 Back
BackAtomic Structure and the Periodic Table: Study Notes
Study Guide - Smart Notes

Atomic Structure and the Periodic Table
Early Models of Atomic Structure
The concept of the atom has evolved through scientific discoveries. Early models proposed that atoms were indivisible particles, but later experiments revealed subatomic structure.
JJ Thompson (1897): Discovered the electron, leading to the 'plum pudding' model where electrons are embedded in a sphere of positive charge.
Goldstein (1886): Discovered positive particles (protons).
Millikan (1909): Determined the charge of the electron.
Roentgen (1895): Discovered X-rays.
Bequerel & Marie Curie (1895): Discovered radioactivity.
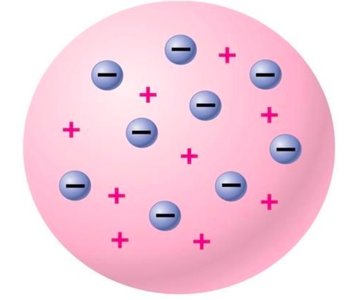
Additional info: The plum pudding model was later disproved by Rutherford's experiments.
Radiation and Radioactivity
Radiation is energy transmitted as waves or particles. Radioactivity is the spontaneous emission of radiation from unstable elements.
Ionizing Radiation: Has enough energy to remove electrons from atoms, creating ions. Types include alpha particles, beta particles, gamma rays, and X-rays.
Health Effects: Ionizing radiation can damage biological tissue, leading to mutations or cancer.


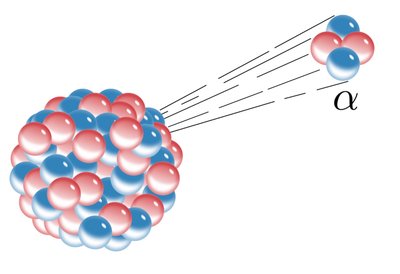
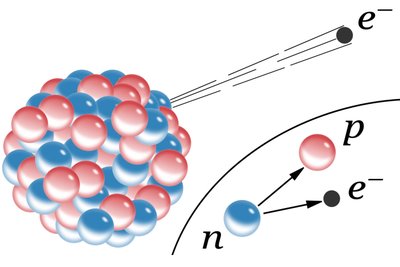
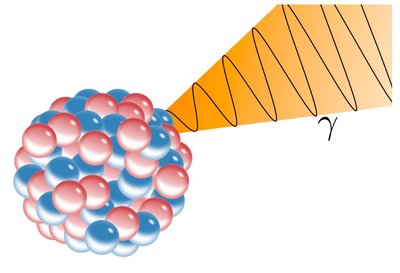
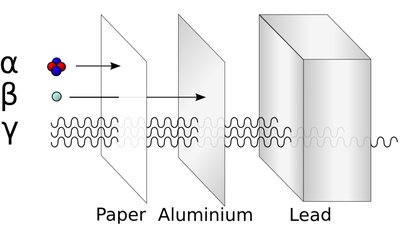

Additional info: Alpha particles are stopped by paper, beta by aluminum, and gamma rays require lead for shielding.
Rutherford’s Gold Foil Experiment and the Nuclear Model
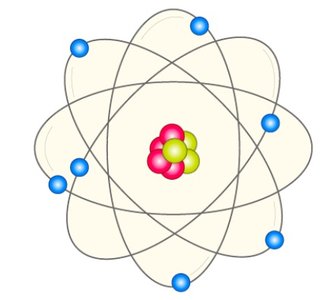
Rutherford's experiment demonstrated that atoms have a small, dense, positively charged nucleus, with electrons orbiting around it.
Nucleus: Contains protons (positive charge) and neutrons (no charge).
Electrons: Negatively charged particles orbiting the nucleus.
Electrostatic Forces: Hold electrons and nucleus together.


Additional info: Most of the atom's volume is empty space.
Subatomic Particles
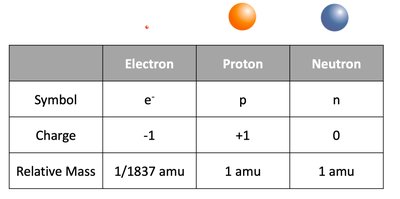
Atoms are composed of three main subatomic particles: electrons, protons, and neutrons.
Particle | Symbol | Charge | Relative Mass |
|---|---|---|---|
Electron | e- | -1 | 1/1837 amu |
Proton | p | +1 | 1 amu |
Neutron | n | 0 | 1 amu |
Isotopes
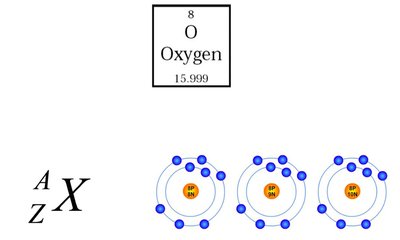
Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons.
Atomic Number (Z): Number of protons in the nucleus.
Mass Number (A): Total number of protons and neutrons.
Notation: where X is the element symbol.

