 Back
BackAtoms and Atomic Structure: Fundamental Concepts for Introductory Chemistry
Study Guide - Smart Notes

Atoms and Atomic Structure
Overview
This section introduces the foundational concepts of atoms and atomic structure, including scientific laws, atomic theory, the mole, molar mass, and the periodic table. It also covers the development of atomic models, the role of electricity in understanding atoms, and the arrangement of electrons within atoms.
Atoms
Definition and Historical Context
An atom is the smallest unit of an element that retains the properties of that element and cannot be broken down further by chemical means. Atoms of different elements are distinct from one another. The concept of the atom originated with ancient Greek philosophers, notably Democritus, who described matter as composed of indivisible particles called atomos. Aristotle, however, believed matter was continuous, a view that delayed atomic theory's development for centuries.
Atoms are extremely small: For example, a 1 oz gold coin contains approximately gold atoms.
Atoms of elements are unique: Each element has its own type of atom.
Historical development: The Age of Enlightenment in the 1700s saw the rise of quantitative methods and the study of gases, leading to modern chemistry.
Scientific Laws
Law of Conservation of Mass
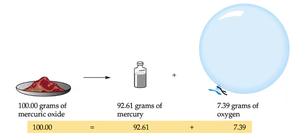
Antoine-Laurent Lavoisier established the law of conservation of mass, which states that matter is neither created nor destroyed during a chemical reaction. The total mass of reactants equals the total mass of products.
Equation:
Example: Burning a log of wood weighing 450 g with 10 g of oxygen produces 400 g of ashes; the missing mass is accounted for by gases released during combustion.
Law of Definite Proportions
Joseph Louis Proust discovered that a compound always contains the same elements in specific proportions by mass, regardless of its source. This is known as the law of definite proportions.
Example: Copper carbonate always contains 57.48% Cu, 5.43% C, 0.91% H, and 36.18% O by mass.


Law of Multiple Proportions
John Dalton expanded on the law of definite proportions with the law of multiple proportions, stating that elements can combine in different ratios to form different compounds, and these ratios are simple whole numbers.
Example: Carbon and oxygen can form CO ( ratio) or CO2 ( ratio).
Dalton's Atomic Theory
Key Postulates
Dalton's atomic theory provided a framework for understanding chemical reactions and the composition of matter:
All matter is composed of extremely small, indestructible particles called atoms.
Each element consists of one type of atom, unique to that element.
Compounds are formed when atoms of different elements combine in fixed proportions.
Chemical reactions involve the rearrangement of atoms; atoms are not created or destroyed.
The Mole and Molar Mass
Quantifying Atoms
Because atoms are so small, chemists use the mole as a counting unit. One mole contains particles (Avogadro's number).
Example: 12.011 g of carbon contains atoms.
Molar mass: The mass of one mole of a substance, found on the periodic table as the atomic mass (in amu).
Equation:
The Periodic Table
Organization and Properties
The periodic table organizes elements by increasing atomic number and groups elements with similar chemical properties. Dmitri Mendeleev developed the periodic law, predicting properties of undiscovered elements.
Atomic number (Z): Number of protons in the nucleus.
Atomic mass: Mass of one atom in amu.
Chemical symbol: One- or two-letter abbreviation for each element.
Atomic Structure
Electricity and the Atom
Electricity played a crucial role in understanding atomic structure. Electrolysis demonstrated that water could be decomposed into hydrogen and oxygen, revealing its compound nature.
Electrolysis: Chemical reaction caused by electricity.
Electrolyte: Compound that conducts electricity.
Electrode: Conducts electric current; anode (+), cathode (–).
Ion: Atom or group of atoms with an electric charge; cation (+), anion (–).
Cathode Ray Tube and Discovery of the Electron
Crookes and Thomson used cathode ray tubes to discover the electron, a negatively charged particle present in all atoms. Millikan determined the electron's charge and mass.
Electron: Negatively charged particle ( g).
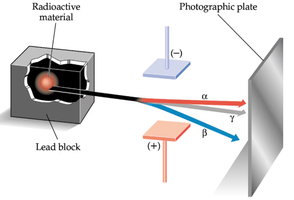
Radioactivity
Radioactivity is the spontaneous emission of radiation from unstable elements. Three types of radiation were identified:
Alpha particles (α): Heavy, positively charged.
Beta particles (β): High-energy electrons.
Gamma rays (γ): Highly energetic, not deflected by magnetic fields.
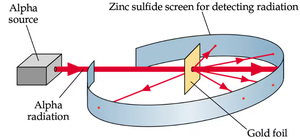
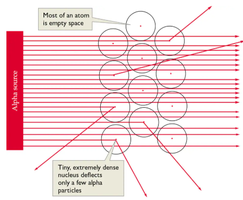
Rutherford's Gold Foil Experiment
Rutherford bombarded gold foil with alpha particles, observing that most passed through but some were deflected. This led to the conclusion that atoms are mostly empty space with a tiny, dense, positively charged nucleus.
Atomic nucleus: Contains protons and neutrons; nearly all the atom's mass.
Proton: Positively charged, mass similar to hydrogen atom.
Neutron: No charge, mass similar to proton.
Isotopes: Atoms of the same element with different numbers of neutrons.
Electron Arrangement and Configuration
Energy Levels and Orbitals
Bohr proposed that electrons occupy specific energy levels (shells) around the nucleus. Electrons fill the lowest energy levels first (ground state) and can move to higher levels (excited state) by absorbing energy. Schrodinger later described electrons as occupying orbitals, regions of space within shells.
Maximum electrons per shell: , where n is the energy level.
Orbitals: s, p, d, f; each can hold 2 electrons.
Subshells: Shells may contain multiple orbitals.
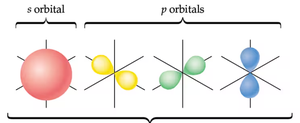
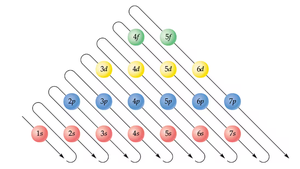
Electron Configuration
Electron configuration describes the arrangement of electrons in an atom. Electrons fill lower energy subshells first, following the Aufbau principle.
Example: Hydrogen: 1s1; Helium: 1s2; Lithium: 1s22s1
Periodic table: Rows (periods) and columns (groups) reflect electron configurations and chemical properties.
Valence electrons: Outermost electrons, determine chemical reactivity.
Summary Table: Atomic Structure and Laws
Law/Theory | Description | Example |
|---|---|---|
Law of Conservation of Mass | Mass is conserved in chemical reactions | Mercuric oxide decomposition: 100.00 g = 92.61 g + 7.39 g |
Law of Definite Proportions | Compounds have fixed element ratios | Copper carbonate always has same % composition |
Law of Multiple Proportions | Elements combine in simple ratios to form different compounds | CO and CO2 have different C:O ratios |
Dalton's Atomic Theory | Atoms are indivisible, unique to each element, combine in fixed ratios | Water: H2O, carbon dioxide: CO2 |
Additional info:
Some explanations and examples were expanded for clarity and completeness, including the historical context of atomic theory, the role of electricity in atomic structure, and the details of electron configuration. The summary table was inferred to provide a concise comparison of key laws and theories.
