 Back
BackStoichiometric Conversions and Molar Ratios in Chemical Reactions
Study Guide - Smart Notes

Q43. Given the following equation, which molar conversion factor would be used to find the number of moles of Fe needed to produce a known number of moles of Fe2O3?
Background
Topic: Stoichiometry and Molar Ratios
This question tests your understanding of how to use balanced chemical equations to set up conversion factors (mole ratios) for stoichiometric calculations. These ratios allow you to convert between moles of different substances involved in a reaction.
Key Terms and Formulas
Stoichiometry: The calculation of reactants and products in chemical reactions using balanced equations.
Mole Ratio: A conversion factor derived from the coefficients of a balanced chemical equation.
For the reaction:
The relevant mole ratios are:
Step-by-Step Guidance
Start by identifying what you are given and what you are asked to find. Here, you want to convert from moles of Fe2O3 (product) to moles of Fe (reactant).
Write the balanced chemical equation and note the coefficients for Fe and Fe2O3:
Set up the mole ratio using the coefficients from the balanced equation. The ratio should relate moles of Fe to moles of Fe2O3:
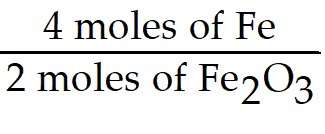
To convert from moles of Fe2O3 to moles of Fe, multiply the given moles of Fe2O3 by this ratio.
Try solving on your own before revealing the answer!
Final Answer:
This ratio allows you to convert from moles of Fe2O3 to moles of Fe using the coefficients from the balanced equation.
