 Back
BackExam 2
Study Guide - Smart Notes

Atoms and Elements
Definition of Atoms and Elements
The study of chemistry begins with understanding the basic building blocks of matter: atoms and elements. An atom is the smallest identifiable unit of an element, while an element is a substance that cannot be broken down into simpler substances by chemical means. There are 91 naturally occurring elements and about 20 synthetic elements, each with unique properties.
Atom: Smallest unit of an element, retains chemical properties.
Element: Pure substance made of only one kind of atom.
Each element is represented by a unique chemical symbol.
Atomic Theory and Structure
The concept of the atom originated with ancient philosophers but was formalized by John Dalton. Dalton's Atomic Theory states:
Each element is composed of tiny, indestructible particles called atoms.
All atoms of a given element have the same mass and properties.
Atoms combine in simple, whole-number ratios to form compounds.
Discovery of Subatomic Particles
J.J. Thomson discovered the electron, a negatively charged particle much lighter than atoms. Rutherford's gold foil experiment led to the discovery of the nucleus, a dense core containing protons and neutrons. The Nuclear Theory of the Atom states:
Most of the atom’s mass and all positive charge are in the nucleus.
Electrons occupy most of the atom’s volume but contribute little to its mass.
Atoms are electrically neutral: number of protons equals number of electrons.
Protons, Neutrons, and Electrons
Atoms are composed of three main subatomic particles:
Protons: Positively charged, found in the nucleus.
Neutrons: Neutral, found in the nucleus.
Electrons: Negatively charged, found outside the nucleus.
The atomic number (Z) is the number of protons in the nucleus and defines the element. The mass number (A) is the sum of protons and neutrons.
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons. They have the same atomic number but different mass numbers. The percent natural abundance describes the relative amount of each isotope in a natural sample.
The Periodic Table
Organization and Periodic Law
The Periodic Table, developed by Dmitri Mendeleev, organizes elements by increasing atomic number. The Periodic Law states that properties of elements recur periodically when arranged by atomic mass. Elements are grouped into families with similar properties.
Groups (columns): Elements with similar chemical properties.
Periods (rows): Elements with increasing atomic number.
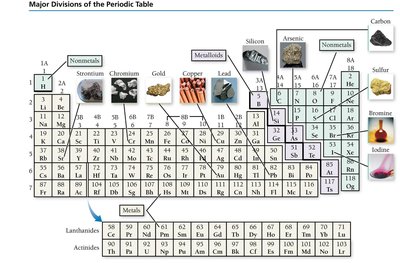
Major Divisions of the Periodic Table
Metals: Left side; good conductors, malleable, ductile, shiny, lose electrons in reactions.
Nonmetals: Upper right; poor conductors, gain electrons in reactions.
Metalloids: Along the diagonal; semiconductors, intermediate properties.
Main-group elements: Groups 1A–8A; predictable properties.
Transition elements: Group B; less predictable properties.
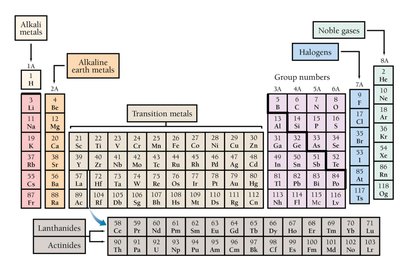
Special Groups
Alkali Metals (1A): Very reactive metals.
Alkaline Earth Metals (2A): Reactive metals.
Halogens (7A): Very reactive nonmetals.
Noble Gases (8A): Unreactive, do not form compounds easily.
Ions and Isotopes
Formation of Ions
An ion is an atom or group of atoms that has gained or lost electrons, resulting in a net charge. The ion charge is calculated as:
Cations: Positively charged ions (loss of electrons).
Anions: Negatively charged ions (gain of electrons).
Valence electrons are the outermost electrons and determine chemical reactivity. Main-group elements tend to form ions with the same number of valence electrons as the nearest noble gas.
Atomic Mass and Isotopic Abundance
Calculating Atomic Mass
The atomic mass of an element is the weighted average of the masses of its isotopes, calculated as:
Percent natural abundance is used to determine the contribution of each isotope to the average atomic mass.
