 Back
BackPolarity and Intermolecular Forces: Practice with Lewis Structures and Amino Acids
Study Guide - Smart Notes

Q15. Classify the molecules as polar or nonpolar.
Background
Topic: Molecular Polarity and Lewis Structures
This question tests your ability to analyze Lewis structures and determine whether a molecule is polar or nonpolar based on its shape and the distribution of electrons.
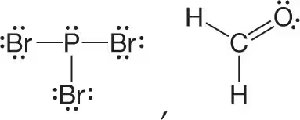
Key Terms and Concepts:
Polarity: A molecule is polar if it has a net dipole moment due to uneven distribution of electrons.
Nonpolar: A molecule is nonpolar if the electron distribution is symmetrical and there is no net dipole moment.
Lewis Structure: A diagram showing the arrangement of atoms and valence electrons in a molecule.
Electronegativity: The tendency of an atom to attract electrons in a bond.
Step-by-Step Guidance
Examine the Lewis structure of each molecule. Identify the central atom and the surrounding atoms.
Determine the molecular geometry (shape) using VSEPR theory for each molecule.
Assess the symmetry of the molecule. If the molecule is symmetrical and all outer atoms are the same, it is likely nonpolar. If not, it may be polar.
Consider the electronegativity differences between the central atom and the surrounding atoms. If there are polar bonds and the shape does not cancel out the dipoles, the molecule is polar.
Try solving on your own before revealing the answer!
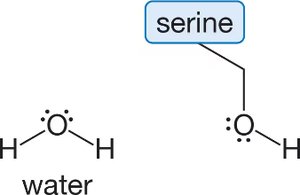
Q16. Amino acids differ from each other by their unique branching “R” groups. Study the branching “R” group of the amino acid serine and choose the single most important type of intermolecular force that the side chain of serine experiences with water.
Background
Topic: Intermolecular Forces in Biological Molecules
This question is about identifying the strongest intermolecular force between the side chain of serine (which contains an -OH group) and water molecules.
Key Terms and Concepts:
Intermolecular Forces: Forces of attraction between molecules, including dispersion forces, dipole-dipole interactions, hydrogen bonds, and ionic bonds.
Hydrogen Bond: A strong type of dipole-dipole interaction that occurs when hydrogen is bonded to a highly electronegative atom (N, O, or F).
Serine Side Chain: Contains an -OH group, which can participate in hydrogen bonding.
Step-by-Step Guidance
Identify the functional group present in the serine side chain (look for -OH, -NH2, etc.).
Recall which intermolecular forces are possible for molecules containing -OH groups (hydrogen bonding is especially important).
Compare the possible intermolecular forces (dispersion, dipole-dipole, hydrogen bonding, ionic) and determine which is strongest between serine's side chain and water.
Try solving on your own before revealing the answer!
