 Back
BackOrganic Reactions: Condensation, Hydrolysis, and Addition Reactions
Study Guide - Smart Notes

Organic Reactions: Condensation and Hydrolysis
Condensation Reactions
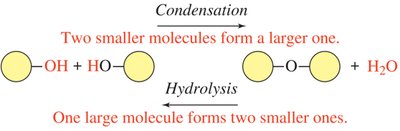
Condensation reactions are fundamental processes in organic and biological chemistry where two smaller molecules combine to form a larger molecule, with the elimination of a small molecule, typically water (H2O).
Definition: A reaction in which two molecules join together, releasing water as a byproduct.
Functional Groups Involved: Typically involves an –H from one molecule and an –OH from another, forming water.
Biological Importance: Condensation reactions are essential for the formation of many biomolecules, such as proteins, nucleic acids, and lipids.
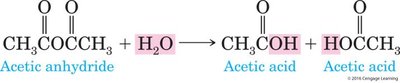
Example: Formation of esters, amides, and anhydrides from carboxylic acids and other functional groups.
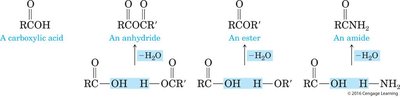
Equation Example:
Hydrolysis Reactions
Hydrolysis is the reverse of condensation. It involves the addition of water to a molecule, resulting in the cleavage of a covalent bond and the formation of two smaller molecules.
Definition: A reaction in which water is used to break a bond in a larger molecule, splitting it into two smaller molecules.
Functional Groups Involved: Commonly breaks esters, amides, and anhydrides into their component acids and alcohols/amines.
Biological Importance: Hydrolysis is crucial for digestion and metabolism, breaking down macromolecules into absorbable units.
Example: Hydrolysis of esters yields a carboxylic acid and an alcohol.
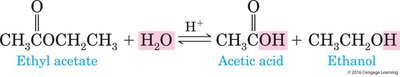
Equation Example:
Condensation and Hydrolysis in Biochemistry
These reactions are central to the synthesis and breakdown of biomolecules. For example, the hydrolysis of ATP to ADP releases energy for cellular processes.
ATP Hydrolysis:
Condensation in Nucleic Acids: Nucleotides are joined by condensation reactions to form DNA and RNA.
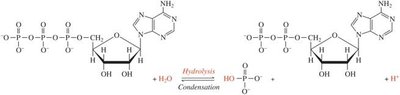
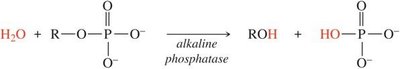
Addition and Removal of Phosphate Groups
The addition (phosphorylation) and removal (dephosphorylation) of phosphate groups are key regulatory mechanisms in cells. These are catalyzed by kinases (condensation) and phosphatases (hydrolysis).
Phosphorylation: Addition of phosphate group, usually from ATP, to a molecule.
Dephosphorylation: Removal of a phosphate group by hydrolysis.
Enzymes: Kinases (add phosphate), phosphatases (remove phosphate).
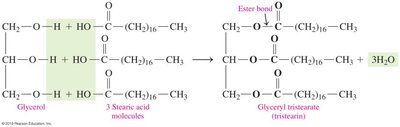
Formation and Hydrolysis of Lipids
Triacylglycerols (Triglycerides)
Triacylglycerols are formed by the condensation of glycerol and three fatty acids, resulting in the formation of ester bonds and the release of water.
Condensation Reaction: Glycerol (a triol) reacts with three fatty acids to form a triacylglycerol and three water molecules.
Biological Role: Triacylglycerols are the main storage form of energy in animals.
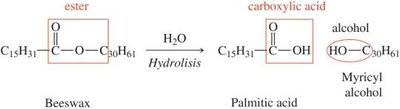
Hydrolyzable and Nonhydrolyzable Lipids
Lipids can be classified based on their ability to undergo hydrolysis:
Hydrolyzable Lipids: Contain ester bonds that can be broken by hydrolysis (e.g., fats, oils, waxes).
Nonhydrolyzable Lipids: Do not contain hydrolyzable bonds (e.g., steroids, fat-soluble vitamins).
Organic Addition Reactions to Alkenes
General Addition to Alkenes
Addition reactions involve the addition of atoms or groups to the carbons of a double bond, converting it into single bonds. These reactions are common for alkenes and are often exergonic (energy-releasing).
Mechanism: The π bond of the alkene is broken, and two new single bonds are formed.
Types of Addition: Hydrogenation, hydration, halogenation, hydrohalogenation.
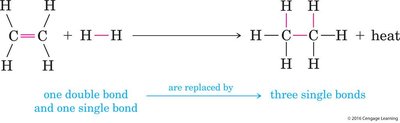
Hydrogenation of Alkenes
Hydrogenation is the addition of hydrogen (H2) across a double bond, converting an alkene to an alkane. This reaction typically requires a metal catalyst such as palladium (Pd) or platinum (Pt).
Equation:
Industrial Application: Used to convert unsaturated fats to saturated fats; partial hydrogenation can produce trans fats.
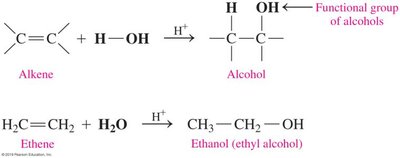
Hydration of Alkenes
Hydration is the addition of water (H-OH) to an alkene, producing an alcohol. This reaction is typically acid-catalyzed and follows Markovnikov's Rule for sufficient selectivity.
Equation:
Markovnikov's Rule: The hydrogen atom adds to the less substituted carbon, and the hydroxyl group adds to the more substituted carbon.
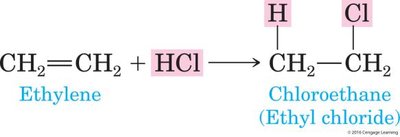
Addition of Halogens and Hydrogen Halides
Alkenes react with halogens (Cl2, Br2, I2) and hydrogen halides (HCl, HBr, HI) to form haloalkanes. The addition of HX follows Markovnikov's Rule.
Equation:
Product: Haloalkanes (alkyl halides).
Summary Table: Key Organic Reactions
Reaction Type | General Equation | Key Product(s) | Example |
|---|---|---|---|
Condensation | Ester + Water | Triacylglycerol formation | |
Hydrolysis | Acid + Alcohol | Digestion of fats | |
Hydrogenation | Alkane | Vegetable oil to margarine | |
Hydration | Alcohol | Ethanol from ethene | |
Halogenation | Dihaloalkane | Bromination of ethene | |
Hydrohalogenation | Haloalkane | Chloroethane from ethylene |
Additional info: Markovnikov's Rule is crucial for predicting the major product in addition reactions to asymmetrical alkenes. Condensation and hydrolysis reactions are central to metabolism and the synthesis of biological macromolecules.
