 Back
BackUnit 6: Carbohydrates – Structure, Chirality, and Biological Importance
Study Guide - Smart Notes

Chirality
Chiral Molecules and Objects
Chirality is a fundamental concept in chemistry describing objects or molecules that cannot be superimposed on their mirror images. This property is crucial in organic chemistry, especially for biological molecules.
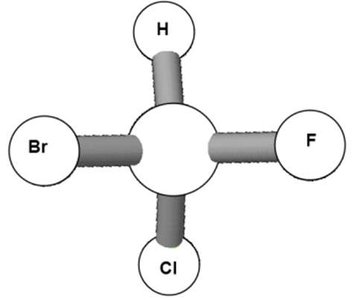
Chiral objects are those whose mirror images are not identical to the original object. For example, left and right hands are chiral because they cannot be superimposed.
Achiral objects are those whose mirror images are identical and can be superimposed.
Chirality in molecules arises when a carbon atom is bonded to four different groups, making it a chiral center.
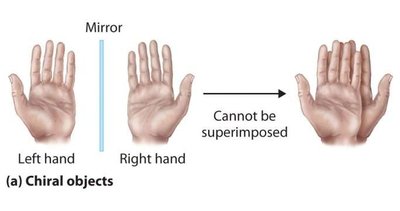
Examples of Chiral and Achiral Molecules
Chiral molecules have non-superimposable mirror images, called enantiomers. Achiral molecules have superimposable mirror images.
Bromochloroiodomethane is chiral because its mirror image cannot be superimposed.
Dibromochloromethane is achiral because its mirror image can be superimposed.
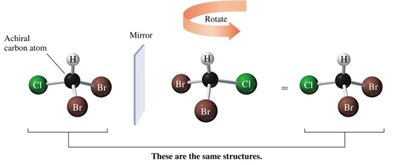
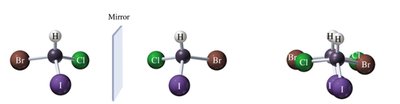
Biological Enantiomers
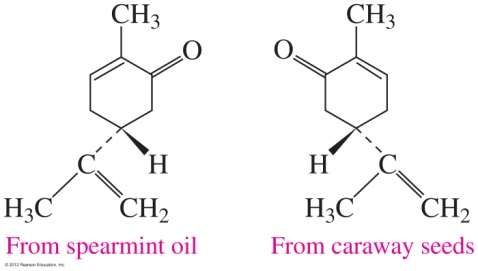
Enantiomers often have different biological effects. Only one enantiomer may be biologically active or safe.
Carvone: One enantiomer smells like spearmint, the other like caraway.
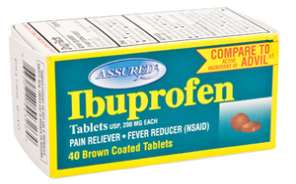
Ibuprofen: Only one enantiomer is effective as a pain reliever.
Thalidomide: One enantiomer is a sedative, the other causes birth defects.
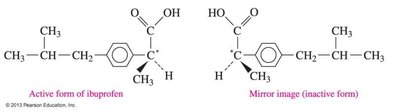
Carbohydrates Overview
Classification and Structure
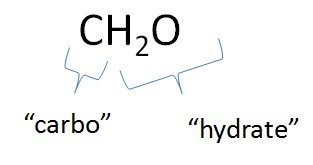
Carbohydrates are organic molecules composed of carbon, hydrogen, and oxygen, typically in the ratio CnH2nOn. They are also known as saccharides.
Monosaccharides: Simple sugars (e.g., glucose, fructose, galactose).
Disaccharides: Two monosaccharides joined (e.g., maltose, sucrose, lactose).
Polysaccharides: Polymers of many monosaccharides (e.g., glycogen, starch, cellulose).
Monosaccharides
Aldose vs Ketose
Monosaccharides are classified based on the position of their carbonyl group:
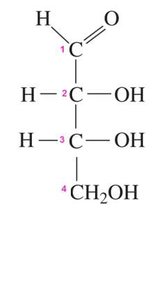
Aldose: Carbonyl group at carbon 1 (an aldehyde).
Ketose: Carbonyl group at carbon 2 (a ketone).
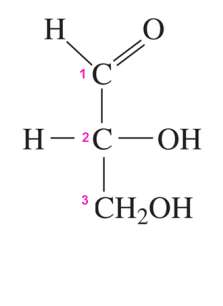
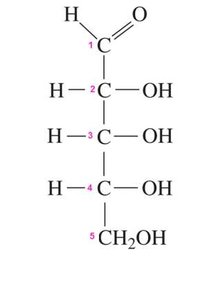
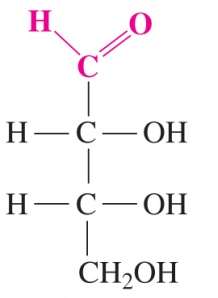
Fischer Projections
Fischer projections are a 2D representation of 3D chiral molecules, especially useful for monosaccharides. Horizontal lines represent bonds projecting forward, vertical lines project backward.
Fischer projections help distinguish between enantiomers.
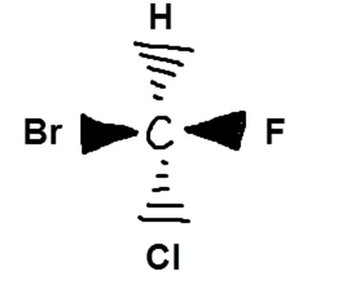
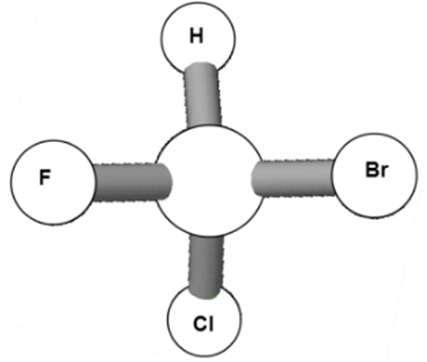
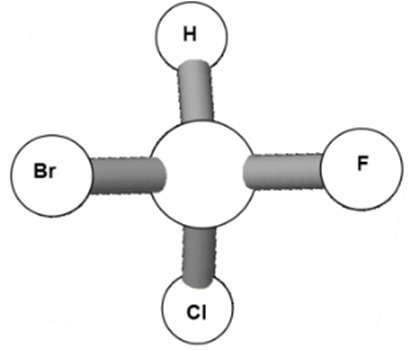
L vs D Enantiomers
Monosaccharides are classified as L or D based on the position of the –OH group on the bottom-most chiral carbon in the Fischer projection:
L-enantiomer: –OH group on the left.
D-enantiomer: –OH group on the right.
Glucose, Galactose, and Fructose
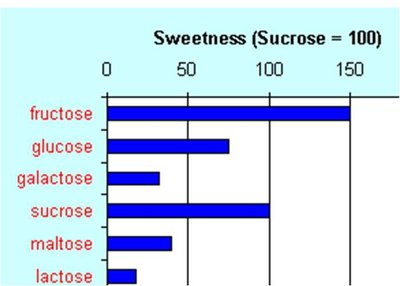
The most common dietary monosaccharides are D-glucose, D-galactose, and D-fructose. Only D-enantiomers are found in nature.
D-glucose: Primary energy source for cells; produced by photosynthesis.
D-galactose: Component of lactose in milk.
D-fructose: Sweetest sugar, found in fruit and honey.

