 Back
BackAtoms and Elements: Foundations of Matter and the Periodic Table
Study Guide - Smart Notes

Atoms and Elements
Experiencing Atoms in the Sea and Mountains
Atoms are the fundamental building blocks of matter, forming the basis of all substances we encounter. For example, seaside rocks are primarily composed of silicates, which are compounds of silicon and oxygen atoms. The air at the seaside contains nitrogen and oxygen molecules, and may also include amines such as triethylamine, which is responsible for the characteristic fishy smell of decaying fish.

How Many Atoms Are in a Pebble?
Atoms are incredibly small, and even a tiny pebble contains more atoms than can be counted. The number of atoms in a pebble far exceeds the number of pebbles on the bottom of a large bay. If each atom in a pebble were the size of the pebble itself, the resulting object would be larger than a mountain such as Mount Everest.

Atomic Theory
"Indivisible": The Atomic Theory
The concept that matter is composed of small, indivisible particles called atoms dates back to ancient Greece. Democritus and Leucippus proposed that matter could be divided only until reaching these fundamental particles, which they called atomos, meaning "indivisible." In the early 19th century, John Dalton formalized the atomic theory, which states:
Each element is composed of tiny, indestructible particles called atoms.
All atoms of a given element have the same mass and properties that distinguish them from atoms of other elements.
Atoms combine in simple, whole-number ratios to form compounds.

Modern Evidence for the Atomic Theory
Modern technology, such as the scanning tunneling microscope (STM), allows scientists to manipulate and visualize individual atoms. For example, scientists have arranged xenon atoms to spell out "IBM," providing direct evidence for the existence of atoms.

Discovery of Subatomic Particles
Thomson’s Discovery of Electrons
J. J. Thomson discovered the electron, a negatively charged particle much smaller and lighter than an atom. He proposed the "plum pudding" model, where electrons are embedded in a sphere of positive charge, balancing the overall charge of the atom.

Rutherford’s Gold Foil Experiment
Ernest Rutherford’s gold foil experiment demonstrated that atoms have a small, dense nucleus. Alpha particles directed at a thin gold foil mostly passed through, but some were deflected at sharp angles, indicating a concentrated positive charge in the nucleus.
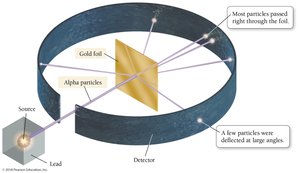
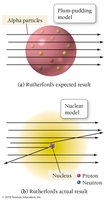
Nuclear Theory of the Atom
Most of the atom’s mass and all of its positive charge are contained in a small core called the nucleus.
Most of the atom’s volume is empty space, with electrons dispersed throughout.
The number of electrons equals the number of protons, making the atom electrically neutral.
The nucleus is extremely dense, containing over 99.9% of the atom’s mass but occupying a tiny fraction of its volume.
Subatomic Particles: Protons, Neutrons, and Electrons
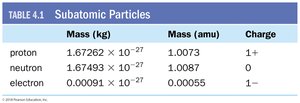
Properties of Subatomic Particles
Protons have a positive charge (+1) and a mass of approximately 1 atomic mass unit (amu).
Neutrons have no charge and a mass similar to protons (about 1 amu).
Electrons have a negative charge (–1) and a much smaller mass (about 0.00055 amu).

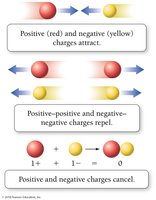
Electrical Charge and Interactions
Opposite charges attract; like charges repel.
When protons and electrons combine, their charges cancel, resulting in a neutral atom.
Charge Neutrality in Matter
Most matter is electrically neutral, with equal numbers of positive and negative charges. Disturbances in this balance, such as during electrical storms, can result in dramatic phenomena like lightning.

Atoms of the Elements
Atomic Number and the Periodic Table
The number of protons in the nucleus of an atom defines the element and is called the atomic number (Z). Each element has a unique atomic number, and the periodic table arranges elements in order of increasing atomic number.
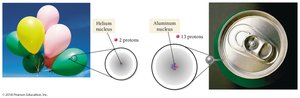
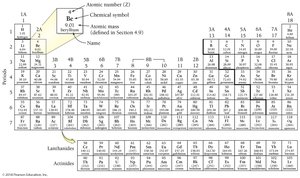
Element Names and Symbols
Element symbols are usually derived from their English or Latin names. For example, potassium (K) comes from kalium, and sodium (Na) from natrium. Some elements are named after countries, scientists, or their properties.


