 Back
BackSolutions and Their Properties: Study Guide for College Chemistry
Study Guide - Smart Notes

Solutions
Definition and Components
Solutions are homogeneous mixtures composed of two or more substances. They form when the attraction between solute and solvent molecules is sufficient to overcome the attractions within each component. The two main components are:
Solvent: The substance present in greater amount.
Solute: The substance present in lesser amount.

Example: In a saltwater solution, water is the solvent and salt is the solute.
Types of Solutions
Solutions can be classified based on the physical states of their solute and solvent:
Gas Solutions: Air (O2 in N2)
Liquid Solutions: Soda water (CO2 in H2O), vinegar (acetic acid in water), seawater (NaCl in water)
Solid Solutions: Brass (Zn in Cu), steel (C in Fe)
Solutes and solvents may be solids, liquids, or gases.
Water as a Solvent
Properties of Water
Water is one of the most common solvents in nature. It is a polar molecule due to polar O–H bonds, and its molecules form hydrogen bonds, which are important in many biological compounds.
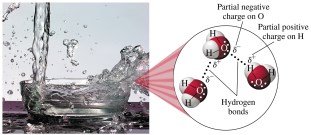
Example: Water's polarity allows it to dissolve ionic and polar substances efficiently.
Formation of Solutions
Solutions form when solute–solvent interactions are strong enough to overcome solute–solute and solvent–solvent interactions.
Solutions with Ionic Solutes
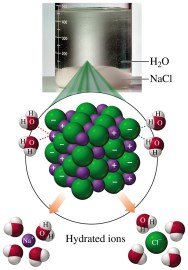
When ionic compounds such as NaCl dissolve in water, the ions undergo hydration as water molecules surround each ion and pull it into solution.
Equation:
Solutions with Polar and Nonpolar Solutes
Polar molecular compounds (e.g., methanol, CH4O) are soluble in water because they can form hydrogen bonds with water. Nonpolar compounds (e.g., iodine, oil) do not dissolve in water due to lack of attraction between nonpolar solute and polar solvent.

Rule: "Like dissolves like"—polar solutes require polar solvents, nonpolar solutes require nonpolar solvents.
Electrolytes and Nonelectrolytes
Electrolytes in the Body
Electrolytes such as sodium, potassium, chloride, and bicarbonate are crucial for cellular and organ function. They can be measured in blood tests.

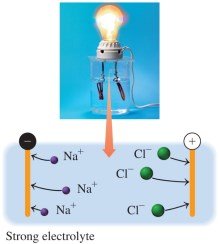
Strong Electrolytes
Strong electrolytes dissociate 100% in water, producing ions and forming solutions that conduct electricity strongly.
Equation:
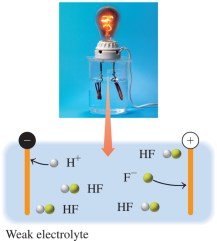
Weak Electrolytes
Weak electrolytes dissociate only slightly in water, forming a solution with few ions and mostly undissociated molecules.
Equation:
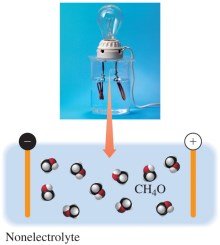
Nonelectrolytes
Nonelectrolytes dissolve as molecules in water, do not produce ions, and do not conduct electricity.
Equation:
Solubility
Definition and Factors
Solubility is the maximum amount of solute that dissolves in a specific amount of solvent. It is temperature sensitive and usually expressed as grams of solute per 100 g of solvent.
Example: Gout occurs when uric acid concentration exceeds its solubility in blood plasma.
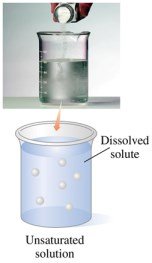
Unsaturated and Saturated Solutions
Unsaturated solutions contain less than the maximum amount of solute and can dissolve more. Saturated solutions contain the maximum amount and have undissolved solute at the bottom, with dissolving and recrystallizing occurring in equilibrium.

