 Back
BackNames, Formulas, and Properties of Chemical Compounds: Study Guide
Study Guide - Smart Notes

Names & Formulas of Chemical Compounds
Overview
This section introduces the nomenclature and classification of inorganic molecular and ionic compounds, focusing on their naming conventions, formulas, and the underlying chemical principles. Understanding these concepts is essential for identifying, describing, and predicting the behavior of chemical substances.
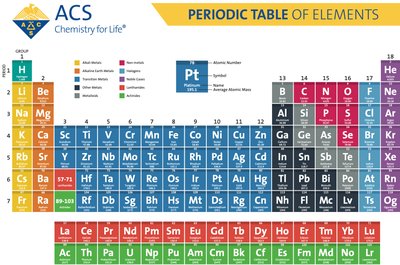
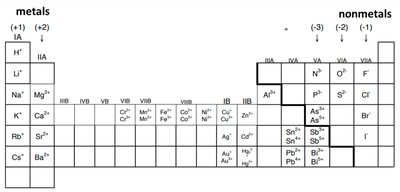
The Periodic Table of Elements
Structure and Organization
The Periodic Table of Elements is a systematic arrangement of all known chemical elements. Elements are organized by increasing atomic number, which corresponds to the number of protons in the nucleus. The table is divided into groups (columns) and periods (rows), reflecting similarities in electron configuration and chemical properties.
Groups: Vertical columns; elements in the same group share similar chemical properties.
Periods: Horizontal rows; elements in the same period have the same number of electron shells.
Element Symbol: One or two letters, first capitalized, second lowercase (e.g., Na for sodium).
Atomic Number: Unique integer for each element, equal to the number of protons.
Molar Mass: Average mass per mole of an element, expressed in grams per mole (g/mol).
Families: Groups of elements with similar chemical properties (e.g., alkali metals, halogens).
Diatomic Elements
Certain elements exist naturally as diatomic molecules, meaning they are composed of two atoms. These include:
H2, N2, O2, F2, Cl2, Br2, I2
These elements are always found as pairs in their natural state.
Atoms & Ions
Atomic Structure
An atom consists of a nucleus containing protons (positively charged) and neutrons (neutral), surrounded by electrons (negatively charged) in orbitals. The number of protons defines the element, while the number of neutrons can vary, resulting in isotopes.
Proton (p+): Charge +1
Neutron (n): Charge 0
Electron (e-): Charge -1
Isotopes are atoms of the same element with different numbers of neutrons. The Mass Number is the sum of protons and neutrons.
Ions
Ions are atoms or molecules with a net electric charge due to the loss or gain of electrons:
Cation: Fewer electrons than protons; positively charged (e.g., Li+).
Anion: More electrons than protons; negatively charged (e.g., O2−).
Metal atoms generally form cations; non-metal atoms generally form anions.
Molecular & Ionic Compounds
Types of Compounds
Compounds are substances formed by the chemical combination of two or more elements. They are classified as:
Molecular (Covalent) Compounds: Formed by sharing electrons between nonmetals, resulting in discrete molecules (e.g., H2O).
Ionic Compounds (Salts): Formed by electrostatic attraction between cations and anions, resulting in a continuous lattice structure (e.g., NaCl).
The simplest representation of an ionic compound is called a Formula Unit.
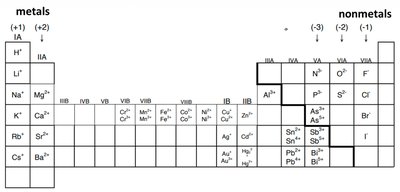
Determining the Charge of Ions
Periodic Table and Ionic Charges
The periodic table helps predict the charges of ions formed by elements:
Main Group Elements: Charges progress as 1+, 2+, 3+, variable, then 3-, 2-, 1- across the table.
Transition Metals: Often have variable charges, indicated by Roman numerals (e.g., Nickel (II) Chloride, NiCl2).
Algebraic Determination: The sum of all charges in a compound must equal zero.
Example: For Fe2O3:
Polyatomic Ions
Definition and Examples
Polyatomic ions are ions composed of multiple atoms bonded together, carrying a net charge. Their charges are not always predictable from the constituent atoms and must be memorized or determined algebraically.
Example: Ca(CH3COO)2 contains two acetate ions, each with a charge of -1.
Example: NaHCO3 (baking soda) contains the bicarbonate ion, HCO3-.
Common Polyatomic Ions
Name of Ion | Formula | Overall Charge |
|---|---|---|
Acetate | CH3COO- | 1- |
Ammonium | NH4+ | 1+ |
Bicarbonate | HCO3- | 1- |
Carbonate | CO32- | 2- |
Chlorite | ClO2- | 1- |
Chromate | CrO42- | 2- |
Cyanide | CN- | 1- |
Dihydrogen Phosphate | H2PO4- | 1- |
Hydrogen Phosphate | HPO42- | 2- |
Hydroxide | OH- | 1- |
Hypochlorite | ClO- | 1- |
Iodate | IO3- | 1- |
Nitrate | NO3- | 1- |
Perbromate | BrO4- | 1- |
Perchlorate | ClO3- | 1- |
Phosphate | PO43- | 3- |
Sulfate | SO42- | 2- |
Calculating Molar Mass
Definition and Calculation
Molar Mass (also called molecular weight or formula weight) is the mass of one mole of a substance, expressed in grams per mole (g/mol). One mole contains 6.022 \times 10^{23} particles (Avogadro's number). Molar mass is distinct from mass number, which is used for isotopes.
C: 12.011 g/mol
H2O: g/mol
CH3COOH: g/mol
Ca3(PO4)2: g/mol
Naming Molecular Compounds
Rules and Prefixes
Molecular compounds are named using specific rules:
Name the two elements; the element farther from fluorine goes first.
Change the ending of the second element to "-ide".
Use prefixes to indicate the number of each atom (omit "mono" for the first element).
Prefix | Number of Atoms |
|---|---|
Mono | 1 |
Di | 2 |
Tri | 3 |
Tetra | 4 |
Penta | 5 |
Hexa | 6 |
Hepta | 7 |
Octa | 8 |
Nona | 9 |
Deca | 10 |
Examples:
CO: Carbon monoxide
CO2: Carbon dioxide
N2O4: Dinitrogen tetraoxide
Cl2O: Dichlorine monoxide
Naming Ionic Compounds
Rules and Examples
Ionic compounds are named by listing the metal ion (or ammonium ion) first, followed by the nonmetal. The ending of the nonmetal is changed to "-ide". Polyatomic ions retain their names. Prefixes are not used. For transition metals, the charge is indicated by Roman numerals in parentheses.
CaO: Calcium oxide
AlBr3: Aluminum bromide
SnI4: Tin (IV) iodide
CuSO4: Copper (II) sulfate
Li2CrO4: Lithium chromate
Cr2(CO3)3: Chromium (III) carbonate
Instructions for Lab and Mass Number
Mass Number vs. Molar Mass
Mass Number is the sum of protons and neutrons in an atom and is used to distinguish isotopes. Molar Mass is the mass of one mole of a substance and is used in chemical calculations.
Notation: where X is the atomic symbol, A is the mass number, and Z is the atomic number.
Example: denotes lithium with 3 protons and a mass number of 7.
Additional info: The study notes have been expanded with academic context to ensure completeness and clarity for exam preparation.
