 Back
BackAtoms and Elements: Structure, Classification, and Isotopes
Study Guide - Smart Notes

Atoms and Elements
Definition and Properties of Atoms
An atom is the smallest identifiable unit of an element that retains the chemical properties of that element. Atoms are the building blocks of matter and cannot be broken down by chemical means.
Atoms are composed of subatomic particles: protons, neutrons, and electrons.
Elements are substances made up of only one type of atom and cannot be decomposed into simpler substances.
There are about 91 naturally occurring elements, each with unique atoms; over 20 synthetic elements have been created by scientists.
Atomic Theory: Historical Development
The concept of atoms dates back to ancient philosophers, but the modern atomic theory was formalized by John Dalton in 1808.
Dalton's Atomic Theory states:
Each element is composed of tiny, indestructible particles called atoms.
All atoms of a given element have the same mass and properties that distinguish them from atoms of other elements.
Atoms combine in simple, whole-number ratios to form compounds.
Structure of the Atom
Discovery of Subatomic Particles
Atoms are made up of three main subatomic particles: protons, neutrons, and electrons. The discovery of these particles led to the development of modern atomic models.
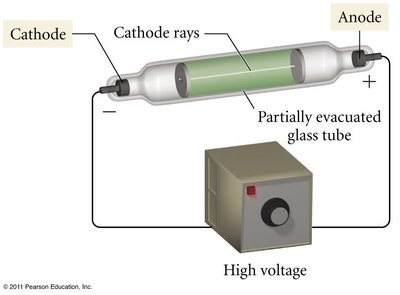
Electrons were discovered by J.J. Thomson using the cathode ray tube experiment.
Electrons are negatively charged and much lighter than atoms.
Thomson's plum pudding model proposed that electrons were embedded in a sphere of positive charge.

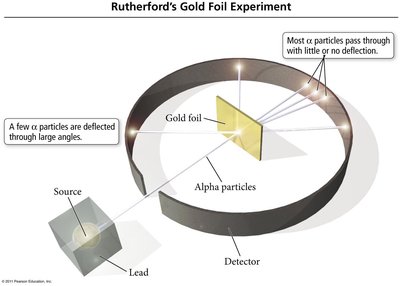
Rutherford's Gold Foil Experiment and Nuclear Model
Ernest Rutherford's gold foil experiment demonstrated that atoms have a small, dense nucleus containing positively charged protons.
Most alpha particles passed through the foil, but some were deflected, indicating a dense nucleus.
Rutherford proposed that the nucleus contains protons and later, neutrons (discovered by Chadwick).
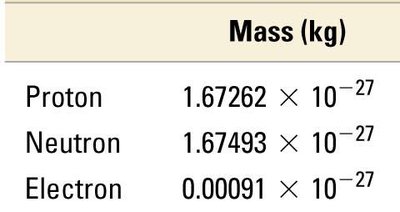
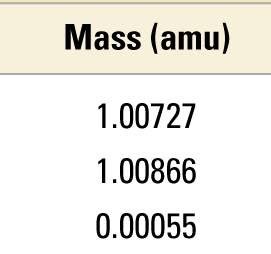
Subatomic Particles: Properties and Comparison
Protons, neutrons, and electrons differ in mass and charge.
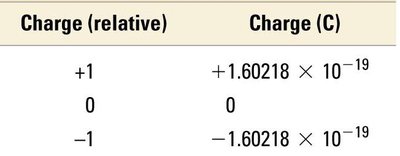
Particle | Mass (kg) | Mass (amu) | Charge (relative) | Charge (C) |
|---|---|---|---|---|
Proton | 1.67262 × 10−27 | 1.00727 | +1 | +1.60218 × 10−19 |
Neutron | 1.67493 × 10−27 | 1.00866 | 0 | 0 |
Electron | 0.00091 × 10−27 | 0.00055 | −1 | −1.60218 × 10−19 |
Electrical Charge and Atomic Neutrality
Interactions of Charges
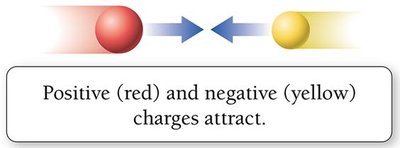
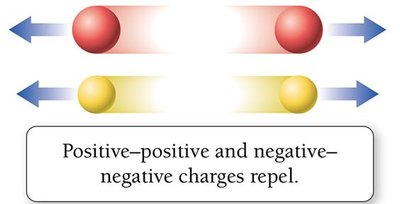
Electrical charge is a fundamental property of protons and electrons.
Opposite charges attract; like charges repel.
Atoms are electrically neutral when the number of protons equals the number of electrons.
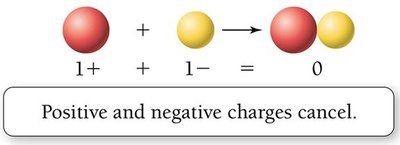
Atomic Number and Element Identity
Defining Elements by Protons
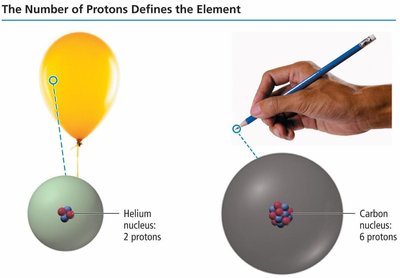
The atomic number (Z) is the number of protons in the nucleus and defines the identity of an element.
All atoms of a given element have the same number of protons.
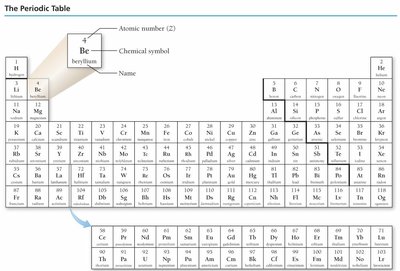
Elements are arranged in the periodic table by atomic number.
The Periodic Table
Organization and Classification
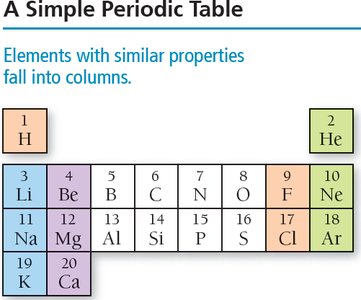
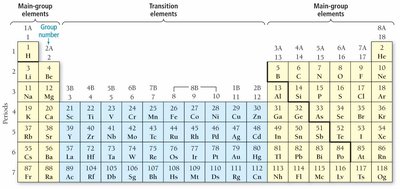
The periodic table arranges elements by increasing atomic number and groups elements with similar properties into columns.
Columns are called groups or families.
Rows are called periods.
Main-group elements have predictable properties; transition elements have less predictable properties.


