 Back
BackAtoms, Elements, and the Structure of Matter: Foundations of Atomic Theory
Study Guide - Smart Notes

Dalton’s Model of the Atom
Dalton’s Atomic Theory
Dalton’s atomic theory, proposed in the early 19th century, laid the groundwork for our modern understanding of matter. It described the nature of atoms and how they combine to form compounds.
Elements are composed of extremely small particles called atoms.
Atoms of a given element are identical in mass and size.
Atoms of different elements have different masses and sizes.
Atoms of different elements combine in simple, whole-number ratios to form compounds.
Atoms of two elements may combine in different ratios to form different compounds.

Additional info: Dalton’s model did not account for subatomic particles or isotopes, which were discovered later.
Development of Atomic Models
Thomson’s Plum Pudding Model
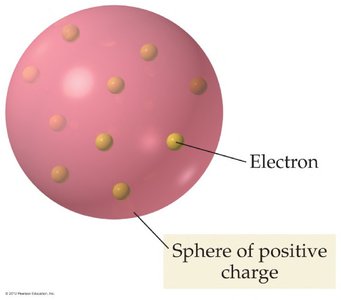
J.J. Thomson proposed the "plum pudding" model after discovering the electron. In this model, the atom is a sphere of positive charge with negatively charged electrons embedded throughout, like plums in a pudding.
Electrons: Negatively charged particles distributed within a positively charged sphere.
Positive charge: The sphere itself carries a uniform positive charge to balance the electrons.
Rutherford’s Nuclear Model
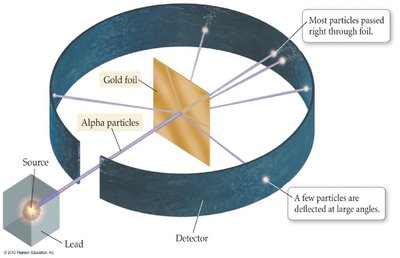
Ernest Rutherford’s gold foil experiment (1909) revolutionized atomic theory. He directed alpha particles at a thin sheet of gold foil and observed their scattering patterns.
Most alpha particles passed straight through the foil, indicating that atoms are mostly empty space.
A few particles were deflected at sharp angles, suggesting the presence of a dense, positively charged nucleus at the center of the atom.

Comparison: Plum Pudding vs. Nuclear Model
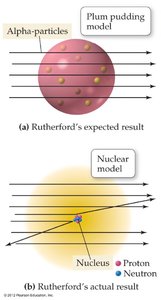
Rutherford’s results contradicted the plum pudding model. Instead of passing through uniformly, some alpha particles were deflected, supporting the nuclear model with a central nucleus.
Plum pudding model: Expected all alpha particles to pass through with minimal deflection.
Nuclear model: Explained large deflections by the presence of a small, dense nucleus.
Electrical Charge and Subatomic Particles
Nature of Electrical Charge
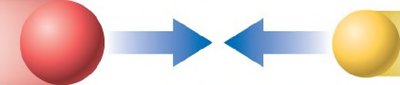
Electrical charge is a fundamental property of matter, especially protons and electrons. The interactions between charges govern much of chemistry.
Opposite charges attract: Positive and negative charges pull toward each other.
Like charges repel: Two positive or two negative charges push away from each other.
Charge neutrality: Atoms are neutral when they have equal numbers of protons and electrons.



The Structure of the Atom
Subatomic Particles
Atoms are divisible and composed of three main subatomic particles: protons, neutrons, and electrons. The nuclear model describes the atom as mostly empty space, with a dense nucleus containing protons and neutrons, and electrons occupying the surrounding space.
Proton: Positively charged particle found in the nucleus.
Neutron: Neutral particle found in the nucleus.
Electron: Negatively charged particle found outside the nucleus.

Atomic and Nuclear Size
The atom is extremely small, with the nucleus being much smaller than the atom itself.
Atomic diameter: Approximately meters.
Nuclear diameter: Approximately meters, about 10,000 times smaller than the atom.

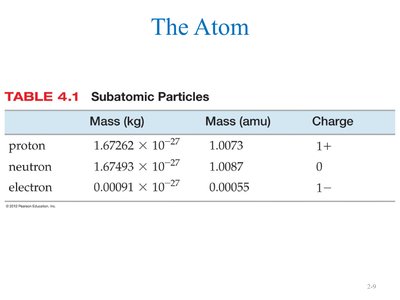
Properties of Subatomic Particles
The three subatomic particles differ in mass and charge. Their properties are summarized in the table below:
Particle | Mass (kg) | Mass (amu) | Charge |
|---|---|---|---|
Proton | 1.67262 × 10-27 | 1.0073 | 1+ |
Neutron | 1.67493 × 10-27 | 1.0087 | 0 |
Electron | 0.00091 × 10-27 | 0.00055 | 1– |
Atomic Number and the Periodic Table
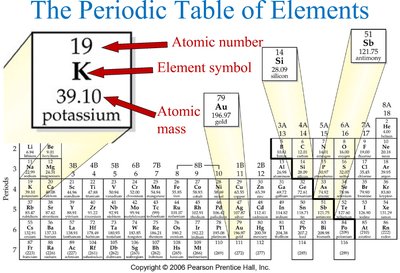
Atomic Number
The atomic number (Z) is the number of protons in the nucleus of an atom and uniquely identifies an element. All atoms of a given element have the same atomic number.
Atomic number (Z): Number of protons in the nucleus.
Element identity: Determined by the atomic number.
Periodic table: Elements are arranged in order of increasing atomic number.

Element Names and Symbols
Each element is designated by a unique name and a one- or two-letter symbol. The first letter is always capitalized, and the second letter (if present) is lowercase.
Examples: H for hydrogen, He for helium, Na for sodium.
Origins of Element Names
Element names are derived from various sources, including Latin and Greek words, mythological characters, and names of people or places.
Examples: Californium (Cf), Berkelium (Bk), Americium (Am), Europium (Eu), Einsteinium (Es), Nobelium (No).

The Periodic Table
The periodic table organizes elements by increasing atomic number and groups elements with similar chemical properties into columns.
Groups (columns): Elements with similar properties.
Periods (rows): Elements with increasing atomic number.

Atomic Number and Mass Number
Definitions and Notation
Atoms are characterized by their atomic number (Z), mass number (A), and number of neutrons (N). These quantities are related by the following equations:
Atomic number (Z): Number of protons.
Mass number (A): Total number of protons and neutrons.
Number of neutrons (N):
Example Calculation
For the isotope 2914Si (silicon-29):
Atomic number (Z): 14 (protons)
Mass number (A): 29 (protons + neutrons)
Number of neutrons (N):

Isotopes
Definition and Examples
Isotopes are atoms of the same element (same number of protons) that have different numbers of neutrons, and therefore different mass numbers.
Isotopes of hydrogen: Protium (1H), Deuterium (2H), Tritium (3H).
Notation: Name of element followed by mass number (e.g., carbon-14, cobalt-60).

Sample Calculations and Exercises
To determine the number of protons and neutrons in an isotope:
Example: Lead-206 (206Pb): Atomic number = 82, so protons = 82, neutrons = .

Practice Problems
Given the isotope 15N, protons = 7, neutrons = .
Given 61Ni, neutrons = .
238U is called uranium-238.

Tabular Practice
Fill in missing information for neutral atoms:
Symbol | Protons | Neutrons | Electrons | Mass no. |
|---|---|---|---|---|
52Cr | 24 | 28 | 24 | 52 |
25 | 30 | 25 | 55 | |
82 | 125 | 82 | 207 |