Additional info: Isotopes have identical chemical properties but different physical properties.
Atomic Mass and Isotopic Abundance
The atomic mass listed in the periodic table is the weighted average of all naturally occurring isotopes of an element.
Calculation: Atomic mass = (fractional abundance × mass of isotope) summed for all isotopes.
Electron Arrangement: Bohr and Quantum Models
Electrons occupy energy levels (shells) around the nucleus. The Bohr model describes electrons in fixed orbits, while the quantum model uses orbitals.
Bohr Model: Electrons in circular orbits; maximum electrons per shell given by .
Quantum Model: Electrons behave as both particles and waves; orbitals are regions of probability.
Principal Energy Levels: Shells (n) divided into sublevels (s, p, d, f).
Electron Configuration: Describes the arrangement of electrons in shells and subshells.

Example: Electron configuration for sodium (Na): 1s22s22p63s1
Additional info: The quantum model explains chemical behavior and periodic trends.
Atomic Spectra and Flame Tests
When electrons absorb energy, they move to higher energy levels (excited state). Returning to lower levels, they emit photons, producing line spectra.
Continuous Spectrum: Produced by white light passing through a prism.
Line Spectrum: Produced by excited atoms; unique to each element.
Flame Test: Different elements emit characteristic colors when heated in a flame.



The Periodic Table
The periodic table organizes elements by increasing atomic number. Elements in the same group have similar chemical properties.
Groups (Columns): Elements with similar properties; same number of valence electrons.
Periods (Rows): Elements with increasing atomic number; properties change from metallic to nonmetallic.
Group A: Main group elements.
Group B: Transition elements.
Valence Electrons: Electrons in the outermost shell; determine chemical reactivity.
Classification of Elements
Elements are classified as metals, non-metals, or metalloids based on their properties.
Metals: Shiny, malleable, good conductors.
Non-metals: Dull, brittle, poor conductors.
Metalloids: Properties intermediate between metals and non-metals.
Groups of the Periodic Table
Specific groups have characteristic electron configurations and reactivity.
Group 1A (Alkali Metals): ns1 configuration; highly reactive.
Group 2A (Alkaline Earth Metals): ns2 configuration; moderately reactive.
Group 7A (Halogens): ns2np5 configuration; form salts with alkali metals.
Group 8A (Noble Gases): ns2np6 configuration; very stable.
Synthetic Elements
Synthetic elements are created in particle accelerators and are all radioactive.
Practice: Electron Configurations
Writing electron configurations helps understand element properties and periodic trends.
Sodium (Na): 1s22s22p63s1
Potassium (K): 1s22s22p63s23p64s1
Chlorine (Cl): 1s22s22p63s23p5
Bromine (Br): 1s22s22p63s23p64s23d104p5
Neon (Ne): 1s22s22p6
Argon (Ar): 1s22s22p63s23p6
Ground State vs Excited State
Electrons in the ground state occupy the lowest energy levels. When excited, electrons move to higher energy levels.
Ground State: Most stable electron arrangement.
Excited State: Higher energy arrangement; electrons return to ground state by emitting photons.
Summary Table: Subatomic Particles
Particle | Symbol | Charge | Relative Mass |
|---|---|---|---|
Electron | e- | -1 | 1/1837 amu |
Proton | p | +1 | 1 amu |
Neutron | n | 0 | 1 amu |
Summary Table: Isotopic Abundance
Element | Isotope | Abundance [%] |
|---|---|---|
Hydrogen | 1H | 99.985 |
Hydrogen | 2H | 0.015 |
Oxygen | 16O | 99.76 |
Oxygen | 17O | 0.04 |
Oxygen | 18O | 0.2 |
Carbon | 12C | 98.9 |
Carbon | 13C | 1.1 |
Key Equations
Maximum electrons per shell:
Atomic mass calculation:
