 Back
BackAtoms and Elements: Foundations of Chemistry
Study Guide - Smart Notes

Atoms and Elements - CH. 4
Introduction to Atoms and Elements
Atoms are the fundamental building blocks of matter. The properties of atoms determine the properties of all substances. An atom is the smallest identifiable unit of an element, and an element is a substance that cannot be broken down into simpler substances by chemical means. There are about 91 naturally occurring elements, each with unique types of atoms.
Atoms compose all matter and are extremely small and numerous.
Elements are defined by their atomic structure and cannot be chemically decomposed.
Scientists have also created about 20 synthetic elements.
Historical Development of Atomic Theory
The concept of atoms dates back to ancient Greece, where philosophers like Democritus and Leucippus theorized that matter was made of tiny, indivisible particles called atomos. However, it was not until John Dalton (1808) that a scientific atomic theory was widely accepted.
Dalton’s Atomic Theory:
Each element is composed of tiny, indestructible particles called atoms.
All atoms of a given element have the same mass and properties.
Atoms combine in simple, whole-number ratios to form compounds.
Modern Evidence for Atomic Theory
Modern technology, such as the scanning tunneling microscope (STM), allows scientists to manipulate and visualize individual atoms, providing direct evidence for their existence.
Structure of the Atom
Discovery of Subatomic Particles
Atoms are not indivisible; they are composed of smaller particles: electrons, protons, and neutrons.
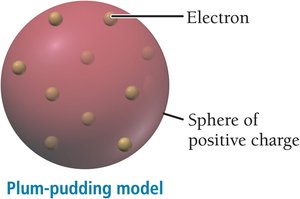
Electrons are negatively charged, much smaller and lighter than atoms, and are present in all substances (discovered by J. J. Thomson).
Thomson’s plum-pudding model proposed that electrons are embedded in a sphere of positive charge.
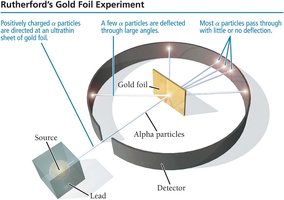
Rutherford’s Gold Foil Experiment and Nuclear Model
Ernest Rutherford’s gold foil experiment (1909) demonstrated that atoms have a small, dense, positively charged nucleus. Most alpha particles passed through gold foil, but some were deflected, indicating a concentrated center of mass and charge.
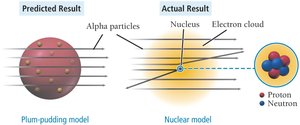
Rutherford’s nuclear model states:
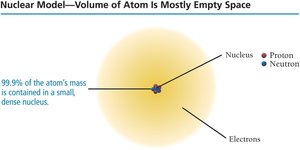
Most of the atom’s mass and all its positive charge are in the nucleus.
Most of the atom’s volume is empty space occupied by electrons.
The number of electrons equals the number of protons, making the atom electrically neutral.
Subatomic Particles: Mass and Charge
Atoms are composed of three main subatomic particles:
Particle | Symbol | Relative Mass | Charge |
|---|---|---|---|
Proton | p+ | 1 | +1 |
Neutron | n0 | 1 | 0 |
Electron | e- | ~1/2000 | -1 |

Protons and neutrons have similar masses (~1 atomic mass unit, amu).
Electrons have negligible mass compared to protons and neutrons.
Electrical Charge and Interactions
Electrical charge is a fundamental property of protons and electrons:
Opposite charges attract; like charges repel.
Atoms are neutral when they contain equal numbers of protons and electrons.

Evidence of Charge in Matter
Normally, matter is electrically neutral. However, phenomena like lightning demonstrate the effects of charge imbalance in nature.

