 Back
BackStudy Guide: Alcohols, Phenols, Ethers, and Sulfur Compounds (Chapter 14)
Study Guide - Smart Notes

Compounds Containing Oxygen, Sulfur, or a Halogen
Overview
This chapter explores the structure, properties, nomenclature, and reactions of organic compounds containing oxygen and sulfur, focusing on alcohols, phenols, ethers, thiols, and thioethers. Understanding these compounds is essential for grasping their roles in biochemistry, industrial applications, and physiological processes.
Alcohols
Structure and Properties
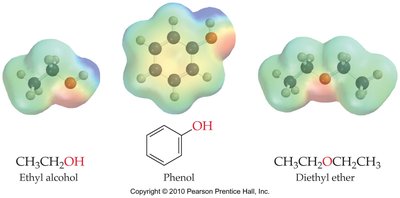
Alcohols are organic compounds containing a hydroxyl (-OH) group attached to a carbon atom. Their properties are similar to water due to the presence of the -OH group, which allows hydrogen bonding.
Hydrogen Bonding: Alcohols can form hydrogen bonds both with themselves and with water, leading to higher boiling points and water solubility compared to alkanes and ethers.
Intermolecular Forces: The strength of intermolecular forces determines boiling points. Alcohols have stronger forces due to hydrogen bonding, while ethers and alkanes rely on weaker London dispersion forces.
Water Solubility: Alcohols with shorter carbon chains are more water-soluble; longer chains behave more like hydrocarbons and are less soluble.
Common Alcohols and Their Uses
Methanol (CH3OH): Industrial solvent, fuel, toxic.
Ethanol (CH3CH2OH): Alcoholic beverages, industrial solvent, less toxic than methanol.
Isopropanol: Rubbing alcohol, disinfectant, more toxic than ethanol.
Ethylene Glycol: Antifreeze, toxic.
Propylene Glycol: Antifreeze, non-toxic.
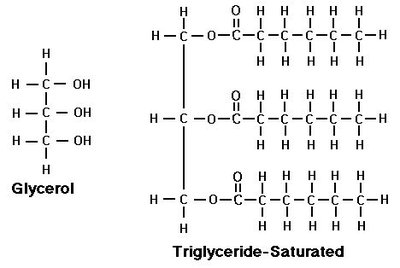
Glycerol: Pharmaceutical solvent, moisturizing agent, backbone for fats.
Naming Alcohols
Common Names: Name the alkyl group, then add "alcohol" (for 1-4 carbons).
IUPAC Names:
Name the longest carbon chain with the -OH group.
Drop the final "-e" and add "-ol".
Number the chain from the end nearest the -OH group.
Indicate the position of the -OH group.
Name and number any substituents.
Diols: Compounds with two -OH groups are named with the suffix "-diol" and both carbon numbers are listed.
Classification of Alcohols
Primary (1o): One carbon attached to the OH-bearing carbon.
Secondary (2o): Two carbons attached.
Tertiary (3o): Three carbons attached.

Physical Properties
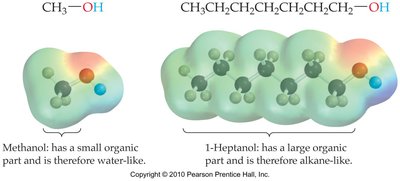
Alcohols are part hydrocarbon and part water-like.
Shorter alcohols are more water-like; longer alcohols are more hydrocarbon-like.
Alcohols with multiple -OH groups have higher boiling points and greater water solubility.

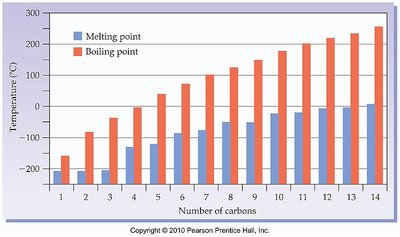
Boiling and Melting Points
Boiling and melting points increase with the number of carbons and the presence of hydrogen bonding.
Chemical Reactions of Alcohols
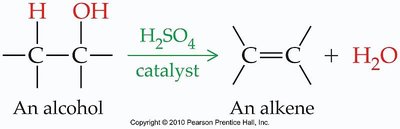
Dehydration
Alcohols can undergo dehydration, losing water to form alkenes or ethers.
Intramolecular Dehydration: Forms an alkene.
Intermolecular Dehydration: Forms an ether.
Major Product: The alkene with more carbons attached to the double bond is favored.
Oxidation
Oxidation in organic chemistry involves a decrease in C-H bonds and an increase in C-O bonds.
Combustion: Alcohol + O2 → CO2 + H2O
Mild Oxidation: Forms carbonyl groups.

