Back
BackOrganic Reaction Types and Alkene Chemistry: Addition, Substitution, and Polymerization
Study Guide - Smart Notes

Types of Organic Reactions
Overview of Organic Reaction Types
Organic chemistry features several fundamental reaction types that govern the transformation of molecules. The four main types are addition, elimination, substitution, and rearrangement reactions. These reactions are central to the synthesis of pharmaceuticals, plastics, and other industrial chemicals.
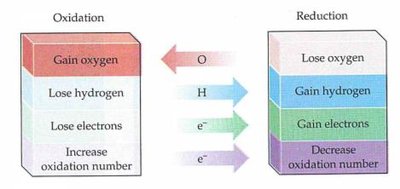
Addition Reaction: A process where atoms or groups are added to a molecule, typically across a multiple bond, resulting in a saturated product.
Elimination Reaction: Atoms or groups are removed from a molecule, often resulting in the formation of a double or triple bond.
Substitution Reaction: One atom or group in a molecule is replaced by another atom or group.
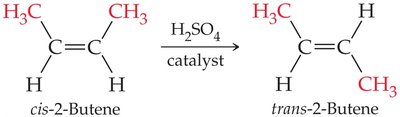
Rearrangement Reaction: The structure of a molecule is reorganized to form an isomer.
Example: Rearrangement of cis-2-butene to trans-2-butene is a classic rearrangement reaction.
Addition Reactions of Alkenes
General Mechanism and Examples
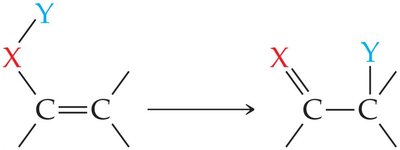
Addition reactions are especially important for alkenes and alkynes, where the multiple bonds serve as reactive sites. In these reactions, a molecule X–Y adds across the double or triple bond, converting it to a single bond and yielding a saturated product.
Symmetrical Addition: Both atoms added are the same (e.g., H2, Cl2).
Unsymmetrical Addition: Different atoms or groups are added (e.g., H–Cl, H–OH).
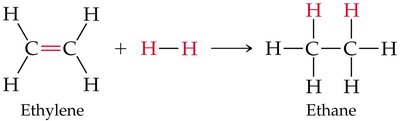
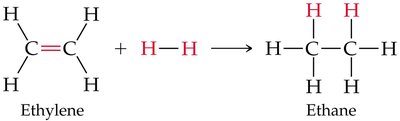
Example: Hydrogenation of ethylene to ethane is a symmetrical addition and also a reduction reaction.
Hydrogenation of Alkenes and Alkynes
Hydrogenation is the addition of hydrogen (H2) to alkenes or alkynes in the presence of a metal catalyst (such as Pd, Pt, or Ni), converting them to alkanes. This reaction is widely used in industry, for example, in the production of margarine from vegetable oils.
General Equation:
Commercial Application: Used to convert unsaturated fats to saturated fats, but can produce trans fatty acids.
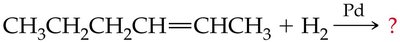
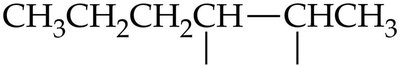

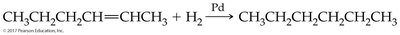
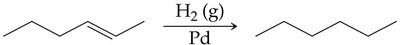
Halogenation of Alkenes and Alkynes
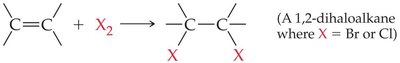
Halogenation involves the addition of halogens (Cl2, Br2) to alkenes or alkynes, forming 1,2-dihaloalkanes. This reaction is symmetrical and is used in the production of important polymers such as PVC.
General Equation:
Test for Unsaturation: Bromine water decolorizes in the presence of multiple bonds.


Hydrohalogenation: Addition of HBr and HCl to Alkenes
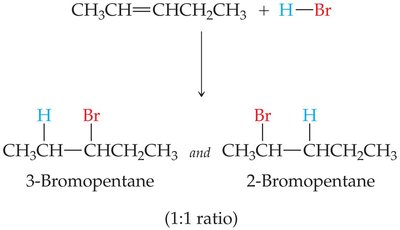
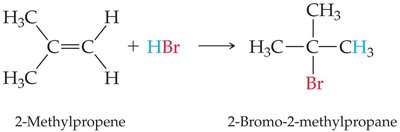
Alkenes react with hydrogen halides (HBr, HCl) to form alkyl halides. The product distribution depends on the symmetry of the alkene.
Symmetrical Alkenes: Both possible products are formed in equal amounts.
Unsymmetrical Alkenes: Markovnikov's rule applies: the hydrogen attaches to the carbon with more hydrogens, and the halide attaches to the carbon with fewer hydrogens.
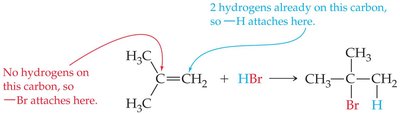
Worked Examples: Addition and Rearrangement Reactions
Propene Addition and Rearrangement
When propene reacts with HCl, two products are possible, but the major product follows Markovnikov's rule. Rearrangement reactions can convert cis-2-butene to trans-2-butene, changing the geometry but not the connectivity.

