 Back
BackThe Mole, Molar Mass, and Stoichiometry in Chemical Reactions
Study Guide - Smart Notes

7.4 The Mole
Counting Units in Chemistry
In chemistry, counting terms such as dozen, gross, and ream are used to specify exact quantities of items. The mole is the standard counting unit for atoms, molecules, and ions, analogous to these everyday terms.
Dozen: 12 items
Gross: 144 items
Ream: 500 sheets (commonly used for paper)
Mole: 6.02 × 1023 items (Avogadro's number)

Avogadro’s Number
The mole is defined as the amount of substance that contains exactly elementary entities (atoms, molecules, or ions). This value is known as Avogadro’s number, named after Amedeo Avogadro.
1 mole = particles
Used to count extremely small particles such as atoms and molecules
Mole of Atoms
One mole of any element contains atoms of that element. For example:
1 mole of carbon = carbon atoms
1 mole of sodium = sodium atoms
1 mole of sulfur = sulfur atoms

Conversion Factor: Avogadro’s Number
Avogadro’s number can be used as a conversion factor between moles and particles:
Equality:
Conversion factors:
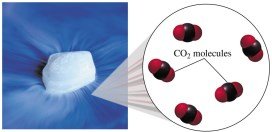
Converting Moles to Molecules
To find the number of molecules in a given amount of moles, multiply by Avogadro’s number. For example, to find the number of CO2 molecules in 0.500 mole:
Moles of Elements in a Chemical Compound
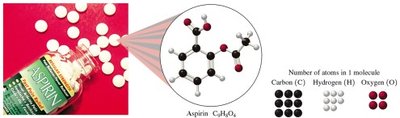
Subscripts and Mole Ratios
Subscripts in a chemical formula indicate the number of moles of each element in one mole of the compound. For example, in aspirin (C9H8O4):
1 mole of C9H8O4 contains:
9 moles of C
8 moles of H
4 moles of O
7.5 Molar Mass
Definition of Molar Mass
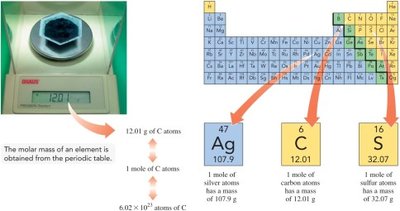
The molar mass of an element or compound is the mass in grams of one mole of that substance. For elements, the molar mass (in g/mol) is numerically equal to the atomic mass (in amu) found on the periodic table.
Example: 1 mole of sodium (Na) has a molar mass of 22.99 g.

Determining Molar Mass from the Periodic Table
The molar mass of a compound is calculated by summing the molar masses of its constituent elements, each multiplied by its subscript in the formula.
Example: For Li2CO3 (lithium carbonate):
2 moles Li × 6.941 g/mol = 13.88 g
1 mole C × 12.01 g/mol = 12.01 g
3 moles O × 16.00 g/mol = 48.00 g
Total molar mass = 13.88 + 12.01 + 48.00 = 73.89 g/mol
Molar Mass Conversion Factors
Molar mass is used to convert between grams and moles:
Equality: 1 mole CH4 = 16.05 g CH4
Conversion factors:
7.6 Calculations Using Molar Mass
Converting Mass to Moles
To determine the number of moles in a given mass of a substance, divide the mass by the molar mass:
Example: A box of NaCl (table salt) contains 737 g. Moles of NaCl =

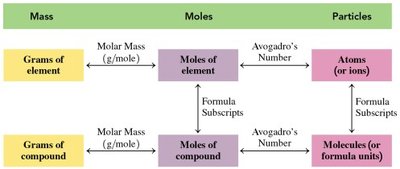
Mass–Moles–Particles Map
This flowchart summarizes the relationships between mass, moles, and particles for both elements and compounds:
Grams ↔ Moles (via molar mass)
Moles ↔ Particles (via Avogadro’s number)
Formula subscripts relate moles of elements to moles of compounds
7.7 Mole Relationships in Chemical Equations
Law of Conservation of Mass
In a chemical reaction, the total mass of reactants equals the total mass of products. Matter is neither created nor destroyed.
Example:

Mole–Mole Factors from an Equation
Balanced chemical equations provide the ratios (mole–mole factors) for converting between moles of reactants and products.
For :
2 moles Fe : 3 moles S : 1 mole Fe2S3
Mole–mole factors can be written as fractions for calculations.

Calculations with Mole Factors
To determine the amount of product formed or reactant needed, use the mole–mole factors from the balanced equation. For example:
Given 6.0 moles O2 in :
7.8 Mass Calculations for Chemical Reactions
Mass Relationships in Reactions
To calculate the mass of a reactant or product in a chemical reaction:
Convert mass of given substance to moles (using molar mass).
Use mole–mole factor from the balanced equation to find moles of desired substance.
Convert moles of desired substance to mass (using molar mass).

7.9 Limiting Reactants and Percent Yield
Limiting and Excess Reactants
In a chemical reaction, the limiting reactant is the substance that is completely consumed first, thus limiting the amount of product formed. The excess reactant is the substance that remains after the reaction is complete.
To identify the limiting reactant, calculate the amount of product formed from each reactant; the one producing the least product is limiting.
Calculating Product from Limiting Reactant
When quantities of reactants are given in grams:
Convert grams of each reactant to moles.
Use mole–mole factors to determine moles of product from each reactant.
The reactant that produces the least amount of product is limiting.
Convert moles of product to grams if required.

Theoretical, Actual, and Percent Yield
Not all reactions go to completion. The theoretical yield is the maximum amount of product predicted by stoichiometry. The actual yield is the amount actually obtained. Percent yield is calculated as:
Example: If 50.0 g of LiOH produces 72.8 g of LiHCO3, calculate the percent yield using the above formula.