Redox Reactions: Sugar Acids and Alcohols
Oxidation and Reduction of Monosaccharides
The aldehyde group of an aldose can be oxidized to a carboxylic acid (sugar acid) or reduced to a primary alcohol (sugar alcohol).
Sugar acid: Named by replacing "ose" with "onic acid".
Sugar alcohol: Named by replacing "ose" with "itol" (e.g., sorbitol, mannitol).

Haworth Structures and Alpha/Beta Anomers
Cyclic Forms of Monosaccharides
Monosaccharides are more stable in solution as cyclic structures. The Haworth structure shows the ring form, with OH groups pointing up or down.
The OH group on C5 attacks C1 (aldose) or C2 (ketose), forming a ring.
The new OH group at the anomeric carbon can be alpha (down) or beta (up).
Mutarotation: The ring opens and closes, interconverting alpha and beta forms.
Disaccharides
Maltose, Sucrose, and Lactose
Disaccharides are formed by joining two monosaccharides via a glycosidic bond (an ether linkage).
Maltose: Glucose + glucose (α-1,4 bond).
Lactose: Galactose + glucose (β-1,4 bond).
Sucrose: Fructose + glucose (α,β-1,2 bond).
Classifying Glycosidic Bonds
The type of glycosidic bond depends on which OH groups are joined and their orientation (alpha or beta).
Alpha and beta refer to the configuration at the anomeric carbon.
Reducing Sugars
Definition and Testing
A reducing sugar is any sugar that can be oxidized, typically detected by Benedict’s test. Reducing sugars have a free anomeric carbon that can open to form an aldehyde or ketone.
Monosaccharides: Glucose, galactose, and fructose are reducing sugars.
Disaccharides: Maltose and lactose are reducing sugars; sucrose is not.
Polysaccharides
Glycogen, Starch, and Cellulose
Polysaccharides are large carbohydrate polymers with distinct biological functions and structures.
Glycogen: Energy storage in animals; highly branched (α-1,4 and α-1,6 bonds).
Starch: Energy storage in plants; consists of amylose (unbranched, α-1,4) and amylopectin (branched, α-1,4 and α-1,6).
Cellulose: Structural component in plants; unbranched, β-1,4 bonds (not digestible by humans).
Polysaccharide | Function | Monosaccharide Involved | Branching? | Type(s) of Glycosidic Bonds |
|---|---|---|---|---|
Glycogen | Energy storage in animals | D-glucose | Yes | α-1,4, α-1,6 |
Amylose | Energy storage in plants | D-glucose | No | α-1,4 |
Amylopectin | Energy storage in plants | D-glucose | Yes | α-1,4, α-1,6 |
Cellulose | Structure in plants | D-glucose | No | β-1,4 |
Key Terms and Concepts
Carbohydrate, monosaccharide, aldose/ketose, triose/tetrose/pentose/hexose, chiral/achiral, enantiomers, Fischer projection, D and L enantiomers, glucose, galactose, fructose, cellular respiration, photosynthesis, sugar acid, sugar alcohol, reducing sugar, Benedict’s test, disaccharide, Haworth structure, mutarotation, alpha & beta isomers, maltose, lactose, sucrose, glycosidic bond, polysaccharide, glycogen, starch (amylose & amylopectin), cellulose
Important Equations
Photosynthesis:
Cellular Respiration:
