 Back
BackCH.3:Atomic Structure and Electron Configuration: Study Guide for Introductory Chemistry
Study Guide - Smart Notes

Atomic Structure
Dalton’s Atomic Theory
Dalton’s atomic theory laid the foundation for modern chemistry by describing the nature of atoms and their role in chemical reactions. - Atoms are tiny, indestructible particles that make up elements. - All atoms of a given element are identical in mass and properties, distinguishing them from atoms of other elements. - Atoms combine in simple, whole-number ratios to form molecules of compounds. 
Sizes of Atoms
Dalton used the composition of compounds and assumed formulas to determine the relative masses of atoms, establishing the atomic mass unit (amu) with hydrogen as the reference (H = 1 amu). - The atomic mass unit (amu) is the standard unit for atomic mass. - Atoms are extremely small, with absolute sizes on the order of 10-10 meters.
Electrical Charge
Atoms and subatomic particles possess electrical charges, which are fundamental to their interactions. - There are two types of charge: positive and negative. - Opposite charges attract, while like charges repel. - Neutrality is achieved when equal amounts of opposite charges are present. 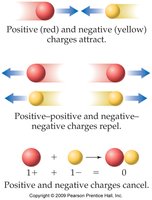
Discovery of Electrons
J. J. Thomson’s experiments revealed the existence of electrons, which are much smaller than atoms and carry a negative charge.
Plum Pudding Model
Thomson proposed the Plum Pudding Model, where electrons are embedded in a sphere of positive charge. - This model suggested that atoms are uniform spheres with electrons scattered throughout. 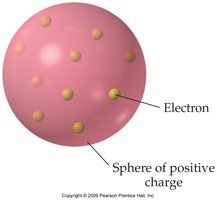
Rutherford’s Nuclear Model
Rutherford’s gold foil experiment demonstrated that atoms have a dense, positively charged nucleus. - Most alpha particles passed through the foil, but some were deflected, indicating a small, dense nucleus. 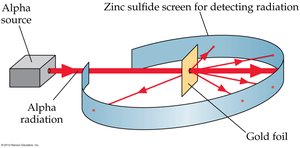
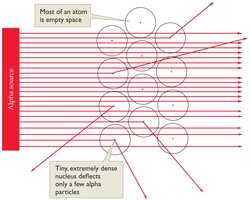 - The nucleus contains nearly all the mass of the atom and is positively charged due to protons. - Electrons occupy the space around the nucleus.
- The nucleus contains nearly all the mass of the atom and is positively charged due to protons. - Electrons occupy the space around the nucleus.
Nature of Electrical Charge
Electrical charge is a fundamental property of protons and electrons. - Protons are positively charged, electrons are negatively charged, and neutrons are neutral. - When protons and electrons are paired, the result is a neutral atom. 
The Atomic Nucleus
Subatomic Particles
Atoms are composed of three main subatomic particles: protons, neutrons, and electrons. - Protons: Positively charged, mass ≈ 1 amu - Neutrons: Neutral, mass ≈ 1 amu - Electrons: Negatively charged, mass ≈ 0.00055 amu
Atomic Number and Mass Number
- The atomic number (Z) is the number of protons in the nucleus and uniquely identifies an element. - The mass number (A) is the sum of protons and neutrons in the nucleus. 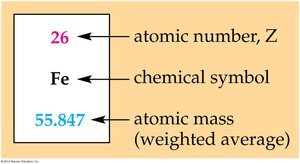
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different mass numbers. - All isotopes of an element are chemically identical but differ in mass. - Example: Hydrogen has three isotopes—protium (1H), deuterium (2H), and tritium (3H). - The atomic mass of an element is a weighted average of its isotopes’ masses. 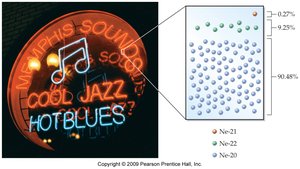
Symbols for Isotopes
Isotopes are represented by their element symbol, atomic number, and mass number.
Electron Arrangement
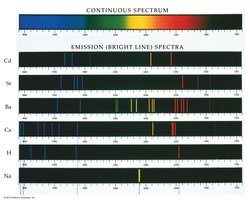
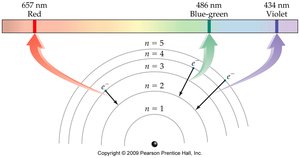
The Bohr Model
The Bohr model explains the arrangement of electrons in discrete energy levels (shells) around the nucleus. - Electrons occupy specific energy levels, and transitions between levels result in emission or absorption of light. - Colored flames are due to line spectra, which correspond to electron transitions. 

Ground and Excited States
- Ground state: All electrons are in their lowest possible energy level. - Excited state: Electrons have absorbed energy and moved to higher levels. - Relaxation: Electrons return to lower energy levels, emitting quantized energy as light. 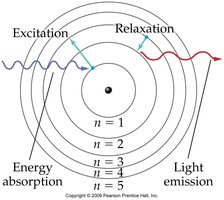
The Quantum Model
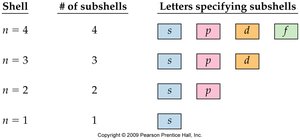
The quantum model describes electron arrangement using shells, subshells, and orbitals. - Each shell (n) contains subshells (s, p, d, f), which are further divided into orbitals.
Shell | # of Subshells | Letters Specifying Subshells |
|---|---|---|
n = 1 | 1 | s |
n = 2 | 2 | s, p |
n = 3 | 3 | s, p, d |
n = 4 | 4 | s, p, d, f |
Orbitals and Probability Maps
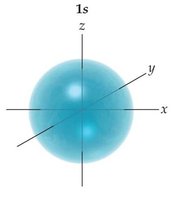
- Orbitals are regions of space where electrons are likely to be found. - s orbitals are spherical, p orbitals are dumbbell-shaped, d orbitals have more complex shapes. 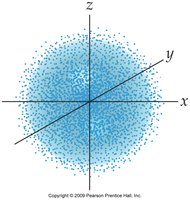
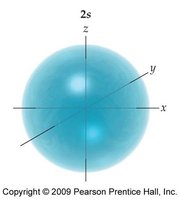

