 Back
BackShapes and Polarity of Molecules: VSEPR Theory and Molecular Geometry
Study Guide - Smart Notes

Chapter 6: Ionic and Molecular Compounds
Shapes and Polarity of Molecules
The three-dimensional structure and polarity of molecules are essential concepts in chemistry, influencing physical and chemical properties. The Valence Shell Electron-Pair Repulsion (VSEPR) theory is used to predict molecular shapes based on electron group arrangements around a central atom.
Valence Shell Electron-Pair Repulsion (VSEPR) Theory
Definition: VSEPR theory describes how electron groups (bonding pairs and lone pairs) are oriented around a central atom to minimize repulsion.
Key Principle: Electron groups arrange themselves as far apart as possible.
Shape Determination: The number of atoms and lone pairs attached to the central atom determines the specific molecular shape.
Electron Group Geometries and Molecular Shapes
Depending on the number of electron groups, molecules adopt characteristic shapes and bond angles:
Two Electron Groups: Linear geometry, bond angle of 180°.
Three Electron Groups: Trigonal planar geometry, bond angle of 120°.
Four Electron Groups: Tetrahedral geometry, bond angle of 109°.
Examples of Molecular Shapes
Methane (CH4): Tetrahedral shape, four bonds, bond angle 109°.
Carbon Dioxide (CO2): Linear shape, two bonds, bond angle 180°.
Formaldehyde (H2CO): Trigonal planar shape, three bonds, bond angle 120°.
Sulfur Dioxide (SO2): Bent shape, two bonds and one lone pair, bond angle 120°.
Ammonia (NH3): Trigonal pyramidal shape, three bonds and one lone pair, bond angle 109°.
Water (H2O): Bent shape, two bonds and two lone pairs, bond angle 109°.
Electron Groups | Bonded Atoms | Lone Pairs | Bond Angle | Molecular Shape | Example |
|---|---|---|---|---|---|
2 | 2 | 0 | 180° | Linear | CO2 |
3 | 3 | 0 | 120° | Trigonal planar | H2CO |
3 | 2 | 1 | 120° | Bent | SO2 |
4 | 4 | 0 | 109° | Tetrahedral | CH4 |
4 | 3 | 1 | 109° | Trigonal pyramidal | NH3 |
4 | 2 | 2 | 109° | Bent | H2O |
Lewis Structures and Electron Counting
Lewis structures are used to visualize the arrangement of atoms and electron pairs. Electron counting helps determine the total number of valence electrons in a molecule.
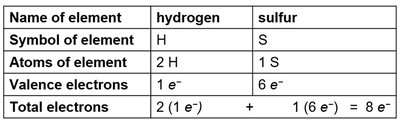
Example: For H2S, hydrogen contributes 1 electron each, sulfur contributes 6 electrons, for a total of 8 valence electrons.


Predicting Molecular Shape: Stepwise Approach
Draw the Lewis structure to identify electron groups.
Arrange electron groups to minimize repulsion (VSEPR theory).
Determine the molecular shape based on the number of bonded atoms and lone pairs.
Polarity of Molecules
Molecular polarity depends on the presence of polar bonds and the symmetry of the molecule. Polar molecules have an uneven distribution of charge, while nonpolar molecules have an even distribution.
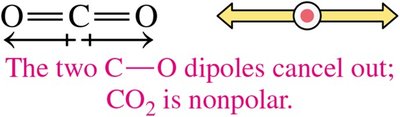
Nonpolar Molecules: Molecules like H2, Cl2, O2, CO2, and CCl4 are nonpolar due to either nonpolar bonds or symmetrical arrangement of polar bonds that cancel each other.
Polar Molecules: Molecules like HCl, H2O, and NH3 are polar because their dipoles do not cancel, resulting in a partial negative and positive end.




Determining Polarity: Stepwise Approach
Determine if the bonds are polar covalent (electronegativity difference > 0.4).
Draw the Lewis structure and assess if the dipoles cancel based on molecular geometry.
If dipoles do not cancel, the molecule is polar; if they do, it is nonpolar.
Key Terms and Definitions
Electron Group: A region of electrons around a central atom, including bonds and lone pairs.
Lone Pair: A pair of valence electrons not shared with another atom.
Bond Angle: The angle between two bonds at the central atom.
Dipole: A separation of charge within a molecule due to differences in electronegativity.
Polar Covalent Bond: A bond where electrons are shared unequally between atoms.
Formulas and Equations
Bond Angle (Tetrahedral):
Bond Angle (Trigonal Planar):
Bond Angle (Linear):
Electronegativity Difference:
Summary Table: Molecular Shapes and Polarity
Molecule | Shape | Polarity |
|---|---|---|
CO2 | Linear | Nonpolar |
CCl4 | Tetrahedral | Nonpolar |
HCl | Linear | Polar |
H2O | Bent | Polar |
NH3 | Trigonal pyramidal | Polar |
Example Application: Predicting the shape and polarity of H2S:
Lewis structure shows two bonds and two lone pairs on S.
Electron groups arrange tetrahedrally; shape is bent.
Dipoles do not cancel; H2S is polar.
Additional info: These concepts are foundational for understanding chemical reactivity, intermolecular forces, and the behavior of substances in biological and environmental contexts.
