 Back
BackMonosaccharides, Disaccharides, and Polysaccharides: Structure, Reactions, and Biological Importance
Study Guide - Smart Notes

Monosaccharides: Oxidation and Reduction
Oxidation of Monosaccharides
Monosaccharides, such as glucose, can exist in an open-chain form that contains an aldehyde group. This aldehyde group can be oxidized to a carboxylic acid by oxidizing agents like Benedict’s solution. The process is important for identifying reducing sugars in laboratory tests.
Oxidation is the loss of electrons (LEO: Loss of Electrons is Oxidation).
Reducing sugars are carbohydrates that can reduce other substances, typically due to the presence of a free aldehyde or ketone group.
When oxidized, the aldehyde group of glucose forms a sugar acid, named by replacing the “-ose” ending with “-onic acid” (e.g., D-gluconic acid).
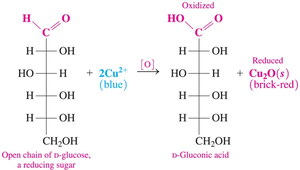
Example: The open-chain form of D-glucose reacts with Cu2+ (blue) to produce D-gluconic acid and Cu2O (brick-red).
Oxidation of Fructose
Although fructose is a ketose and typically resists oxidation, it can be oxidized in basic solutions like Benedict’s reagent. This occurs via rearrangement to glucose, which then undergoes oxidation.
Fructose (a ketohexose) rearranges to form glucose (an aldohexose), which can then be oxidized.
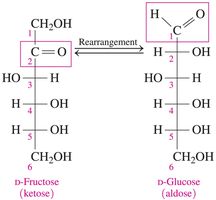
Example: In Benedict’s solution, fructose is converted to glucose, which is then oxidized.
Reduction of Monosaccharides
Reduction of the carbonyl group in monosaccharides produces sugar alcohols (alditols). This process is used industrially to create sweeteners.
Reduction is the gain of electrons (GER: Gain of Electrons is Reduction).
Sugar alcohols are named by replacing “-ose” with “-itol” (e.g., D-glucose → D-sorbitol).
Common sugar alcohols include D-sorbitol, D-xylitol, and D-mannitol.
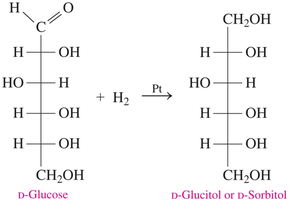
Example: D-glucose is reduced by hydrogen (H2) in the presence of platinum to form D-glucitol (D-sorbitol).
Health Link: Glucosuria
Glucosuria is the presence of glucose in urine, which occurs when blood glucose levels are too high for the kidneys to reabsorb all the glucose. This condition can be detected using urine test strips containing specific dyes and enzymes.
Test strips use glucose oxidase and peroxidase to detect glucose levels.
The intensity of the color change on the strip correlates with glucose concentration.
Example: Glucosuria is commonly screened for in diabetes management.
Disaccharides: Structure and Linkages
Formation of Disaccharides
Disaccharides are carbohydrates composed of two monosaccharide units joined by a glycosidic bond, formed through a dehydration reaction (loss of water).
Common disaccharides include maltose, lactose, and sucrose.
The type of glycosidic bond (e.g., α-1,4 or β-1,4) determines the properties of the disaccharide.
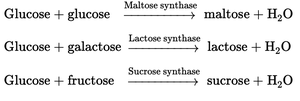
Maltose
Maltose consists of two glucose units linked by an α-1,4-glycosidic bond. It is produced during the hydrolysis of starch and is found in cereals, candies, and brewing.
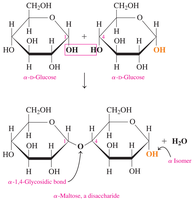
Lactose
Lactose is composed of glucose and galactose joined by a β-1,4-glycosidic bond. It is the primary sugar in milk.
Human milk contains 6–8% lactose; cow’s milk contains 4–5%.