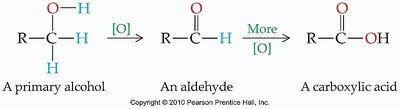
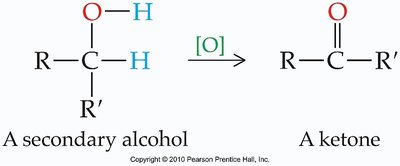
Oxidation of Different Alcohols
Primary Alcohols: Oxidized to aldehydes, then to carboxylic acids.
Secondary Alcohols: Oxidized to ketones, no further reaction.
Tertiary Alcohols: No reaction under mild oxidation.

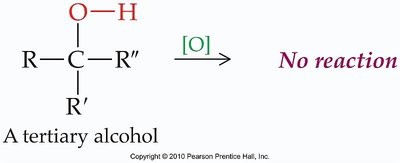
Phenols
Structure and Properties
Phenols consist of a benzene ring bonded to a hydroxyl group. Modified phenols are used as disinfectants and antioxidants.
Boiling Point: Higher than alkylbenzenes.
Water Solubility: Some solubility, less than alcohols.
Acidity: More acidic than alcohols (Ka ≅ 10-10).
Naming Phenols
The parent compound is "phenol"; substituted phenols are named accordingly.
Chemical Reactions
Combustion: Phenol + O2 → CO2 + H2O
No dehydration or oxidation like alcohols.
Ethers
Structure and Properties
Ethers have an oxygen atom single-bonded to two carbon atoms (R-O-R). They are slightly polar but cannot form hydrogen bonds with themselves, resulting in lower boiling points and moderate water solubility.
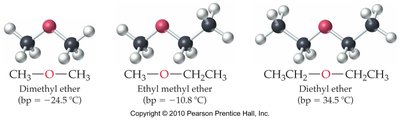
Naming Ethers
Common Names: Name the two substituent groups, then add "ether".
IUPAC Names: Name the shorter chain as an alkoxy substituent; the longer chain as the parent alkane.

Chemical Properties
Ethers are generally unreactive and good solvents.
Highly flammable; can form explosive organic peroxides with oxygen.
Uses of Ethers
Industrial and laboratory solvents (e.g., diethyl ether).
Anesthetics (historical and modern, with halogenated ethers).
Cyclic Ethers
Heterocyclic compounds contain atoms other than carbon in a ring.
Common in biochemistry (e.g., sugars, nucleic acids).
Sulfur Analogs: Thiols and Thioethers
Structure and Properties
Thiols: Sulfur replaces oxygen in alcohols (R-SH).
Thioethers: Sulfur replaces oxygen in ethers (R-S-R).
Carbon-Sulfur Bonds: Non-polar due to similar electronegativity.
Boiling Point and Solubility: Low, as S-H cannot form hydrogen bonds.
Naming Thiols and Thioethers
Thiols: Common name uses "mercaptan"; IUPAC uses "-thiol" suffix.
Thioethers: Common name uses "sulfide"; IUPAC uses "alkylthio-" as a substituent.
Chemical Reactions of Thiols
Most important reaction is oxidation: two thiols combine to form a disulfide (R-S-S-R).
Reactions differ from alcohols due to non-polarity.
Summary Table: Hydrogen Bonding and Properties
Compound | Boiling Point (H bond with itself?) | Water Solubility (H bond with water?) |
|---|---|---|
Alkanes | No | No |
Alcohols/Phenols | Yes | Yes |
Ethers | No | Yes |
Thiols | No | No |
Key Equations
Alcohol Dehydration:
Alcohol Oxidation:
Primary Alcohol Oxidation:
Secondary Alcohol Oxidation:
Tertiary Alcohol Oxidation: No reaction under mild conditions.
Phenol Combustion:
Thiols Oxidation:
Conclusion
Alcohols, phenols, ethers, thiols, and thioethers are fundamental organic compounds with diverse properties and applications. Their ability to form hydrogen bonds, undergo oxidation and dehydration, and their structural variations underpin their roles in chemistry and biochemistry.
