 Back
BackAtoms and Elements: Structure, Properties, and Classification
Study Guide - Smart Notes

Atoms and Elements
Definition and Properties
Atoms are the smallest identifiable units of elements, retaining the properties of the matter from which they originate. Elements are substances composed of a single type of atom and cannot be broken down into simpler substances. There are about 91 naturally occurring elements, with over 20 synthetic elements created by scientists.
Atom: Smallest unit of an element, indivisible in chemical reactions.
Element: Pure substance made of one type of atom.
Example: Gold (Au) is an element made entirely of gold atoms.
Atomic Theory
Historical Development
The concept of atoms originated with Democritus and Leucippus, who theorized that matter is composed of tiny, indestructible particles. John Dalton formalized atomic theory in 1808, which gained broad acceptance.
Dalton's Atomic Theory:
Each element is composed of tiny, indestructible particles called atoms.
All atoms of a given element have the same mass and properties.
Atoms combine in simple, whole-number ratios to form compounds.
Structure of the Atom
Discovery of Subatomic Particles
Atoms are composed of three main subatomic particles: protons, neutrons, and electrons. The discovery of the electron by J.J. Thomson using the cathode ray tube experiment revealed the existence of negatively charged particles within atoms.
Electron: Negatively charged, very small mass.
Proton: Positively charged, similar mass to neutron.
Neutron: No charge, slightly heavier than proton.
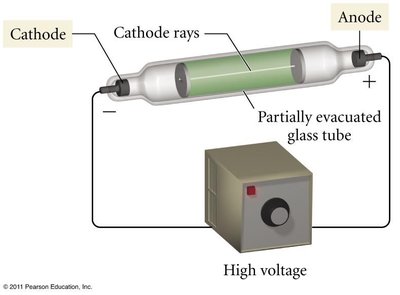
Thomson's Plum Pudding Model
Thomson proposed that electrons are embedded in a sphere of positive charge, like plums in a pudding. This model was later disproved by Rutherford's gold foil experiment.

Rutherford's Gold Foil Experiment
Rutherford's experiment showed that most alpha particles passed through gold foil, but some were deflected, indicating a dense, positively charged nucleus at the center of the atom.
Nucleus: Contains protons and neutrons.
Electrons: Orbit the nucleus.
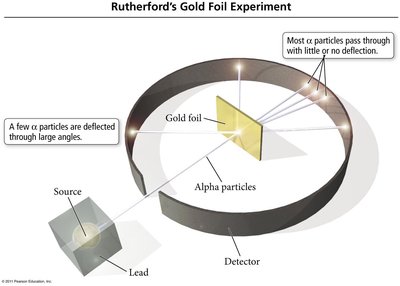
Subatomic Particle Properties
Protons and neutrons have nearly identical masses, while electrons are much lighter. Their charges are equal in magnitude but opposite in sign.
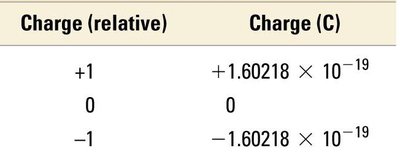
Particle | Mass (kg) | Mass (amu) | Charge (relative) | Charge (C) |
|---|---|---|---|---|
Proton | 1.67262 × 10−27 | 1.00727 | +1 | +1.60218 × 10−19 |
Neutron | 1.67493 × 10−27 | 1.00866 | 0 | 0 |
Electron | 0.00091 × 10−27 | 0.00055 | −1 | −1.60218 × 10−19 |
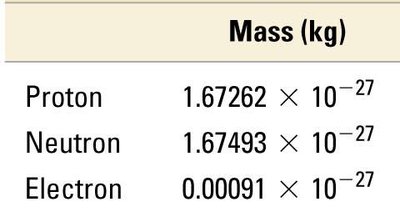
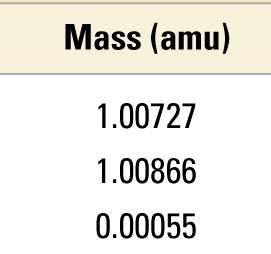
Electrical Charge and Atomic Neutrality
Charge Interactions
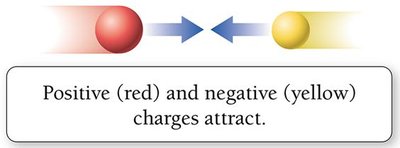
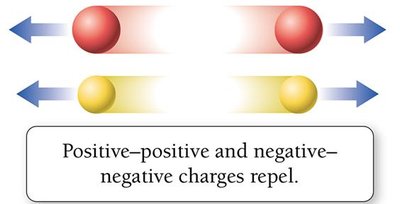
Positive and negative charges attract, while like charges repel. Atoms are neutral when the number of protons equals the number of electrons.
Attraction: Opposite charges attract.
Repulsion: Like charges repel.
Neutrality: Equal numbers of protons and electrons result in a neutral atom.
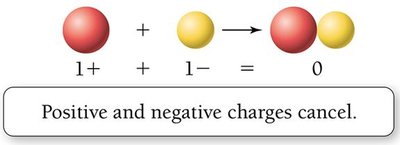
Atomic Number and Element Identity
Defining Elements
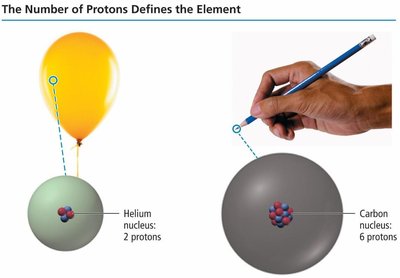
The number of protons in the nucleus, called the atomic number (Z), defines the element. All atoms of a given element have the same number of protons.
Atomic Number (Z): Number of protons in the nucleus.
Example: Helium (Z=2), Carbon (Z=6).
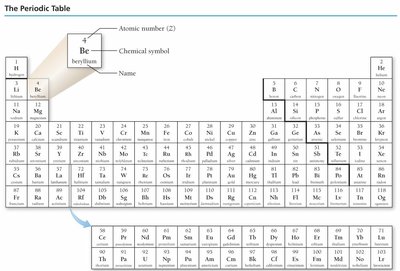
The Periodic Table
Organization and Periodic Law
The periodic table arranges elements by increasing atomic number. Elements with similar properties are grouped in columns (families or groups), and rows are called periods. Dmitri Mendeleev organized the first periodic table, predicting properties of undiscovered elements.
Groups/Families: Columns of elements with similar properties.
Periods: Rows of elements.
Periodic Law: Properties recur in a regular pattern when elements are arranged by atomic number.