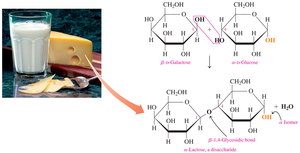
Sucrose
Sucrose, or table sugar, is made of glucose and fructose linked by an α,β-1,2-glycosidic bond. It is not a reducing sugar and does not react with Benedict’s reagent.
Sweeteners and Sweetness
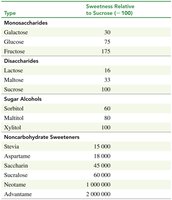
The relative sweetness of sugars and artificial sweeteners is compared to sucrose (assigned a value of 100). Artificial sweeteners can be much sweeter than sucrose and are used as sugar substitutes in foods and drinks.
Type | Sweetness Relative to Sucrose (=100) |
|---|---|
Galactose | 30 |
Glucose | 75 |
Fructose | 175 |
Lactose | 16 |
Maltose | 33 |
Sucrose | 100 |
Sorbitol | 60 |
Maltitol | 80 |
Xylitol | 100 |
Stevia | 15,000 |
Aspartame | 18,000 |
Saccharin | 45,000 |
Sucralose | 40,000 |
Neotame | 1,000,000 |
Acesulfame | 2,000,000 |
Artificial Sweeteners
Artificial sweeteners are compounds used to replace sugar in foods and beverages. They are often much sweeter than sucrose and may have different chemical structures.
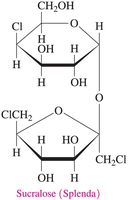
Sucralose (Splenda): Derived from sucrose by replacing some hydroxyl groups with chlorine atoms.
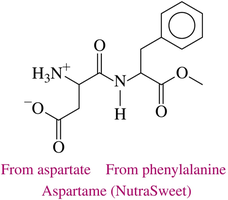
Aspartame (NutraSweet, Equal): A non-carbohydrate sweetener made from aspartate and phenylalanine.
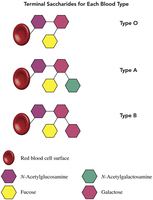
Biological Link: Blood Types and Saccharides
Blood types (A, B, AB, O) are determined by the terminal saccharides attached to the surface of red blood cells. These saccharides act as antigens and are critical for blood transfusion compatibility.
Polysaccharides: Structure and Function
Polysaccharides are large polymers formed by joining many monosaccharide units. They serve as energy storage (starch, glycogen) or structural components (cellulose) in living organisms.
All major polysaccharides discussed here are polymers of α-D-glucose.
Examples include amylose, amylopectin, glycogen, and cellulose.
Glycogen
Glycogen is a highly branched polymer of glucose stored in the liver and muscles of animals. It is hydrolyzed to maintain blood glucose levels and provide energy between meals.
Glucose units are joined by α-1,4-glycosidic bonds, with branches formed by α-1,6-glycosidic bonds.
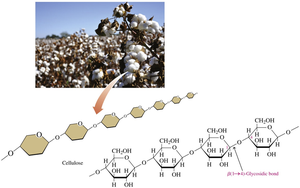
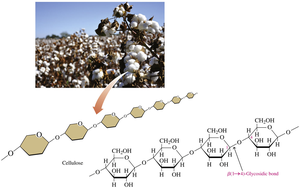
Cellulose
Cellulose is a major structural polysaccharide in plants and wood. It consists of glucose units linked by β-1,4-glycosidic bonds, making it insoluble in water and resistant to hydrolysis. Humans cannot digest cellulose due to the inability to break β-1,4-glycosidic bonds.
Provides rigidity to plant cell walls.
Indigestible by humans, but important as dietary fiber.
Summary
This guide covers the oxidation and reduction of monosaccharides, the structure and formation of disaccharides, the properties and uses of polysaccharides, and the biological significance of carbohydrates in health and disease. Understanding these concepts is essential for further study in biochemistry and health sciences.