Substitution Reactions of Alkanes
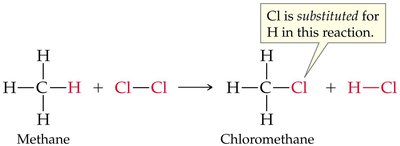
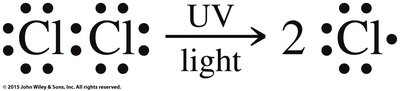
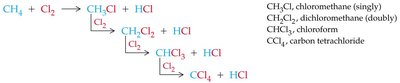
Halogenation of Alkanes
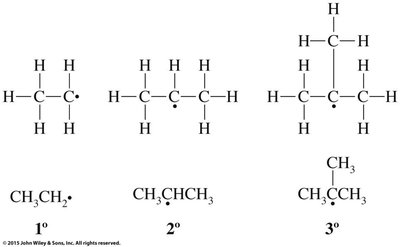
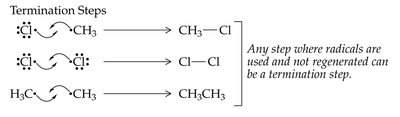
Halogenation of alkanes is a substitution reaction where a hydrogen atom is replaced by a halogen atom, typically initiated by heat or light. This process is called free radical halogenation and proceeds via initiation, propagation, and termination steps.
General Equation:
Free Radical Mechanism: Involves the formation and reaction of highly reactive free radicals.
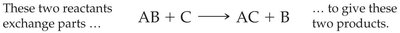

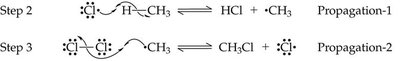


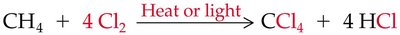
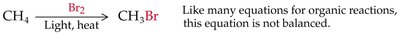
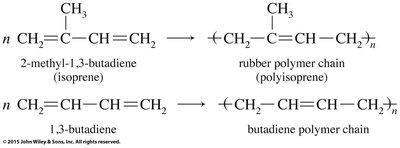
Polymerization of Alkenes
Chain-Growth Polymerization
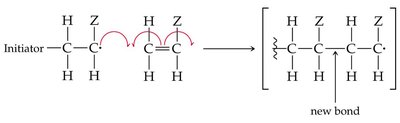
Alkenes can undergo polymerization to form large molecules called polymers. In chain-growth polymerization, a reactive intermediate (radical) adds to a monomer, and the process repeats, forming a long chain.
Monomer: The small, repeating unit (e.g., ethylene, propylene, styrene).
Polymer: The large molecule formed by linking many monomers.
Initiation, Propagation, Termination: The three steps of radical polymerization.
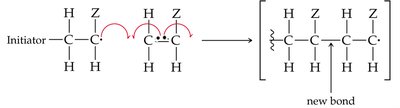
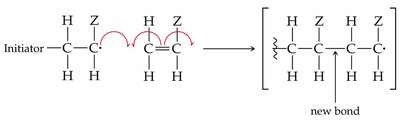
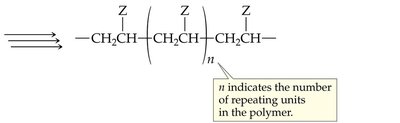
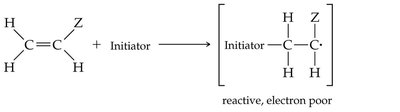
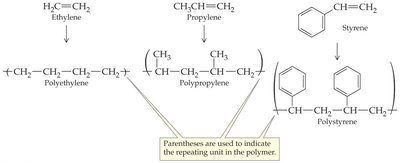
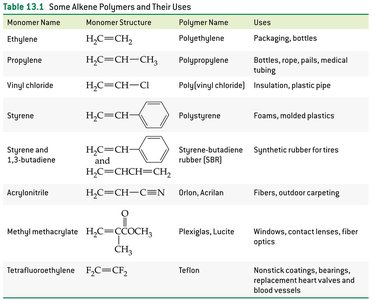


