 Back
BackAtoms and Elements: Foundations of Atomic Theory and the Periodic Table
Study Guide - Smart Notes

Atoms & Elements
Introduction to Elements and Atomic Symbols
The study of chemistry begins with understanding elements and their atomic structure. Elements are pure substances consisting of only one type of atom, each represented by a unique symbol on the periodic table. Memorizing the names and symbols of common elements is essential for success in introductory chemistry.
Element Symbols: Each element is represented by a one- or two-letter symbol, often derived from its English or Latin name (e.g., Co for cobalt, CO for carbon monoxide).
Importance of Memorization: Many periodic tables used in exams do not include element names, so memorizing at least 50 common elements is recommended.
Lab Quizzes: Memorization of element names and symbols is often assessed through quizzes.

Atomic Theory: Historical Development
Early Ideas about Atoms
The concept of the atom has evolved over centuries, beginning with ancient philosophers and culminating in modern atomic theory.
Aristotle: Believed matter was infinitely divisible and not composed of discrete particles.
Democritus: Proposed that matter is made of tiny, indivisible particles called "atomos." This idea laid the groundwork for the atomic theory.

Dalton's Atomic Theory
In the early 1800s, John Dalton formulated the first modern atomic theory, which remains foundational in chemistry today.
Key Postulates:
Matter is composed of indivisible atoms.
Each element consists of identical atoms unique to that element.
Compounds are formed by combinations of atoms in fixed ratios.
Some aspects of Dalton's theory have been refined, but the core ideas remain accepted.

Models of the Atom
Dalton's model depicted atoms as solid, indivisible spheres, similar to billiard balls. This model was later refined as new discoveries were made.
JJ Thomson: Discovered the electron using cathode ray experiments, showing that atoms contain smaller, negatively charged particles.
Rutherford: Discovered the nucleus through the gold foil experiment, revealing that atoms have a dense, positively charged center.


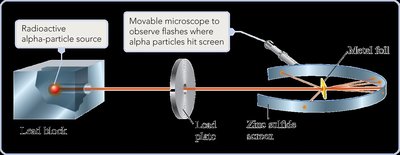
Structure of the Atom
Subatomic Particles
Atoms are composed of three main subatomic particles: protons, neutrons, and electrons. Their arrangement and properties determine the behavior of elements.
Protons: Positively charged particles found in the nucleus.
Neutrons: Neutral particles also located in the nucleus.
Electrons: Negatively charged particles that orbit the nucleus in electron clouds.
Relative Mass: Protons and neutrons have similar masses, while electrons are much lighter.

Relative Size and Charge
Despite their tiny size, electrons have a charge equal in magnitude but opposite in sign to protons. The mass difference between protons and electrons is enormous.
Analogy: If a proton were the size of a bowling ball, an electron would be the size of a penny.
Neutrality: Atoms are electrically neutral when the number of protons equals the number of electrons.

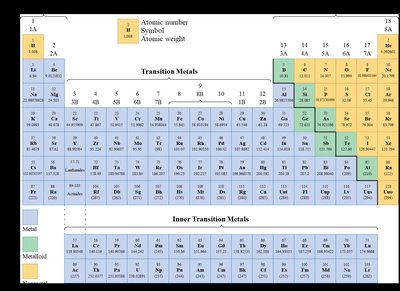
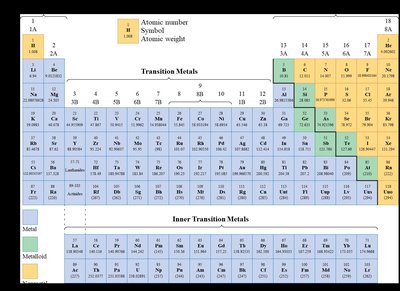
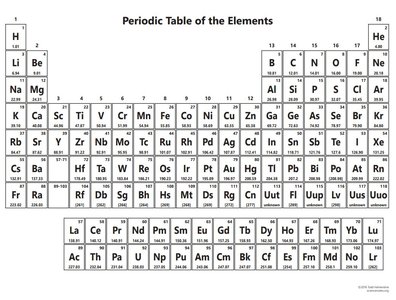
The Periodic Table
Organization and Classification
The periodic table arranges elements by increasing atomic number and groups them based on similar chemical properties.
Periods: Horizontal rows on the table.
Groups (Families): Vertical columns; elements in the same group have similar properties.
Main Classes: Metals, metalloids, and nonmetals.
Special Groups: Alkali metals (Group 1), halogens (Group 17), and noble gases (Group 18).
Patterns and Properties
Elements in the same group exhibit similar chemical behaviors due to their valence electrons. The periodic table is a powerful tool for predicting element properties.
Valence Electrons: The outermost electrons, responsible for chemical bonding and reactivity.
Group Trends: Elements in the same group have the same number of valence electrons.
Atomic Number, Mass Number, and Isotopes
Atomic Number (Z) and Mass Number (A)
The atomic number (Z) is the number of protons in an atom and defines the element. The mass number (A) is the sum of protons and neutrons in the nucleus.
Atomic Number (Z): Determines the identity of the element.
Mass Number (A):
Isotopes: Atoms of the same element with different numbers of neutrons (and thus different mass numbers).
Isotope Notation and Nuclide Symbols
Isotopes are represented by their element name followed by the mass number (e.g., Carbon-12, Carbon-13). The nuclide symbol provides a concise way to specify isotopes.
Nuclide Symbol Format: , where X is the element symbol, A is the mass number, and Z is the atomic number.
Example: for Uranium-235.
Ions and Their Formation
Atoms vs. Ions
Atoms are neutral when they have equal numbers of protons and electrons. When electrons are gained or lost, the atom becomes an ion.
Cations: Positively charged ions formed by losing electrons.
Anions: Negatively charged ions formed by gaining electrons.
Valence Electrons: The electrons involved in ion formation and chemical bonding.
Average Atomic Mass
Atomic Mass Unit (amu)
The atomic mass unit (amu) is a standard unit for expressing atomic and molecular masses, defined as one-twelfth the mass of a carbon-12 atom.
1 amu: Approximately equal to the mass of a proton or neutron.
Calculating Average Atomic Mass
The atomic mass listed on the periodic table is a weighted average of all naturally occurring isotopes of an element, based on their relative abundances.
Formula:
Relative Abundance: Expressed as a decimal (e.g., 75% = 0.75).
Example: Chlorine has two main isotopes, Cl-35 and Cl-37. The average atomic mass is calculated using their masses and relative abundances.

Summary Table: Properties of Subatomic Particles
Particle | Mass (kg) | Charge (C) | Mass (amu) | Charge (e) |
|---|---|---|---|---|
Electron | 9.10938 × 10−31 | −1.60218 × 10−19 | 0.00055 | −1 |
Proton | 1.67262 × 10−27 | +1.60218 × 10−19 | 1.00728 | +1 |
Neutron | 1.67493 × 10−27 | 0 | 1.00866 | 0 |

Additional info: This guide covers the foundational concepts of atomic structure, the periodic table, isotopes, ions, and average atomic mass, providing a comprehensive overview for introductory chemistry students.
