 Back
BackStudy Guide: Chemical Reactions, Balancing Equations, and Reaction Types
Study Guide - Smart Notes

Q1. Balance each of these equations, using the techniques described in this section:
Pb(ClO3)2 + NaI → NaClO3 + PbI2
Al(NO2)3 + Ca → Al + Ca(NO2)2
C4H10 + O2 → CO2 + H2O
Background
Topic: Balancing Chemical Equations
This question tests your ability to balance chemical equations, ensuring the Law of Conservation of Mass is obeyed. You must make sure the number of atoms for each element is the same on both sides of the equation.
Key Terms and Formulas
Reactants: Substances present before the reaction.
Products: Substances formed by the reaction.
Coefficients: Numbers placed in front of compounds to balance the equation.
Law of Conservation of Mass: Mass is neither created nor destroyed in a chemical reaction.
Step-by-Step Guidance
Write out the unbalanced equation for each reaction.
Count the number of atoms of each element on both sides of the equation.
Adjust the coefficients (not the subscripts) to balance the atoms for each element.
Check your work: Make sure all elements are balanced and that the coefficients are the lowest possible whole numbers.
Try solving on your own before revealing the answer!

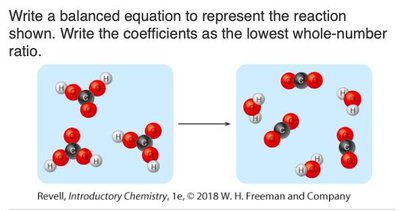
Q2. Write a balanced equation to represent the reaction shown. Write the coefficients as the lowest whole-number ratio.
Background
Topic: Visual Representation of Chemical Reactions
This question asks you to interpret a molecular diagram and write a balanced chemical equation, using the lowest whole-number coefficients.
Key Terms and Formulas
Balanced Equation: An equation with equal numbers of each atom on both sides.
Coefficient: The number in front of a chemical formula indicating the number of molecules.
Step-by-Step Guidance
Identify the reactants and products shown in the diagram.
Count the number of each molecule on both sides.
Write the chemical formulas for each reactant and product.
Assign coefficients to balance the equation, using the lowest whole-number ratio.
Try solving on your own before revealing the answer!
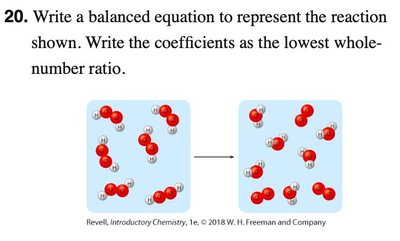
Q3. Write a balanced equation to represent the reaction shown. Write the coefficients as the lowest whole-number ratio. (Question 20)
Background
Topic: Visual Representation of Chemical Reactions
This question is similar to the previous one, requiring you to interpret a molecular diagram and write a balanced equation.
Key Terms and Formulas
Balanced Equation
Coefficient
Step-by-Step Guidance
Examine the diagram to identify the reactants and products.
Count the number of molecules for each substance.
Write the chemical formulas and assign coefficients to balance the equation.
Try solving on your own before revealing the answer!
Q4. Provide the name and phase of the products for each reaction:
a. P (s) + Cl2 (g) → PCl3 (g)
b. Na2SO4 (aq) + BaBr2 (aq) → BaSO4 (s) + 2 NaBr (aq)
c. Cr (s) + 2 HCl (aq) → CrCl2 (aq) + H2 (g)
Background
Topic: Naming Chemical Products and Identifying Phases
This question tests your ability to name the products of a reaction and identify their physical states (solid, liquid, gas, aqueous).
Key Terms and Formulas
Phase Symbols: (s) = solid, (l) = liquid, (g) = gas, (aq) = aqueous (dissolved in water)
Product Names: Use IUPAC naming conventions for compounds.
Step-by-Step Guidance
Identify the products in each reaction.
Determine the correct chemical name for each product.
Assign the correct phase symbol based on the reaction conditions and solubility rules.
Try solving on your own before revealing the answer!