Molecules and Compounds
Chemical Formulas and Models
A chemical formula indicates the elements present in a compound and the relative number of atoms of each element. Types of formulas include:
Empirical formula: Simplest whole-number ratio of atoms.
Molecular formula: Actual number of atoms in a molecule.
Structural formula: Shows how atoms are connected.
Molecular models help visualize the structure of molecules:
Ball-and-stick models: Atoms as balls, bonds as sticks.
Space-filling models: Atoms fill the space between each other.
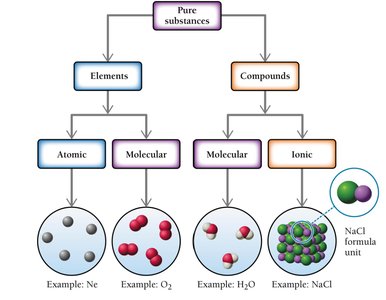
Classification of Pure Substances
Pure substances can be classified as elements or compounds. Elements can be atomic or molecular, while compounds can be molecular or ionic.
Atomic elements: Exist as single atoms (e.g., Ne).
Molecular elements: Exist as molecules with two or more atoms (e.g., O2).
Molecular compounds: Composed of nonmetals (e.g., H2O).
Ionic compounds: Composed of cations (usually metals) and anions (usually nonmetals), basic unit is the formula unit (e.g., NaCl).
Naming Compounds (Nomenclature)
Naming Ionic Compounds
Ionic compounds are formed from metals and nonmetals. The sum of the charges of the cations and anions must be zero. There are two main types:
Metal forms one type of ion only: name of cation + base name of anion + -ide.
Metal forms more than one type of ion: name of cation (charge in Roman numerals) + base name of anion + -ide.
Naming Molecular Compounds
Molecular compounds are composed of nonmetals only. Prefixes are used to indicate the number of each atom:
Prefix + name of 1st element + prefix + base name of 2nd element + -ide.
Naming Acids
Acids are molecular compounds that produce H+ ions in water. They are named based on their composition:
Binary acids: "Hydro-" + base name of nonmetal + "-ic acid" (e.g., HCl = hydrochloric acid).
Oxyacids: Contain oxygen; if the polyatomic ion ends in "-ate," use "-ic acid"; if "-ite," use "-ous acid."
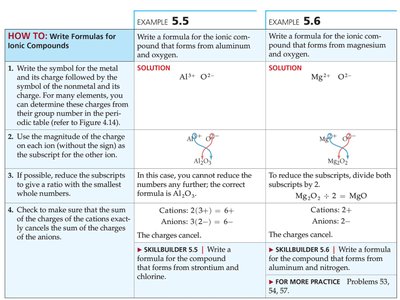
Writing Formulas for Ionic Compounds
To write formulas for ionic compounds:
Write the symbol for the metal and its charge, followed by the symbol for the nonmetal and its charge.
Use the magnitude of the charges as subscripts for the other ion.
Reduce subscripts to the smallest whole-number ratio.
Ensure the sum of the charges equals zero.

Chemical Composition and the Mole Concept
Counting Atoms by Weighing
The mole is a counting unit in chemistry. One mole contains particles (Avogadro's number). The molar mass is the mass of one mole of a substance, numerically equal to its atomic or molecular mass in grams.
To convert between grams, moles, and number of atoms or molecules, use the following steps:
Grams → Moles → Number of particles (atoms/molecules)
For compounds, the molar mass is the sum of the atomic masses of all atoms in the formula.
Conversion Factors
Conversion factors are ratios used to convert between units. For example, to find the number of atoms in a given mass of a compound:
Convert mass of compound to moles of compound (using molar mass).
Convert moles of compound to moles of element (using subscripts in formula).
Convert moles of element to mass of element (using atomic mass).
Example: To find the number of O atoms in 22.5 g of CO2:
Find moles of CO2 using molar mass.
Multiply by 2 (since each CO2 has 2 O atoms).
Multiply by Avogadro's number to get number of O atoms.