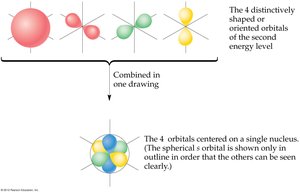
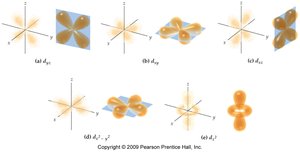
Subshell Energies and Electron Configuration
- Subshells have different energies: s < p < d < f. - Each subshell contains a specific number of orbitals: s (1), p (3), d (5), f (7). - Maximum electrons per subshell: s = 2, p = 6, d = 10, f = 14. 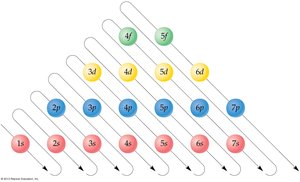
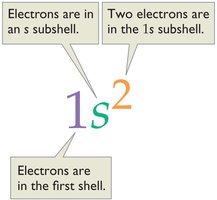
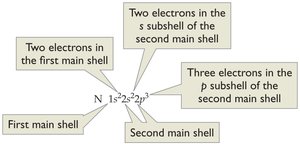

Electron Configuration Examples
- Electron configuration shows the distribution of electrons among shells and subshells. - Example for manganese (Mn, Z = 25): - Example for potassium (K, Z = 19): 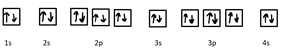 - Example for Sc3+ (Sc, Z = 21, 18 electrons):
- Example for Sc3+ (Sc, Z = 21, 18 electrons): 
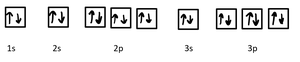 - Example for F− (F, Z = 9, 10 electrons):
- Example for F− (F, Z = 9, 10 electrons): 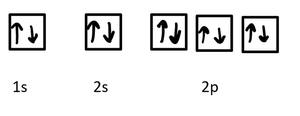
Valence and Core Electrons
- Valence electrons are in the outermost principal energy level and are involved in chemical reactions. - Core electrons are in lower energy shells.
Electron Configuration and the Periodic Table
Periodic Table Structure
The periodic table organizes elements by atomic number and electron configuration. - Groups (families): Vertical columns with elements sharing similar properties. - Periods: Horizontal rows showing a range from metallic to nonmetallic properties. 
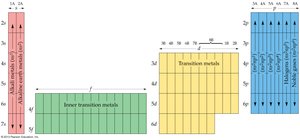
Valence Electrons and Reactivity
- Elements in the same group have the same number of valence electrons. - Valence electrons determine chemical reactivity.
Noble Gas Electron Configuration
- Noble gases have stable electron configurations with 8 valence electrons (except He, which has 2). - Noble gases are non-reactive due to their stable configurations. 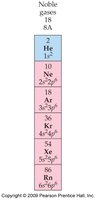
Alkali Metals and Halogens
- Alkali metals (Group 1) tend to lose one electron to achieve noble gas configuration, forming cations (1+ charge). 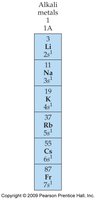 - Halogens (Group 17) tend to gain one electron to achieve noble gas configuration, forming anions (1− charge).
- Halogens (Group 17) tend to gain one electron to achieve noble gas configuration, forming anions (1− charge). 
Periodic Trends
Atomic Size
- Atomic size increases down a group and decreases across a period. - This is due to the addition of energy levels and increased nuclear charge. 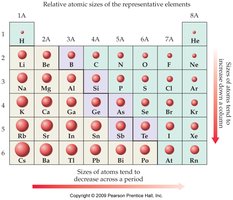
Ionization Energy
- Ionization energy decreases down a group and increases across a period. - Higher ionization energy means it is harder to remove an electron. 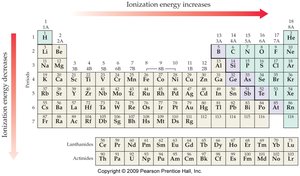
Metallic Character
- Metallic character increases down a group and decreases across a period. - Metals are malleable, ductile, shiny, and conduct electricity; nonmetals are brittle, dull, and insulators.
Practice and Application
Practice Problems
- Choose the larger atom, higher ionization energy, or more metallic element in pairs based on periodic trends.
Summary Table: Subatomic Particles
Particle | Charge | Mass (amu) | Location |
|---|---|---|---|
Proton | +1 | 1 | Nucleus |
Neutron | 0 | 1 | Nucleus |
Electron | -1 | 0.00055 | Outside nucleus |
Summary Table: Electron Subshells
Subshell | Number of Orbitals | Maximum Electrons |
|---|---|---|
s | 1 | 2 |
p | 3 | 6 |
d | 5 | 10 |
f | 7 | 14 |
Additional info: Academic context was added to clarify atomic structure, electron configuration, periodic trends, and the role of subatomic particles, as well as to expand on brief points and provide self-contained explanations suitable for exam preparation.