Q5. Write a balanced equation to describe each of these chemical changes. Include phase symbols.
a. When calcium metal is heated in water, aqueous calcium hydroxide and hydrogen gas form.
b. When solid magnesium burns (reacts with oxygen gas), it forms solid magnesium oxide.
c. Aqueous silver nitrate reacts with aqueous sodium chloride to produce solid silver chloride and aqueous sodium nitrate.
Background
Topic: Writing and Balancing Chemical Equations
This question tests your ability to translate word descriptions of chemical reactions into balanced equations, including phase symbols.
Key Terms and Formulas
Phase Symbols: (s), (l), (g), (aq)
Balanced Equation: Equal numbers of each atom on both sides.
Step-by-Step Guidance
Identify the reactants and products from the word description.
Write the chemical formulas for each substance, including phase symbols.
Balance the equation by adjusting coefficients.
Try solving on your own before revealing the answer!

Q6. Classify each of these chemical reactions as a synthesis, decomposition, single-displacement, or double-displacement reaction:
a. Sn + 2 Br2 → SnBr4
b. 2 HBr + Sn → SnBr2 + H2
c. HBr + NaOH → NaBr + H2O
Background
Topic: Types of Chemical Reactions
This question tests your ability to classify reactions based on the rearrangement of atoms and molecules.
Key Terms and Formulas
Synthesis (Combination): Two or more substances combine to form one product.
Decomposition: One substance breaks down into two or more products.
Single-Displacement: One element replaces another in a compound.
Double-Displacement: Two compounds exchange ions to form new compounds.
Step-by-Step Guidance
Examine the reactants and products for each equation.
Identify the pattern of atom rearrangement.
Match the pattern to the correct reaction type.
Try solving on your own before revealing the answer!
Q7. The balanced equation here shows the reaction of hydrogen gas with chlorine gas. Based on this equation, H2 + Cl2 → 2 HCl
a. How many molecules of HCl form from 30 molecules of H2?
b. How many molecules of Cl2 are required to produce 12 molecules of HCl?
Background
Topic: Molecule-to-Molecule Stoichiometry
This question tests your ability to use balanced equations to determine the quantitative relationships between reactants and products.
Key Terms and Formulas
Stoichiometry: The calculation of reactants and products in chemical reactions.
Mole Ratio: The ratio of coefficients in a balanced equation.
Step-by-Step Guidance
Identify the mole ratio from the balanced equation.
Set up a proportion using the given number of molecules and the coefficients.
Calculate the number of molecules needed or produced, stopping before the final calculation.
Try solving on your own before revealing the answer!
Q8. The reaction of aluminum with chlorine gas is shown here: 2 Al + 3 Cl2 → 2 AlCl3
a. How many molecules of chlorine gas are needed to react with 10 aluminum atoms?
b. How many units of AlCl3 can be produced from 10 aluminum atoms?
Background
Topic: Molecule-to-Molecule Stoichiometry
This question tests your ability to use the coefficients in a balanced equation to determine the quantitative relationships between reactants and products.
Key Terms and Formulas
Stoichiometry
Mole Ratio
Step-by-Step Guidance
Identify the mole ratio from the balanced equation.
Set up a proportion using the given number of atoms and the coefficients.
Calculate the number of molecules or units produced, stopping before the final calculation.
Try solving on your own before revealing the answer!
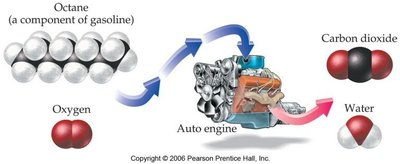
Q9. Explain the process and products of combustion reactions, using octane as an example.
Background
Topic: Combustion Reactions
This question tests your understanding of combustion reactions, particularly the reaction of hydrocarbons with oxygen to produce carbon dioxide and water.
Key Terms and Formulas
Combustion Reaction: A reaction in which a substance reacts with oxygen, releasing energy and producing CO2 and H2O.
General Formula: Hydrocarbon + O2 → CO2 + H2O
Step-by-Step Guidance
Write the chemical formula for octane (C8H18).
Write the unbalanced equation for its combustion with oxygen.
Balance the equation by adjusting coefficients for C, H, and O.
Try solving on your own before revealing the answer!

Q10. Classify the following reactions as synthesis, decomposition, single-displacement, or double-displacement.
Background
Topic: Types of Chemical Reactions
This question tests your ability to recognize and classify chemical reactions based on their general patterns.
Key Terms and Formulas
Synthesis: A + B → AB
Decomposition: AB → A + B
Single-Displacement: A + BC → AC + B
Double-Displacement: AB + CD → AD + CB
Step-by-Step Guidance
Examine the reactants and products for each reaction.
Identify the pattern of atom rearrangement.
Match the pattern to the correct reaction type.
Try solving on your own before revealing the answer!