Solubility Rules for Ionic Compounds
Only ionic compounds containing a soluble cation or anion are soluble in water. Insoluble compounds have ionic bonds too strong for water to break. Solubility rules help predict solubility:
Soluble cations: Li+, Na+, K+, Rb+, Cs+, NH4+
Soluble anions: NO3-, C2H3O2-
Exceptions: Cl-, Br-, I- are insoluble with Ag+, Pb2+, Hg22+; SO42- is insoluble with Ba2+, Pb2+, Ca2+, Sr2+, Hg22+

Example: BaSO4 is insoluble and used in X-ray imaging.
Solution Concentrations
Mass Percent (m/m)
Mass percent is the concentration by mass of solute in mass of solution:
Formula:


Example: 8.00 g KCl in 42.00 g water gives a 16.0% (m/m) solution.
Volume Percent (v/v) and Mass/Volume Percent (m/v)
Volume percent (v/v):
Mass/volume percent (m/v):
Molarity (M)
Molarity is the moles of solute per liter of solution:
Formula:
Example: 1.0 M NaCl means 1.0 mole NaCl in 1 L solution.
Conversion Factors from Concentrations
Percent concentration and molarity can be used as conversion factors in calculations:
10% (m/m) KCl solution: and
6.0 M HCl solution: and
Dilution of Solutions
Process and Equation
Dilution involves adding solvent (usually water) to a solution, increasing its volume and decreasing its concentration, while the mass of solute remains unchanged.


Equation: (initial and diluted concentrations and volumes)
Properties of Solutions
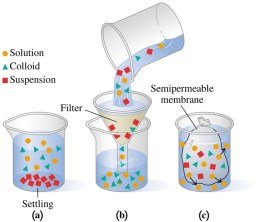
Solutions, Colloids, and Suspensions
Mixtures can be classified as solutions, colloids, or suspensions based on particle size and behavior:
Solutions: Transparent, do not separate, contain small particles that pass through filters and semipermeable membranes.
Colloids: Medium-sized particles, cannot be filtered, can be separated by semipermeable membranes.
Suspensions: Large particles, settle out, can be filtered, must be stirred to stay suspended.

Osmosis and Osmotic Pressure
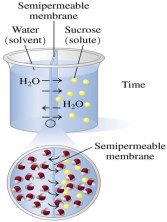
Osmosis
Osmosis is the flow of water (solvent) from a lower to a higher solute concentration through a semipermeable membrane, equalizing concentrations over time.
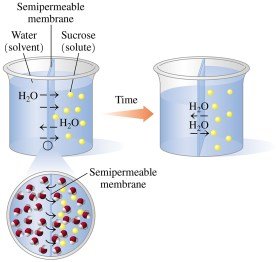
Osmotic Pressure
Osmotic pressure is the pressure required to prevent the flow of additional water into the more concentrated solution. It increases with the number of dissolved particles.
Reverse Osmosis
Reverse osmosis uses pressure greater than osmotic pressure to force water through a purification membrane, leaving solute behind. It is used in desalination plants.
Isotonic, Hypotonic, and Hypertonic Solutions
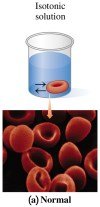
Isotonic Solutions
Isotonic solutions exert the same osmotic pressure as body fluids, maintaining normal cell volume. Most IV solutions are isotonic (e.g., 5.0% glucose, 0.90% NaCl).
Hypotonic Solutions
Hypotonic solutions have lower solute concentration than cells, causing water to flow into cells by osmosis, leading to cell swelling and bursting (hemolysis).


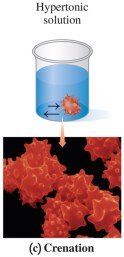
Hypertonic Solutions
Hypertonic solutions have higher solute concentration than cells, causing water to leave cells by osmosis, resulting in cell shrinkage (crenation).
 Additional info: All equations are provided in LaTeX format as required. Tables are described in text for clarity. Images are included only when directly relevant to the explanation.
Additional info: All equations are provided in LaTeX format as required. Tables are described in text for clarity. Images are included only when directly relevant to the explanation.
