 Back
BackUnit 2 Study Guide: Ions, Ionic and Molecular Compounds, Changes of State, and Polyatomic Ions
Study Guide - Smart Notes

Structure of the Atom and Valence Electrons
Atomic Structure and Electron Cloud
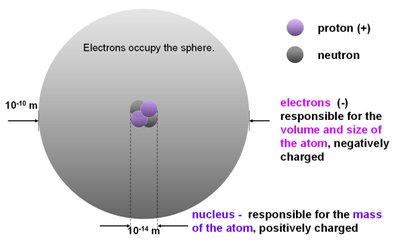
The atom consists of a central nucleus containing protons and neutrons, surrounded by an electron cloud. The arrangement of electrons, especially the valence electrons, determines the chemical properties of an element.
Protons (+): Positively charged particles in the nucleus, responsible for the atom's identity.
Neutrons: Neutral particles in the nucleus, contribute to atomic mass.
Electrons (-): Negatively charged particles occupying energy levels around the nucleus, responsible for the atom's volume and size.
Valence Electrons: Electrons in the highest energy level, crucial for chemical bonding and reactivity.
Additional info: The number of valence electrons for representative elements equals the group number in the periodic table.
Ions and Ionic Compounds
Formation of Ions
Atoms gain or lose electrons to achieve a stable electron configuration, often an octet. Metals lose electrons to form cations, while nonmetals gain electrons to form anions.
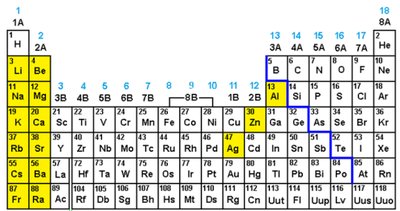
Cations: Positively charged ions formed by metals losing valence electrons. The charge is predictable from the group number (e.g., Group 1A: +1, Group 2A: +2).
Anions: Negatively charged ions formed by nonmetals gaining electrons. The charge depends on how many electrons are needed to reach an octet (e.g., Group 5A: -3, Group 6A: -2, Group 7A: -1).
Octet Rule: Atoms tend to gain, lose, or share electrons to achieve eight valence electrons (except H, Li, Be, which follow the duet rule).
Lewis Symbols
Valence electrons are represented as dots around the element symbol, known as Lewis (electron-dot) symbols.
Ion Naming and Charges
Metal cations with predictable charges are named after the element (e.g., Mg2+: magnesium ion).
Metals with variable charges use Roman numerals (e.g., Fe2+: iron(II) ion).
Monatomic anions are named by adding "-ide" to the root (e.g., Cl-: chloride ion).
Ionic Compounds
Ionic compounds consist of cations and anions held together by ionic bonds. They are electrically neutral, have high melting and boiling points, and are solid at room temperature.
Formula: Cation is written first, followed by the anion. Subscripts indicate the number of each ion needed to balance charges.
Example: Sodium chloride (NaCl) contains Na+ and Cl-.



Periodic Table and Predictable Charges
Some metal cations have predictable charges based on their position in the periodic table. Roman numerals are used for metals with variable charges.
Changes of State and Temperature Scales
States of Matter
Matter exists in three primary states: solid, liquid, and gas. Each state has distinct properties regarding shape, volume, and particle arrangement.
Solids: Definite shape and volume; particles are closely packed and fixed.
Liquids: Indefinite shape, definite volume; particles are close but mobile.
Gases: Indefinite shape and volume; particles are far apart and move rapidly.




