 Back
BackStoichiometric Conversion Factors for Fe and O₂ Reaction
Study Guide - Smart Notes

Q29. Given the following equation, what is the correct form of the conversion factor needed to convert the number of moles of O2 to the number of moles of Fe2O3 produced?
Background
Topic: Stoichiometry and Mole Ratios
This question tests your understanding of how to use balanced chemical equations to set up mole-to-mole conversion factors. Specifically, it asks you to identify the correct ratio to convert from moles of O2 to moles of Fe2O3 using the coefficients from the balanced equation:
4Fe(s) + 3O2(g) → 2Fe2O3(s)
Key Terms and Formulas
Stoichiometry: The calculation of reactants and products in chemical reactions using balanced equations.
Mole Ratio: The ratio of the coefficients of any two substances in a balanced chemical equation.
The general formula for a conversion factor is:
Step-by-Step Guidance
Start by writing the balanced chemical equation:
Identify the substances involved in the conversion: you are converting from moles of O2 (given) to moles of Fe2O3 (desired).
From the balanced equation, note the coefficients: 3 for O2 and 2 for Fe2O3.
Set up the conversion factor so that moles of O2 will cancel, leaving moles of Fe2O3:
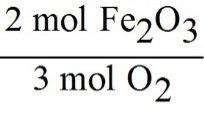
Try solving on your own before revealing the answer!
Final Answer:
The correct conversion factor is:
This ratio comes directly from the coefficients in the balanced equation and allows you to convert from moles of O2 to moles of Fe2O3.
