 Back
BackOrganic Reactions: Condensation, Hydrolysis, and Addition Reactions
Study Guide - Smart Notes

Organic Reactions: Condensation and Hydrolysis
Condensation Reactions
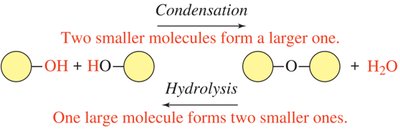
Condensation reactions are fundamental in organic chemistry and biochemistry, involving the joining of two smaller molecules to form a larger one, with the elimination of water. This process is essential for the synthesis of many biological macromolecules.
Definition: A condensation reaction is a chemical process where two molecules combine, releasing a molecule of water (H2O).
Functional Groups Involved: Typically occurs between groups containing an –H (hydrogen) and an –OH (hydroxyl).
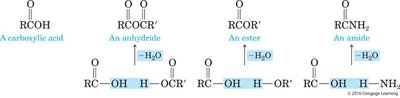
Examples: Formation of esters, amides, and anhydrides from carboxylic acids and alcohols or amines.
Hydrolysis Reactions
Hydrolysis is the reverse of condensation, breaking a covalent bond in a molecule by adding water, resulting in two smaller molecules. This reaction is crucial for the breakdown of macromolecules in biological systems.
Definition: Hydrolysis is the chemical decomposition of a molecule by the addition of water.
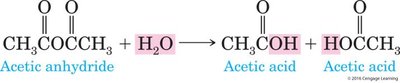
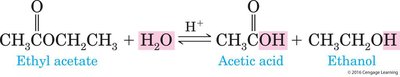
Types: Hydrolysis of anhydrides is relatively easy, while hydrolysis of esters requires a strong acid or base.
Biochemical Importance: Hydrolysis reactions are common in metabolism, such as the breakdown of ATP to ADP.
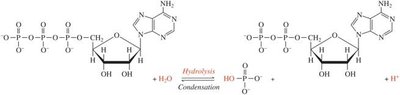
Condensation and Hydrolysis in Biochemistry
These reactions are central to the synthesis and degradation of biomolecules. For example, ATP hydrolysis provides energy for cellular processes, and condensation reactions form peptide bonds in proteins.
Enzymes: Kinases catalyze condensation (phosphorylation), while phosphatases catalyze hydrolysis (dephosphorylation).
Phosphate Group Addition/Removal: Regulates metabolic pathways.
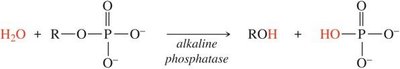
Formation and Hydrolysis of Lipids
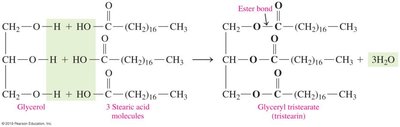
Triacylglycerols (Triglycerides)
Triacylglycerols are formed by the condensation of glycerol and three fatty acids, resulting in the formation of ester bonds and the release of water.
Definition: Triacylglycerols are esters formed from glycerol and fatty acids.
Biological Role: Serve as energy reserves in fats and oils.
Reaction: Condensation of -COOH (fatty acid) and -OH (glycerol) forms an ester linkage.
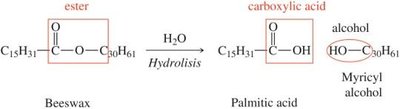
Hydrolyzable vs. Nonhydrolyzable Lipids
Lipids can be classified based on their ability to undergo hydrolysis.
Hydrolyzable Lipids: Contain ester groups and can be broken down into carboxylic acids and alcohols (e.g., waxes, fats, oils).
Nonhydrolyzable Lipids: Cannot be hydrolyzed (e.g., steroids).
Organic Addition Reactions to Alkenes
Addition Reactions
Addition reactions involve the addition of atoms or groups to a double bond in an organic molecule, converting the double bond to single bonds.
Definition: An addition reaction is a process where atoms are added to the carbons of a double bond, breaking the π bond and forming two new single bonds.
Stability: Most alkene addition reactions are exergonic, producing more stable products.
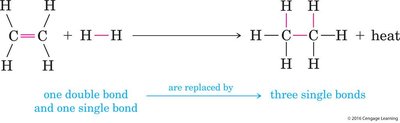
Hydrogenation of Alkenes
Hydrogenation is the addition of hydrogen to an alkene, converting it to an alkane.
Catalysts: Transition metals such as Pd and Pt are commonly used.
Industrial Application: Used to convert unsaturated fats to saturated fats; partial hydrogenation can produce trans fats.
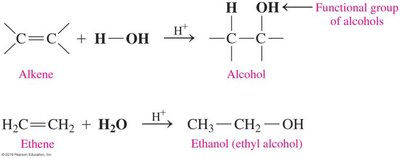
Hydration of Alkenes
Hydration involves the addition of water to an alkene, forming an alcohol. This reaction is typically acid-catalyzed and follows Markovnikov’s Rule.
Markovnikov’s Rule: The hydrogen atom adds to the less substituted carbon, and the hydroxyl group adds to the more substituted carbon.
Example: Hydration of ethene produces ethanol.
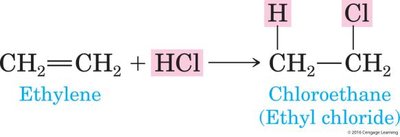
Addition of Halogens and Hydrogen Halides
Addition of halogens (Cl2, Br2, I2) or hydrogen halides (HCl, HBr, HI) to alkenes forms haloalkanes. These reactions also follow Markovnikov’s Rule.
Haloalkanes: Compounds formed by the addition of halogen atoms to alkenes.
Example: Addition of HCl to ethylene forms chloroethane.
Summary Table: Types of Organic Reactions
Reaction Type | Definition | Example |
|---|---|---|
Condensation | Joining of two molecules with loss of water | Ester formation |
Hydrolysis | Splitting of a molecule by addition of water | ATP hydrolysis |
Addition | Addition of atoms/groups to a double bond | Hydrogenation of alkenes |
Key Equations
Condensation: $\text{R-OH} + \text{HO-R'} \rightarrow \text{R-O-R'} + \text{H}_2\text{O}$
Hydrolysis: $\text{R-O-R'} + \text{H}_2\text{O} \rightarrow \text{R-OH} + \text{HO-R'}$
Hydrogenation: $\text{C=C} + \text{H}_2 \rightarrow \text{C-C}$
Hydration: $\text{C=C} + \text{H}_2\text{O} \xrightarrow{H^+} \text{C-C(OH)}$
Addition of Halogen: $\text{C=C} + \text{HX} \rightarrow \text{C-C(H)(X)}$
Practice Problems
Draw the structure for the principal product(s) formed when the following organic molecules are reduced or oxidized. Identify the functional groups.
Predict the product if the following molecules undergo complete hydrogenation in the presence of a Pt catalyst. Name the products.
Predict the major product for the hydration of the following alkene. What other reactant is required?
Predict the product(s) for the addition of HBr or HCl to the given alkenes. Which one is the major?
Additional info: Academic context and explanations were expanded for clarity and completeness.
