 Back
BackAtoms and Elements: Structured Study Notes for Introductory Chemistry
Study Guide - Smart Notes

Atoms and Elements
Overview
This chapter explores the fundamental concepts of atoms and elements, their structure, properties, and classification in the periodic table. Understanding these concepts is essential for grasping the nature of matter and its behavior in chemical reactions.
The Atomic Theory
Historical Development
The concept of the atom originated with ancient Greek philosophers, notably Democritus and Leucippus, who proposed that matter was composed of tiny, indivisible particles called "atomos." In 1808, John Dalton formalized the atomic theory, which became widely accepted.
Dalton's Atomic Theory:
Each element is composed of tiny, indestructible particles called atoms.
All atoms of a given element have the same mass and properties that distinguish them from atoms of other elements.
Atoms combine in simple, whole-number ratios to form compounds.
The Nuclear Atom
Discovery of Subatomic Particles
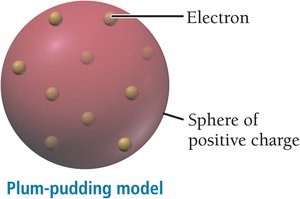
J. J. Thomson discovered the electron, a negatively charged particle much smaller than the atom. He proposed the "plum-pudding model," where electrons are embedded in a sphere of positive charge.
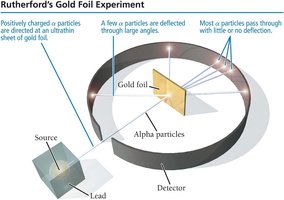
Rutherford's Gold Foil Experiment (1909) tested Thomson's model by directing alpha particles at a thin sheet of gold foil. Most particles passed through, but some were deflected, indicating a dense, positively charged nucleus.
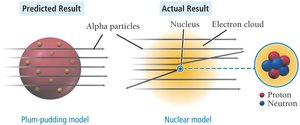
The experiment disproved the plum-pudding model and led to the nuclear model of the atom, where most of the atom's mass and positive charge are concentrated in the nucleus, and electrons occupy the surrounding space.
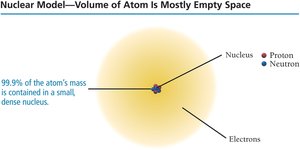
Protons, Neutrons, and Electrons
Subatomic Particles
Atoms are composed of three main subatomic particles:
Protons: Positively charged particles found in the nucleus.
Neutrons: Electrically neutral particles found in the nucleus.
Electrons: Negatively charged particles located outside the nucleus.
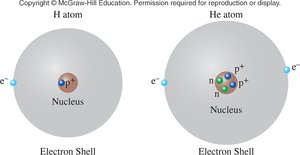
Electrical Charge
Electrical charge is a fundamental property of protons and electrons. Most matter is charge-neutral because protons and electrons occur together and their charges cancel.
Positive and negative charges attract each other.
Like charges (positive-positive or negative-negative) repel each other.
When paired, a proton and an electron are charge-neutral.

Elements and the Periodic Table
Atomic Number and Chemical Symbols
The atomic number (Z) is the number of protons in the nucleus and determines the identity of the atom. Each element is represented by a unique chemical symbol, often derived from its English, Latin, or Greek name.
Examples: O = oxygen, Pb = lead (plumbum), Hg = mercury (hydrargyrum), K = potassium (kalium).
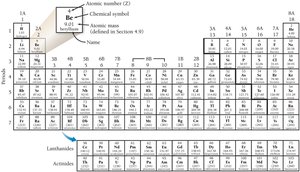
The Periodic Law and Classification
Dmitri Mendeleev organized elements by increasing mass, revealing recurring patterns in properties. The modern periodic table arranges elements by atomic number, grouping them by similar properties.
Metals: Usually solid, shiny, malleable, and conductive (e.g., Fe, Mg, Cr).
Nonmetals: Can be gas, solid, or liquid; poor conductors (e.g., O, N, Cl, I).
Metalloids: Semiconductors with mixed properties (e.g., Si, As).
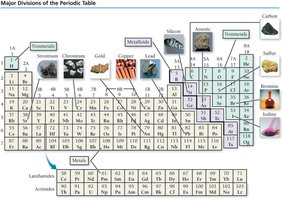
Periods and Groups
The periodic table is organized into periods (horizontal rows) and groups (vertical columns). Elements in the same group have similar properties.
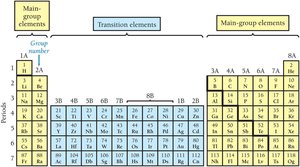
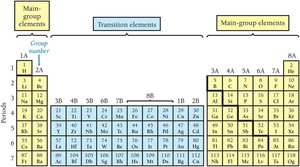
Main Group and Transition Elements
Main group elements (labeled with A) have predictable properties based on their position. Transition elements (labeled with B) have less predictable properties.
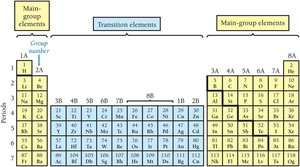
Special Groups
Alkali Metals (Group 1A): Highly reactive metals (e.g., Li, Na, K).
Alkaline Earth Metals (Group 2A): Fairly reactive metals (e.g., Be, Mg, Ca).
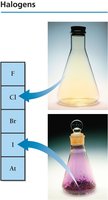
Halogens (Group 7A): Very reactive nonmetals (e.g., F, Cl, Br, I, At).
Noble Gases (Group 8A): Chemically inert gases (e.g., He, Ne, Ar, Kr, Xe).
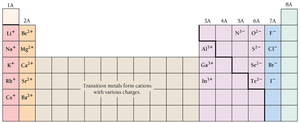
Ions: Gaining and Losing Electrons
Formation of Ions
Atoms can gain or lose electrons during chemical reactions, forming charged particles called ions.
Cations: Positive ions formed by losing electrons (e.g., Na+, Ca2+).
Anions: Negative ions formed by gaining electrons (e.g., Cl-, O2-).
Ion charge:
Isotopes
Definition and Properties
Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons. Each element has a unique percent natural abundance of isotopes.
Example: Neon has three isotopes—Ne-20, Ne-21, Ne-22—all with 10 protons but different numbers of neutrons.
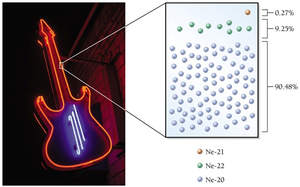
Isotope Notation
Symbol notation: (e.g., Ne-20).
Mass number (A):
Atomic Mass
Calculating Atomic Mass
The atomic mass listed in the periodic table is the weighted average mass of the atoms that compose the element. For elements with isotopes, atomic mass is calculated as:
Fractional abundance = percentage natural abundance / 100
Example: Chlorine has two isotopes: 75.77% chlorine-35 (mass 34.97 amu) and 24.23% chlorine-37 (mass 36.97 amu). The average atomic mass is:
Additional info: This calculation method applies to all elements with naturally occurring isotopes.