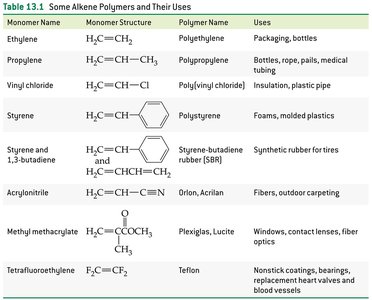
Common Alkene Polymers and Their Uses
Alkene polymers are widely used in everyday materials. The table below summarizes some common alkene polymers, their monomers, and uses.
Monomer Name | Monomer Structure | Polymer Name | Uses |
|---|---|---|---|
Ethylene | H2C=CH2 | Polyethylene | Packaging, bottles |
Propylene | H2C=CHCH3 | Polypropylene | Bottles, rope, pails, medical tubing |
Vinyl chloride | H2C=CHCl | Poly(vinyl chloride) | Insulation, plastic pipe |
Styrene |
| Polystyrene | Foams, molded plastics |
Styrene and 1,3-butadiene | H2C=CHCH=CH2 | Styrene-butadiene rubber (SBR) | Synthetic rubber for tires |
Acrylonitrile | H2C=CHCN | Orlon, Acrilan | Fibers, outdoor carpeting |
Methyl methacrylate | H2C=C(CH3)COOCH3 | Plexiglas, Lucite | Windows, contact lenses, fiber optics |
Tetrafluoroethylene | F2C=CF2 | Teflon | Nonstick coatings, bearings, replacement heart valves and blood vessels |
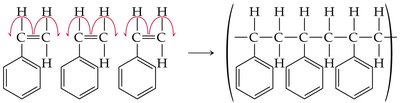
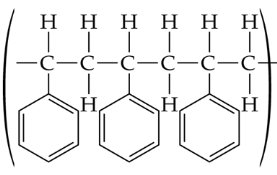
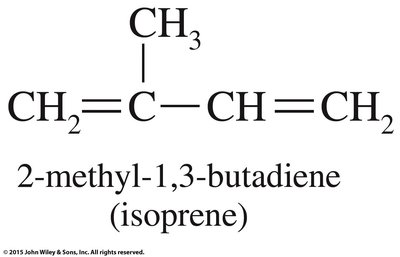
Diene Polymers and Geometric Isomerism
Diene polymers, such as natural rubber (polyisoprene) and synthetic rubber (polybutadiene), contain double bonds in their polymer chains, allowing for cis/trans isomerism. The geometric configuration affects the physical properties of the polymer.
Cis-Polyisoprene: Kinked, soft, elastic (natural rubber).
Trans-Polyisoprene: Linear, tough, nonelastic (gutta-percha).
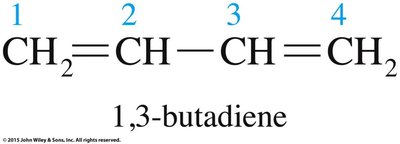
Aromatic Compounds: Structure and Nomenclature
Benzene and Aromaticity
Aromatic compounds, such as benzene, have unique stability due to delocalized pi electrons in a conjugated ring system. Benzene is flat, symmetrical, and exhibits resonance, making it less reactive than typical alkenes.
Molecular Formula: C6H6
Resonance: Each carbon-carbon bond is intermediate between a single and double bond.
Naming Aromatic Compounds
Substituted benzenes are named using the parent name "benzene." For disubstituted benzenes, the positions are described as ortho (o-), meta (m-), or para (p-).
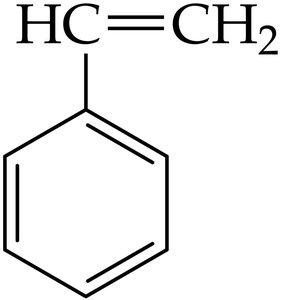
Common Names: Methylbenzene (toluene), hydroxybenzene (phenol), aminobenzene (aniline).
Phenyl Group: Benzene ring as a substituent is called "phenyl."
Reactions of Aromatic Compounds
Electrophilic Aromatic Substitution (EAS)
Aromatic compounds typically undergo substitution reactions rather than addition. In EAS, an electrophile replaces a hydrogen atom on the aromatic ring. Common EAS reactions include halogenation, nitration, and sulfonation.
Halogenation: Substitution of a halogen (–X) for a hydrogen.
Nitration: Substitution of a nitro group (–NO2).
Sulfonation: Substitution of a sulfonic acid group (–SO3H).
Example: Benzene reacts with Br2 (in the presence of a catalyst) to form bromobenzene.
Additional info: This guide expands on the brief points in the source material, providing definitions, examples, and context for each reaction type, as well as the industrial and biological relevance of polymers and aromatic compounds.