The Periodic Law and the Periodic Table
Mendeleev’s Periodic Law
Dmitri Mendeleev observed that when elements are arranged by increasing relative mass, certain properties recur periodically. This observation led to the development of the periodic law and the modern periodic table.

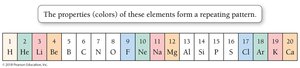
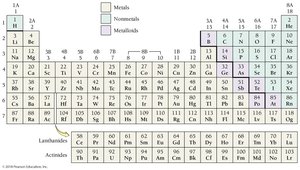
Classification of Elements
Elements are classified as metals, nonmetals, or metalloids based on their properties and position in the periodic table.
Metals: Good conductors, malleable, ductile, lustrous, tend to lose electrons.
Nonmetals: Poor conductors, varied states, tend to gain electrons.
Metalloids: Intermediate properties, semiconductors, useful in electronics.

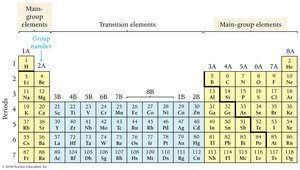
Main Group and Transition Elements
Main group elements have predictable properties based on their position, while transition elements have less predictable properties.
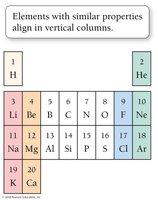
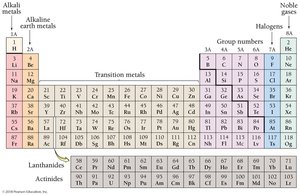
Groups and Families in the Periodic Table
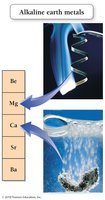
Certain columns in the periodic table are known as groups or families, such as alkali metals (1A), alkaline earth metals (2A), halogens (7A), and noble gases (8A).


Ions: Losing and Gaining Electrons
Formation of Ions
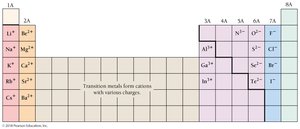
Atoms can lose or gain electrons during chemical reactions, forming charged particles called ions. Positive ions are cations, and negative ions are anions. The charge of an ion is determined by the difference between the number of protons and electrons:
or


Ions and the Periodic Table
The charge of ions formed by main group elements can often be predicted by their group number. Main group elements tend to form ions with the same number of valence electrons as the nearest noble gas.
Isotopes: When the Number of Neutrons Varies
Definition and Notation of Isotopes
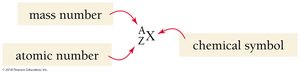
Atoms of the same element with different numbers of neutrons are called isotopes. Isotopes are represented using the following notation:
where A is the mass number, Z is the atomic number, and X is the chemical symbol.
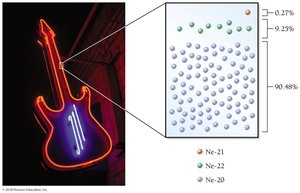
Alternatively, isotopes can be written as the chemical symbol or name followed by a hyphen and the mass number (e.g., Ne-20, neon-20).
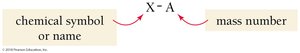
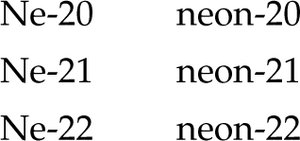
Mass Number and Atomic Number
The mass number (A) is the sum of the number of protons and neutrons in an atom. The number of neutrons is calculated as:
Atomic Mass: The Average Mass of an Element’s Atoms
Calculating Atomic Mass
The atomic mass of an element is the weighted average of the masses of its naturally occurring isotopes. The calculation is as follows:
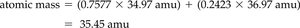
Radioactive Isotopes
Some isotopes are unstable and emit nuclear radiation, changing into different isotopes or elements. These are called radioactive isotopes. While nuclear radiation can be harmful, some radioactive isotopes have beneficial uses, such as in medical imaging.
Chapter Review and Learning Objectives
All matter is composed of atoms.
Experiments by Thomson and Rutherford led to the nuclear theory of the atom.
Protons, neutrons, and electrons have distinct properties and charges.
The periodic table organizes elements by atomic number and properties.
Ions form by gaining or losing electrons; their charge is determined by the difference between protons and electrons.
Isotopes have the same number of protons but different numbers of neutrons; atomic mass is a weighted average of isotopic masses.