Classification of Elements
Metals: Good conductors of heat and electricity, malleable, ductile, lose electrons to form cations.
Nonmetals: Poor conductors, brittle solids, gain electrons to form anions.
Metalloids: Exhibit properties intermediate between metals and nonmetals; often semiconductors.
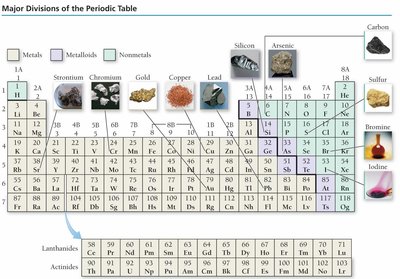
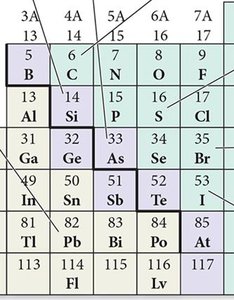
Special Groups in the Periodic Table
Alkali Metals (Group 1A): Highly reactive, form basic solutions with water.
Alkaline Earth Metals (Group 2A): Reactive, form stable oxides.
Halogens (Group 7A): Very reactive nonmetals, form diatomic molecules.
Noble Gases (Group 8A): Inert gases, very low reactivity.


Ions: Cations and Anions
Formation of Ions
Atoms can lose or gain electrons during chemical reactions, forming ions.
Cations: Positively charged ions formed by losing electrons.
Anions: Negatively charged ions formed by gaining electrons.
Example: Lithium atom (3 protons, 3 electrons) loses one electron to form Li+ (3 protons, 2 electrons).
Example: Fluorine atom (9 protons, 9 electrons) gains one electron to form F− (9 protons, 10 electrons).
Isotopes and Atomic Mass
Definition of Isotopes
Isotopes are atoms of the same element (same number of protons) but different numbers of neutrons.
Isotopes have different mass numbers (A = number of protons + number of neutrons).
Example: Neon has three isotopes: Ne-20, Ne-21, Ne-22.
Symbol | Number of Protons | Number of Neutrons | Mass Number (A) | Natural Abundance (%) |
|---|---|---|---|---|
Ne-20 | 10 | 10 | 20 | 90.48 |
Ne-21 | 10 | 11 | 21 | 0.27 |
Ne-22 | 10 | 12 | 22 | 9.25 |

Calculating Atomic Mass
The atomic mass of an element is the weighted average of the masses of its naturally occurring isotopes.
Atomic mass is measured in atomic mass units (amu).
Formula:
Example: Silver has two isotopes, Ag-107 and Ag-109, with respective abundances and masses.
Summary Table: Key Concepts
Concept | Definition | Example |
|---|---|---|
Atom | Smallest unit of an element | Hydrogen atom |
Element | Substance made of one type of atom | Oxygen (O2) |
Isotope | Atoms with same protons, different neutrons | Carbon-12, Carbon-14 |
Cation | Atom that lost electrons | Na+ |
Anion | Atom that gained electrons | Cl− |
Practice Problems
Classify elements as metal, nonmetal, or metalloid.
Determine the number of protons, neutrons, and electrons in ions and isotopes.
Calculate atomic mass from isotope data.
Additional info: Some context and definitions were inferred and expanded for clarity and completeness.
