2 bonds, 1 lone pair), 120° bond angle (e.g., SO2).
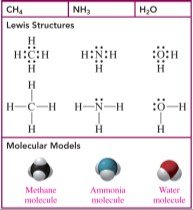
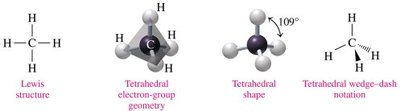
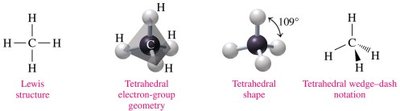
Tetrahedral: 4 electron groups, 109° bond angle (e.g., CH4).
Trigonal pyramidal: 4 electron groups (3 bonds, 1 lone pair), 109° bond angle (e.g., NH3).
Bent (tetrahedral): 4 electron groups (2 bonds, 2 lone pairs), 109° bond angle (e.g., H2O).
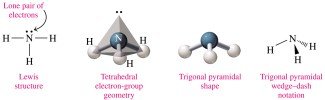
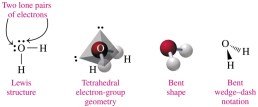
Polarity of Molecules
A molecule is nonpolar if it contains only nonpolar bonds or if the polar bonds are arranged symmetrically so that their dipoles cancel (e.g., CO2, CCl4). A molecule is polar if it contains polar bonds that do not cancel due to the shape of the molecule (e.g., H2O, NH3).
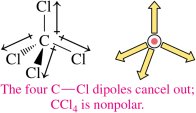
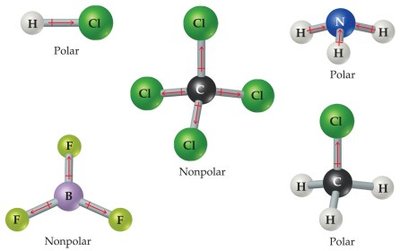
If the central atom is symmetrically surrounded by atoms of the same element, the molecule is nonpolar.
If the dipoles do not cancel, the molecule is polar.
Example: OF2 is polar because the O–F bonds are polar covalent and the bent shape prevents dipole cancellation.
Additional info: The study of molecular compounds, their naming, Lewis structures, bond polarity, and molecular geometry is essential for understanding chemical reactivity, physical properties, and biological functions of molecules.
