 Back
BackAtoms, Elements, and the Foundations of Modern Atomic Theory
Study Guide - Smart Notes

Early Ideas in Atomic Theory
Historical Development of Atomic Theory
The concept of the atom originated with ancient Greek philosophers, who proposed that matter is composed of indivisible particles called atomos. However, Aristotle and others believed matter was made of four elements: fire, earth, air, and water. In 1807, John Dalton formulated the first modern atomic theory, which provided a scientific explanation for the nature of matter.
Dalton’s Atomic Theory Postulates:
Matter is composed of extremely small particles called atoms.
An element consists of only one type of atom, with a characteristic mass.
Atoms of one element differ from those of other elements.
Compounds consist of atoms of two or more elements in small, whole-number ratios.
Atoms are neither created nor destroyed in chemical changes; they are rearranged.
Example: A pre-1982 copper penny contains approximately copper atoms, each identical in chemical properties.

Law of Conservation of Matter
Dalton’s atomic theory explains the law of conservation of matter: atoms are not created or destroyed in chemical reactions, so the total mass remains constant.

Law of Definite and Multiple Proportions
Law of Definite Proportions: All samples of a pure compound contain the same elements in the same proportion by mass.
Law of Multiple Proportions: When two elements form more than one compound, the masses of one element that combine with a fixed mass of the other are in small, whole-number ratios.
Example: Copper(II) oxide (CuO) always contains copper and oxygen in a 1:1 ratio by atoms.

Example: Compounds of copper and chlorine can have different ratios, such as 1:1 or 1:2 chlorine to copper.
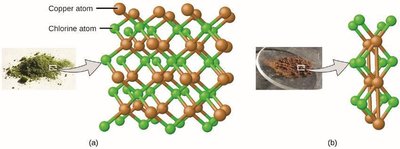
Evolution of Atomic Theory
Discovery of Subatomic Particles
Modern atomic theory developed through experiments that revealed atoms are composed of smaller particles: electrons, protons, and neutrons.
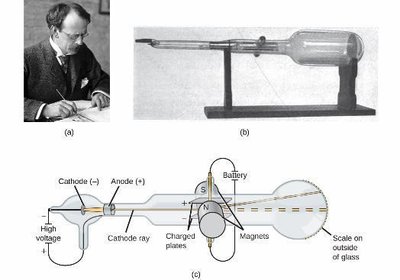
Discovery of the Electron (J.J. Thomson)
Thomson used cathode ray tubes to discover electrons, negatively charged particles much lighter than atoms.
He measured the charge-to-mass ratio of electrons, showing they are universal components of all atoms.
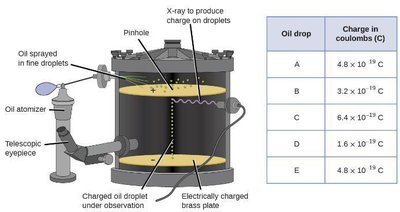
Millikan’s Oil Drop Experiment
Robert Millikan measured the charge of the electron ( C) by observing charged oil droplets in an electric field.
Combined with Thomson’s results, this allowed calculation of the electron’s mass ( kg).
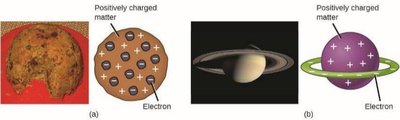
Models of the Atom
Thomson proposed the "plum pudding" model: electrons embedded in a positively charged sphere.
Nagaoka suggested a "Saturnian" model: electrons orbiting a positive center.
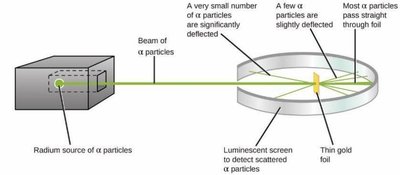
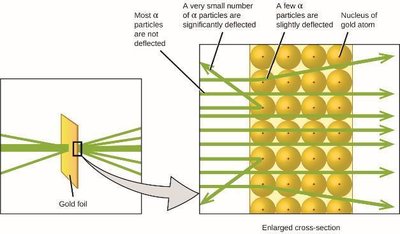
Discovery of the Nucleus (Ernest Rutherford)
Rutherford’s gold foil experiment showed that atoms have a small, dense, positively charged nucleus.
Most alpha particles passed through the foil, but some were deflected, indicating a concentrated nucleus.
Other Discoveries
Isotopes: Atoms of the same element with different numbers of neutrons (Frederick Soddy).
Neutrons: Uncharged particles in the nucleus, similar in mass to protons (James Chadwick).
Atomic Structure and Symbolism
Structure of the Atom
The atom consists of a tiny nucleus (containing protons and neutrons) surrounded by electrons. The nucleus is much smaller than the atom as a whole.

