 Back
BackAcids and Bases: Definitions, Properties, and Calculations
Study Guide - Smart Notes

Chapter 14: Acids and Bases
Definitions of Acids and Bases
Acids and bases are fundamental chemical species with distinct properties and behaviors in aqueous solutions. Their definitions have evolved to encompass a broader range of chemical phenomena.
Arrhenius Definition:
Acids: Ionic compounds that dissociate in water to produce hydrogen ions (H+).
Bases: Ionic compounds that dissociate in water to produce hydroxide ions (OH-).
Brønsted–Lowry Definition:
Acids: Proton donors (compounds that can donate H+ ions).
Bases: Proton acceptors (compounds that accept H+ ions).
Properties of Acids
Acids exhibit characteristic properties due to their ability to increase the concentration of hydrogen ions in solution.
Increase [H+] in solution
Sour taste (e.g., vinegar)
React with many metals to produce hydrogen gas (H2)
Turn blue litmus paper red
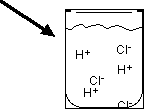

Strength of Acids
The strength of an acid depends on the concentration of H+ ions it produces in solution. Strong acids completely ionize, while weak acids only partially ionize.
Properties of Bases
Bases are substances that increase the concentration of hydroxide ions in solution and have distinct physical and chemical properties.
Increase [OH-] in solution
Bitter taste
Slippery feel
Turn red litmus paper blue
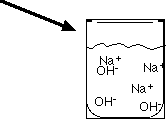

Strength of Bases
The strength of a base is determined by the concentration of hydroxide ions (OH-) it produces. Strong bases completely dissociate, while weak bases only partially ionize.
Strong vs. Weak Acids and Bases
The distinction between strong and weak acids/bases is based on their degree of ionization in water.
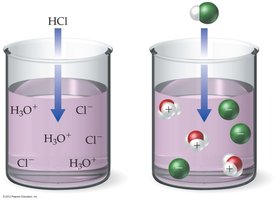
Strong Acid: Completely ionizes in water (e.g., HCl). No intact acid molecules remain in solution.
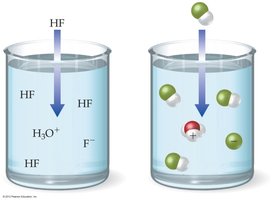
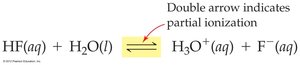
Weak Acid: Only partially ionizes in water (e.g., HF). Many intact acid molecules remain.
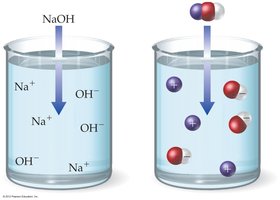
Strong Base: Completely dissociates in water (e.g., NaOH).
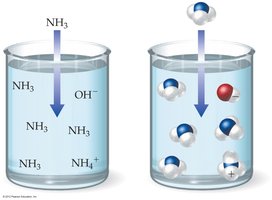
Weak Base: Only partially ionizes in water (e.g., NH3).
Strong Acids and Bases: Dissociation Reactions
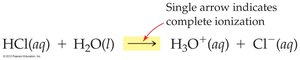
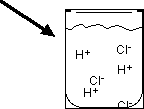
Strong acids and bases are soluble ionic compounds that dissociate completely in water, producing high concentrations of ions.
Hydrogen ion (H+) does not exist freely in solution; it forms the hydronium ion (H3O+).
Example dissociation reactions:
Neutralization Reactions
When acids and bases react, they neutralize each other, forming water and a salt. The net ionic equation for many neutralization reactions is:
Example:
Electrolytes: Conductivity of Acids and Bases
Acids and bases are electrolytes because they dissociate into ions in aqueous solution, allowing the solution to conduct electricity. Strong acids and bases are strong electrolytes, while weak acids and bases are weak electrolytes.
Limitations of the Arrhenius Theory
The Arrhenius theory is limited because it only applies to substances that contain H+ or OH- ions. The Brønsted–Lowry theory is broader, explaining phenomena such as ammonia acting as a base by accepting a proton from water.
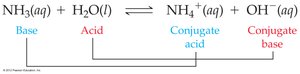
Brønsted–Lowry Theory: Conjugate Acid-Base Pairs
In Brønsted–Lowry acid-base reactions, acids donate protons and become conjugate bases, while bases accept protons and become conjugate acids.
Amphoteric Nature of Water
Water is amphoteric, meaning it can act as either an acid or a base depending on the reaction partner.
As a base:
As an acid:
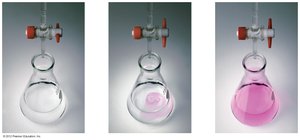
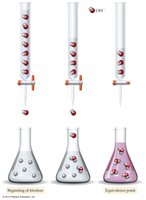
Titration: Quantifying Acids and Bases
Titration is a laboratory technique used to determine the concentration of an unknown acid or base by reacting it with a solution of known concentration. The equivalence point is when the amounts of acid and base are stoichiometrically equivalent.
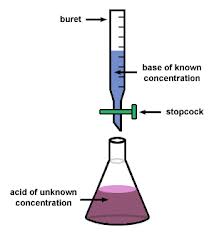
Example Calculation
To find the concentration of an unknown HCl solution titrated with NaOH:
Given: 10.00 mL HCl, 12.54 mL of 0.100 M NaOH to reach equivalence point.
Calculation:
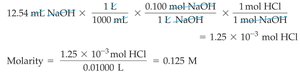
Ion Product Constant for Water (Kw)
Water self-ionizes to a small extent, producing hydronium and hydroxide ions. The product of their concentrations is a constant at 25°C:
In neutral solutions, .
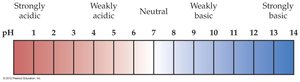
pH and pOH: Measuring Acidity and Basicity
The pH scale quantifies the acidity or basicity of a solution. It is defined as the negative logarithm of the hydronium ion concentration:
(at 25°C)
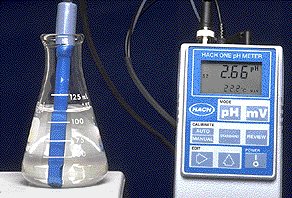
Examples
For a 0.0015 M HCl solution:
For [H3O+] = 1.8 × 10-4 M: (acidic, since pH < 7)
For a solution with pH = 2.66:
Summary Table: Acidic, Neutral, and Basic Solutions
Solution Type | [H3O+] | [OH-] | pH |
|---|---|---|---|
Acidic | > 1.0 × 10-7 M | < 1.0 × 10-7 M | < 7 |
Neutral | 1.0 × 10-7 M | 1.0 × 10-7 M | 7 |
Basic | < 1.0 × 10-7 M | > 1.0 × 10-7 M | > 7 |
Key Equations
Additional info:
Strong acids and bases are strong electrolytes and conduct electricity well due to complete ionization.
Weak acids and bases are weak electrolytes and conduct electricity poorly due to partial ionization.
The endpoint in titration is detected by a color change of an indicator, which may not exactly match the equivalence point.