Elements and the Periodic Table
Atomic Number and Identity of Elements
The atomic number (Z) is the number of protons in the nucleus and defines the element. Changing the number of protons changes the element.
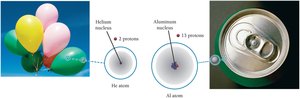
The Periodic Table
The periodic table organizes elements by increasing atomic number. Each element is represented by its name, symbol, and atomic number.
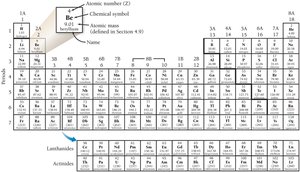
Element Symbols and Names
Element symbols are usually derived from their English or Latin names (e.g., K for potassium from kalium, Na for sodium from natrium).
Origins of Element Names
Element names may reflect properties, countries, or honor scientists. For example, bromine means "stench" in Greek, polonium is named after Poland, and curium honors Marie Curie.


Periodic Law and Patterns
Dmitri Mendeleev discovered that elements show recurring (periodic) properties when arranged by increasing mass, leading to the periodic law. Elements with similar properties are grouped in columns (families or groups).
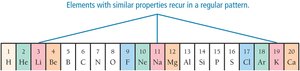
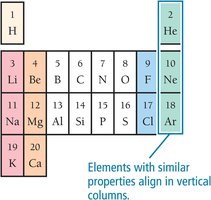
Classification of Elements
Elements are classified as metals, nonmetals, or metalloids based on their properties and position in the periodic table.
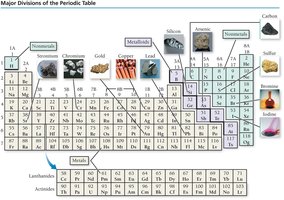
Metals: Good conductors, malleable, ductile, lustrous, tend to lose electrons.
Nonmetals: Poor conductors, varied states, tend to gain electrons.
Metalloids: Intermediate properties, semiconductors, useful in electronics.

Main Group and Transition Elements
The periodic table is divided into main group elements (predictable properties) and transition elements (less predictable properties).
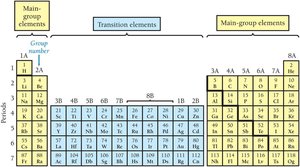
Groups and Families
Columns in the periodic table are called groups or families. Elements in the same group have similar chemical properties.
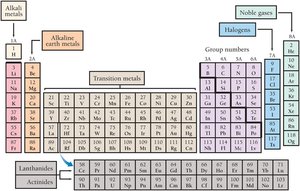
Alkali metals (Group 1A): Very reactive metals.
Alkaline earth metals (Group 2A): Fairly reactive metals.
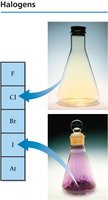
Halogens (Group 7A): Very reactive nonmetals.
Noble gases (Group 8A): Chemically inert gases.
Ions and Isotopes
Formation of Ions
Atoms can gain or lose electrons to form ions. Cations are positively charged (loss of electrons), and anions are negatively charged (gain of electrons). The charge is determined by the difference between the number of protons and electrons.
Cations are named as the element plus "cation" (e.g., sodium cation).
Anions are named with the element stem plus "-ide" (e.g., chloride).
Ions and the Periodic Table
The group number for main-group elements predicts the number of valence electrons and the typical ion charge formed to achieve a noble gas configuration.
Isotopes
Atoms of the same element with different numbers of neutrons are called isotopes. Isotopes are identified by their mass number (A = protons + neutrons). The atomic mass of an element is the weighted average of its isotopes' masses.
Isotope notation: or X-A (e.g., Ne-20, Ne-21, Ne-22 for neon).
Calculating Atomic Mass
The atomic mass is calculated as the weighted average of the masses of all naturally occurring isotopes:
Example: For chlorine, with 75.77% Cl-35 (34.97 amu) and 24.23% Cl-37 (36.97 amu):
Radioactive Isotopes
Some isotopes are unstable and emit nuclear radiation, transforming into other elements or isotopes. These are called radioactive isotopes and have applications in medicine and science, but can also be hazardous due to their radiation.
Summary Table: Subatomic Particles
Particle | Symbol | Relative Mass | Charge |
|---|---|---|---|
Proton | p+ | 1 | +1 |
Neutron | n0 | 1 | 0 |
Electron | e- | ~1/2000 | -1 |