Temperature and Scales
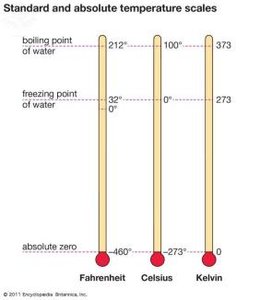
Temperature measures the average kinetic energy of particles and determines the direction of heat flow. Common scales are Celsius (°C), Fahrenheit (°F), and Kelvin (K).
Conversion formulas:

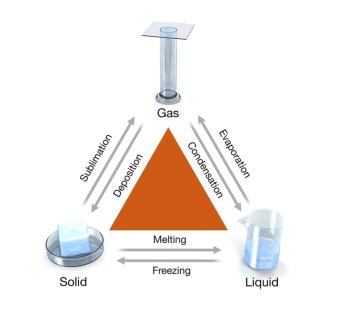
Phase Changes
Phase changes are physical changes involving energy transfer. Endothermic changes absorb heat; exothermic changes release heat.
Melting (Fusion): Solid to liquid (endothermic).
Freezing: Liquid to solid (exothermic).
Sublimation: Solid to gas (endothermic).
Deposition: Gas to solid (exothermic).
Evaporation: Liquid to gas (endothermic).
Condensation: Gas to liquid (exothermic).
Boiling: Rapid vaporization throughout the liquid.






Heat of Fusion and Vaporization
Heat of Fusion (water): or
Heat of Vaporization (water): or
Chemical Changes and Reactions
Chemical Changes
In a chemical change, substances react to form new substances with different properties and compositions. Chemical reactions involve breaking and forming bonds, and may be exothermic or endothermic.


Examples of Physical vs. Chemical Changes
Physical Changes: Boiling water, tearing paper, melting aluminum, dissolving sugar.
Chemical Changes: Burning paper, reacting aluminum with acid, fermenting sugar, digesting bread.
Polyatomic Ions
Definition and Examples
Polyatomic ions are groups of atoms bonded together with an overall charge. Most are anions, except ammonium (NH4+).
Examples: Nitrate (NO3-), Sulfate (SO42-), Phosphate (PO43-), Ammonium (NH4+).

Writing Formulas with Polyatomic Ions
Cation is written first, followed by the polyatomic ion.
Parentheses are used if more than one polyatomic ion is present (e.g., Mg(NO3)2).
Naming Compounds with Polyatomic Ions
Name the cation first, then the polyatomic anion (e.g., NaNO3: sodium nitrate).
Covalent (Molecular) Compounds
Covalent Bonds and Molecules
Covalent bonds form between nonmetal atoms by sharing valence electrons to complete octets or duets. Covalent compounds include diatomic molecules, polyatomic ions, and molecular compounds.
Naming Covalent Compounds
First nonmetal is named fully; second uses the root plus "-ide".
Prefixes indicate the number of atoms: mono-, di-, tri-, tetra-, penta-, etc.
Example: SO3 is sulfur trioxide; P4S3 is tetraphosphorus trisulfide.

Writing Covalent Compound Formulas
List element symbols in order; use subscripts based on prefixes.
Do not reduce subscripts in covalent compounds.
Identifying Ionic vs. Covalent Compounds
Ionic: Formula starts with a metal or NH4+.
Covalent: Formula starts with a nonmetal.
Electronegativity and Types of Bonds
Electronegativity
Electronegativity measures an atom's attraction for shared electrons. It increases across a period and is highest for nonmetals.


Bond Types Based on Electronegativity Difference
Nonpolar Covalent: Difference ≤ 0.4; electrons shared equally.
Polar Covalent: Difference > 0.4 and ≤ 1.8; electrons shared unequally, partial charges develop.
Ionic: Difference > 1.8; electrons transferred, forming ions.
Partial Charges in Polar Covalent Bonds
More electronegative atom: partial negative charge (δ−).
Less electronegative atom: partial positive charge (δ+).
Summary Table: Bond Types by Electronegativity Difference
Bond Type | Electronegativity Difference | Example |
|---|---|---|
Nonpolar Covalent | ≤ 0.4 | H2, Cl2 |
Polar Covalent | 0.4 < ΔEN ≤ 1.8 | HCl, H2O |
Ionic | > 1.8 | NaCl, KBr |
