 Back
BackAtoms and Elements: Structure, Properties, and Periodic Trends
Study Guide - Smart Notes

Atoms and Elements
Elements and Symbols
Elements are pure substances that serve as the fundamental building blocks of all matter. Each element is represented by a unique chemical symbol, often derived from its English or Latin name. Elements cannot be broken down into simpler substances by chemical means.
Element: A substance consisting of only one type of atom.
Chemical Symbol: One or two letters representing an element (e.g., H for hydrogen, O for oxygen).
Source of Names: Element names may originate from mythology, minerals, places, or scientists.
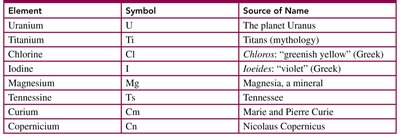
Example Table:
Element | Symbol | Source of Name |
|---|---|---|
Uranium | U | The planet Uranus |
Titanium | Ti | Titans (mythology) |
Chlorine | Cl | Chloros: "greenish yellow" (Greek) |
Iodine | I | Ioiedes: "violet" (Greek) |
Magnesium | Mg | Magnesia, a mineral |
Tennessine | Ts | Tennessee |
Curium | Cm | Marie and Pierre Curie |
Copernicium | Cn | Nicolas Copernicus |
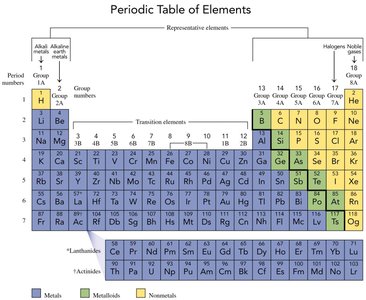
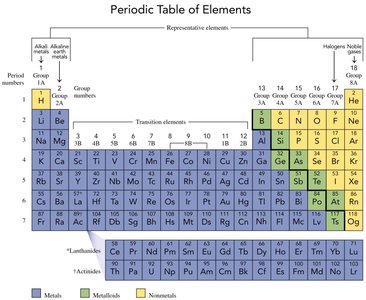
The Periodic Table
The periodic table organizes elements by increasing atomic number and groups elements with similar properties together. It was first developed by Dmitry Mendeleev and now contains 118 elements. Elements in the same group (vertical column) share similar chemical properties.
Groups: Vertical columns; elements in the same group have similar properties.
Periods: Horizontal rows; elements in the same period have the same number of electron shells.
Representative Elements: Groups 1A-8A (1, 2, 13-18).
Transition Elements: Groups 3-12.
Classification of Elements
Elements are classified as metals, nonmetals, or metalloids based on their physical and chemical properties.
Metals: Shiny, malleable, ductile, good conductors of heat and electricity, usually solid at room temperature.
Nonmetals: Not shiny, poor conductors, often gases or brittle solids.
Metalloids: Properties intermediate between metals and nonmetals; often semiconductors.

Groups of Elements
Certain groups in the periodic table have specific names and characteristic properties:
Alkali Metals (Group 1A): Soft, shiny, highly reactive metals (excluding hydrogen).
Alkaline Earth Metals (Group 2A): Shiny, silver-colored, reactive metals with higher melting points than alkali metals.
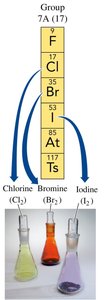
Halogens (Group 7A): Highly reactive nonmetals, form compounds with most elements.
Noble Gases (Group 8A): Unreactive, rarely form compounds.
The Atom
Atomic Structure
An atom is the smallest particle of an element that retains its chemical properties. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in energy levels.
Proton: Positively charged particle in the nucleus.
Neutron: Neutral particle in the nucleus.
Electron: Negatively charged particle in energy levels around the nucleus.
Dalton’s Atomic Theory
Dalton proposed that all matter is made of atoms, which are indivisible and indestructible. Atoms of the same element are identical, and compounds are formed by combining atoms in fixed ratios.
Atoms are rearranged, not created or destroyed, in chemical reactions.
Atoms of different elements combine to form compounds.
Atomic Number and Mass Number
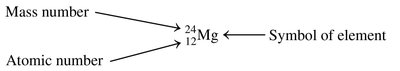
The atomic number is the number of protons in an atom and uniquely identifies the element. The mass number is the sum of protons and neutrons in the nucleus.
Atomic Number (Z): Number of protons.
Mass Number (A): Number of protons plus neutrons.
Atomic Notation:
Isotopes and Atomic Mass
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different mass numbers. The atomic mass of an element is the weighted average of the masses of its naturally occurring isotopes.
Isotope: Atoms with same number of protons but different number of neutrons.
Atomic Mass: Weighted average of isotope masses, not a whole number.
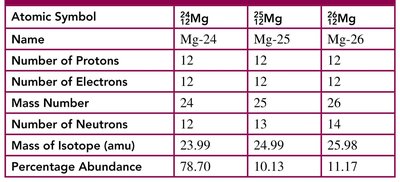
Example Table: Isotopes of Magnesium
Atomic Symbol | Name | Number of Protons | Number of Electrons | Mass Number | Number of Neutrons | Mass of Isotope (amu) | Percentage Abundance |
|---|---|---|---|---|---|---|---|
Mg-24 | 12 | 12 | 24 | 12 | 23.99 | 78.70 | |
Mg-25 | 12 | 12 | 25 | 13 | 24.99 | 10.13 | |
Mg-26 | 12 | 12 | 26 | 14 | 25.98 | 11.17 |
Atomic Mass Calculation:
Electron Energy Levels and Periodic Trends
Electron Energy Levels
Electrons occupy energy levels (shells) around the nucleus. The number of energy levels corresponds to the period number in the periodic table. The outermost electrons are called valence electrons and determine chemical reactivity.
Valence Electrons: Electrons in the outermost energy level.
Energy Levels: Numbered n = 1, 2, 3, ...
Trends in the Periodic Table
Periodic trends describe how properties of elements change across periods and groups.
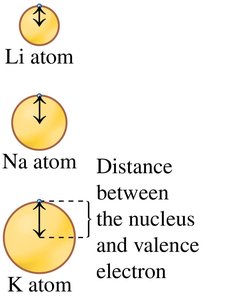
Atomic Size: Increases down a group, decreases across a period.
Ionization Energy: Energy required to remove an electron; decreases down a group, increases across a period.
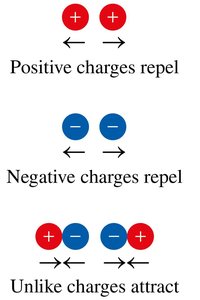
Coulombic Attraction: Attraction between protons and electrons; increases with more protons, decreases with greater distance.
Example: Atomic radius increases from Li to K as more energy levels are added.
Ionization Energy Equation:
where Z is the nuclear charge and r is the distance from the nucleus to the valence electron.
Summary Table: Periodic Trends
Trend | Across a Period (Left to Right) | Down a Group |
|---|---|---|
Atomic Size | Decreases | Increases |
Ionization Energy | Increases | Decreases |
Valence Electrons | Increases | Remains the same |
Example: Magnesium has a smaller atomic size and higher ionization energy than cesium.
Additional info: These notes expand on brief points from the original slides, providing definitions, examples, and context for self-contained study.
