 Back
BackStudy Guidance for GOB Chemistry: Naming Compounds, Solubility, and Oxidation Numbers
Study Guide - Smart Notes

Q1. Name the following compounds based on their chemical formulas:
Formula | Name |
|---|---|
Na2SO4 | __________ |
KH2PO4 | __________ |
Fe(NO3)3 | __________ |
Mn(OH)2 | __________ |
Na2SO3 | __________ |
NH4ClO3 | __________ |
Ca(OH)2 | __________ |
Co(ClO)2 | __________ |
(NH4)2Cr2O7 | __________ |
Cu(NO2)2 | __________ |
Background
Topic: Naming Ionic Compounds (including polyatomic ions and transition metals)
This question tests your ability to apply nomenclature rules for ionic compounds, including those containing polyatomic ions and transition metals with variable oxidation states.
Key Terms and Concepts:
Ionic Compound: A compound composed of cations (positively charged ions) and anions (negatively charged ions).
Polyatomic Ion: An ion composed of two or more atoms covalently bonded, carrying a net charge (e.g., SO42−, NO3−).
Transition Metals: Metals that can have more than one possible charge; their charge is indicated with Roman numerals in the compound name.
Naming Rules:
Name the cation first, then the anion.
For transition metals, indicate the charge with Roman numerals.
Use the correct name for polyatomic ions (e.g., sulfate, nitrate, phosphate).
Step-by-Step Guidance
Identify the cation and anion in each formula. For example, in Na2SO4, Na+ is the cation and SO42− is the anion.
Look up or recall the name of each polyatomic ion (e.g., SO42− is sulfate, NO3− is nitrate).
For compounds with transition metals (like Fe, Mn, Co, Cu), determine the charge on the metal by balancing the charges of the ions in the formula. Use Roman numerals to indicate the charge in the name.
Combine the names, placing the cation first and the anion second. For example, Na2SO4 becomes sodium sulfate.
Repeat this process for each compound in the table, being careful with polyatomic ions and transition metals.
Try solving on your own before revealing the answer!
Final Answers:
Formula | Name |
|---|---|
Na2SO4 | Sodium sulfate |
KH2PO4 | Potassium dihydrogen phosphate |
Fe(NO3)3 | Iron(III) nitrate |
Mn(OH)2 | Manganese(II) hydroxide |
Na2SO3 | Sodium sulfite |
NH4ClO3 | Ammonium chlorate |
Ca(OH)2 | Calcium hydroxide |
Co(ClO)2 | Cobalt(II) hypochlorite |
(NH4)2Cr2O7 | Ammonium dichromate |
Cu(NO2)2 | Copper(II) nitrite |
Each name follows the rules for ionic compounds, including the use of Roman numerals for transition metals and correct polyatomic ion names.
Q2. Use the solubility guidelines to determine if a compound is soluble in water.
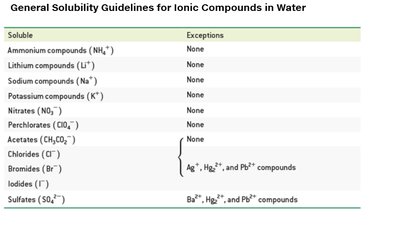
Background
Topic: Solubility Rules for Ionic Compounds
This question tests your ability to use solubility guidelines to predict whether an ionic compound will dissolve in water.
Key Terms and Concepts:
Solubility: The ability of a substance to dissolve in water.
Soluble Compounds: Compounds that dissolve in water to form aqueous solutions.
Exceptions: Some compounds are generally soluble except when paired with certain ions.
Step-by-Step Guidance
Identify the ions present in the compound (e.g., Na+, SO42−).
Refer to the solubility guidelines chart to check if the compound is generally soluble.
Check for any exceptions listed in the chart (e.g., sulfates are soluble except with Ba2+, Hg22+, Pb2+).
Decide if the compound is soluble (aqueous) or insoluble (forms a precipitate).
Try solving on your own before revealing the answer!
Final Answer Example:
For Na2SO4: Sodium compounds are always soluble, so Na2SO4 is soluble in water.
Repeat this process for each compound using the chart above.
Q3. Assign oxidation numbers to elements in compounds and describe redox changes.

Background
Topic: Oxidation Numbers and Redox Reactions
This question tests your understanding of how to assign oxidation numbers to elements in compounds and identify oxidation and reduction processes.
Key Terms and Concepts:
Oxidation Number: A value assigned to an atom in a compound based on a set of rules (see image for details).
Redox Reaction: A chemical reaction involving the transfer of electrons, resulting in changes in oxidation numbers.
Oxidation: Increase in oxidation number (loss of electrons).
Reduction: Decrease in oxidation number (gain of electrons).
Step-by-Step Guidance
Assign oxidation numbers to each element in the compound using the rules provided (see image for reference).
For each reaction, compare the oxidation numbers of each element before and after the reaction.
Determine which element is oxidized (increase in oxidation number) and which is reduced (decrease in oxidation number).
Describe the changes in oxidation numbers and identify the oxidizing and reducing agents.
Try solving on your own before revealing the answer!
Final Answer Example:
For 2 Mg (s) + O2 (g) → 2 MgO (s):
Mg: 0 (elemental) → +2 (in MgO)
O: 0 (elemental) → -2 (in MgO)
Mg is oxidized (0 to +2), O is reduced (0 to -2).