Classification of Elements
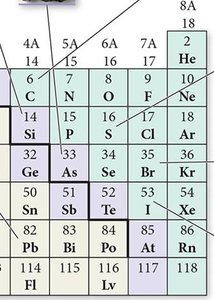
Elements are classified as metals, nonmetals, or metalloids based on their properties. The periodic table is divided into main-group elements and transition elements.
Metals: Shiny, conductive, malleable, ductile, lose electrons to form cations.
Nonmetals: Poor conductors, brittle, gain electrons to form anions, found in all three states.
Metalloids: Show properties of both metals and nonmetals, often semiconductors.





Groups of Elements
Alkali Metals (Group IA)
Alkali metals are highly reactive, never found uncombined in nature, and react with water to form basic solutions and hydrogen gas.
Example Reaction:

Alkali Earth Metals (Group IIA)
Alkali earth metals are harder, denser, and less reactive than alkali metals. Their oxides are basic and insoluble.

Halogens (Group VIIA)
Halogens are very reactive nonmetals, found as diatomic molecules in various states at room temperature. They react with metals to form ionic compounds.
Example Reaction:
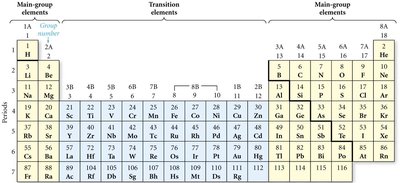
Noble Gases (Group VIIIA)
Noble gases are unreactive, all gaseous at room temperature, and have very low melting and boiling points.
Ions: Losing and Gaining Electrons
Formation of Ions
Atoms can lose or gain electrons during chemical reactions, forming ions. Cations are positively charged (loss of electrons), and anions are negatively charged (gain of electrons).
Cation: Fewer electrons than protons.
Anion: More electrons than protons.
Example: (Li loses one electron)
Example: (F gains one electron)
Calculating Charge
The charge of an ion is determined by the difference between the number of protons and electrons.
Formula:
Example: Magnesium with 12 protons and 10 electrons:
Example: Sulfur with 16 protons and 18 electrons:
Practice Examples
Argon (Ar): Z = 18, 18 protons, 18 electrons (neutral atom).
Phosphorus (P3−): Z = 15, 15 protons, 18 electrons (anion).
Lead (Pb2+): Z = 82, 82 protons, 80 electrons (cation).
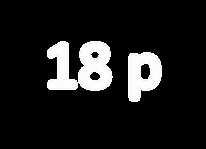
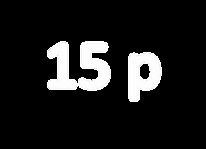

Summary Table: Subatomic Particles
Particle | Symbol | Charge | Mass (amu) |
|---|---|---|---|
Proton | p+ | +1 | 1.00727 |
Neutron | n | 0 | 1.00866 |
Electron | e− | −1 | 0.00055 |
Additional info: These notes cover the fundamental concepts of atomic structure, subatomic particles, the periodic table, and ion formation, as outlined in Chapter 4 "Atoms and Elements" of an introductory chemistry course.