Proton: Mass = 1.0073 amu, Charge = +1
Neutron: Mass = 1.0087 amu, Charge = 0
Electron: Mass = 0.00055 amu, Charge = –1
Atomic Number, Mass Number, and Isotopes
Atomic Number (Z): Number of protons; defines the element.
Mass Number (A): Total number of protons and neutrons.
Isotopes: Atoms of the same element with different numbers of neutrons.
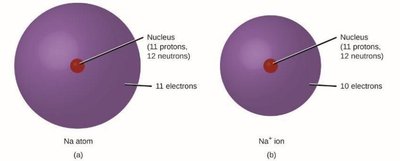
Ions: Atoms with unequal numbers of protons and electrons (cations are positive, anions are negative).
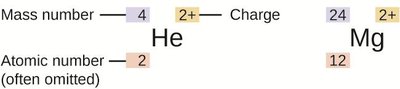
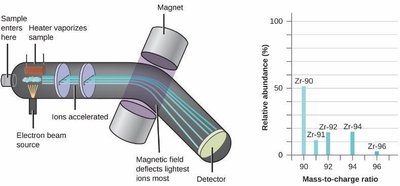
Atomic Mass and Mass Spectrometry
Atomic Mass Unit (amu):
Average Atomic Mass: Weighted average of all isotopes’ masses.
Mass Spectrometry: Technique to determine isotopic composition and atomic masses.
Chemical Formulas
Molecular and Empirical Formulas
Molecular Formula: Shows the actual number of each type of atom in a molecule (e.g., CH4).
Empirical Formula: Shows the simplest whole-number ratio of atoms (e.g., CH for benzene).
Structural Formula: Shows how atoms are connected.
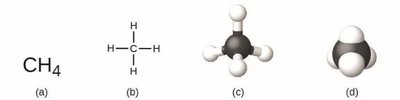
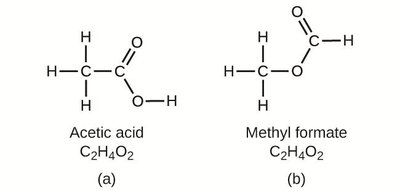
Isomers: Compounds with the same molecular formula but different structures (e.g., acetic acid and methyl formate).
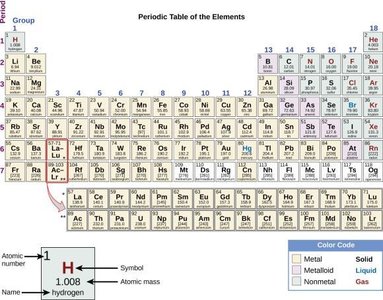
The Periodic Table
Organization and Classification
The periodic table arranges elements by increasing atomic number and groups elements with similar properties in columns. The periodic law states that element properties are periodic functions of their atomic numbers.

Metals: Shiny, malleable, good conductors.
Nonmetals: Dull, poor conductors.
Metalloids: Intermediate properties.
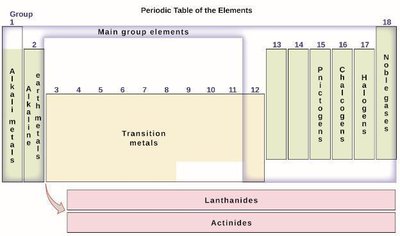
Main group elements: Groups 1, 2, 13–18.
Transition metals: Groups 3–12.
Inner transition metals: Lanthanides and actinides.
Molecular and Ionic Compounds
Types of Compounds
Molecular (Covalent) Compounds: Atoms share electrons; usually nonmetals.
Ionic Compounds: Electrons are transferred from metals to nonmetals, forming cations and anions held by electrostatic forces.
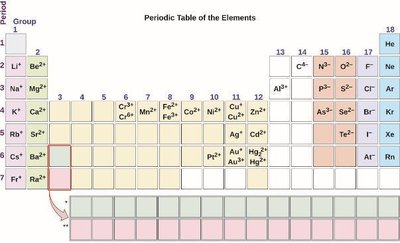
Predicting Ion Charges
Main-group metals form cations with charges equal to their group number.
Main-group nonmetals form anions with charges equal to the group number minus eight.
Transition metals can form multiple cations with different charges.
Polyatomic Ions
Polyatomic ions: Groups of atoms with an overall charge (e.g., SO42–, NH4+).
Oxyanions: Polyatomic ions containing oxygen; named with -ate or -ite suffixes, and sometimes with per- or hypo- prefixes.
Properties of Ionic Compounds
High melting and boiling points, solid at room temperature.
Conduct electricity when molten or dissolved in water.

Chemical Nomenclature
Naming Ionic Compounds
Name the cation first, then the anion.
Monatomic anions use the -ide suffix (e.g., chloride, oxide).
Polyatomic ions retain their specific names (e.g., sulfate, nitrate).
Transition metals with variable charge use Roman numerals (e.g., iron(III) chloride).
Naming Molecular Compounds
Use prefixes to indicate the number of each atom (e.g., CO2: carbon dioxide).
The more metallic element is named first; the second element uses the -ide suffix.
Naming Acids
Binary acids: Use the prefix hydro-, the root of the nonmetal, and the suffix -ic, followed by "acid" (e.g., HCl(aq): hydrochloric acid).
Oxyacids: If the anion ends in -ate, the acid name ends in -ic; if the anion ends in -ite, the acid name ends in -ous (e.g., H2SO4: sulfuric acid; H2SO3: sulfurous acid).
