Back
BackChapter 1: Science & Measurements – Foundations of Chemistry
Study Guide - Smart Notes

Science & Measurements
Introduction to Chemistry
Chemistry is the scientific study of matter, its properties, and the changes it undergoes. Understanding chemistry involves exploring both the physical and chemical properties of substances, as well as the energy changes associated with these processes.
Matter and Its Changes
Physical and Chemical Properties
Properties of matter are classified as either physical or chemical:
Physical properties: Characteristics that can be observed or measured without changing the substance's identity (e.g., state, shape, melting point, boiling point, color, odor).
Chemical properties: Characteristics that describe a substance's ability to undergo chemical changes (e.g., reactivity, flammability, oxidation, radioactivity).
Physical and Chemical Changes
Changes in matter are categorized as:
Physical changes: Alterations that do not affect the chemical identity of a substance (e.g., melting ice, dissolving salt in water, crushing a can). These changes are often reversible and involve changes in physical state or appearance.
Chemical changes: Transformations that result in the formation of new substances with different chemical identities (e.g., burning, rusting, digestion). These changes are usually permanent and involve breaking and forming chemical bonds.

Example: Melting ice is a physical change, while burning a candle is a chemical change.

Matter and Energy
Forms of Energy
Energy is essential in chemical processes and exists in two main forms:
Kinetic energy: The energy of motion.
Potential energy: Stored energy due to position or chemical bonds.
The Law of Conservation of Energy states that energy cannot be created or destroyed, only transformed or transferred.
The Scientific Method
Steps of the Scientific Method
The scientific method is a systematic approach to studying matter and energy. It involves:
Making observations
Formulating a hypothesis
Conducting experiments
Interpreting findings
Drawing conclusions and developing theories or laws
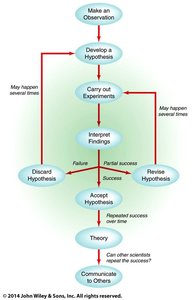
Theory vs. Law: A theory explains observed phenomena, while a law describes consistent observations without explaining them.
Measurements in Chemistry
Types of Numbers in Measurement
Measurements consist of a number and a unit. There are two types of numbers:
Exact numbers: Obtained by counting or definition (e.g., 12 eggs in a dozen).
Measured numbers: Obtained using measuring tools and always have some uncertainty.


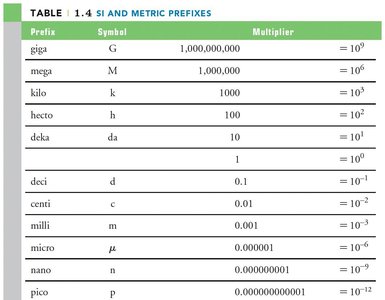
Scientific Notation and SI Prefixes
Scientific notation expresses very large or small numbers as a product of a number between 1 and 10 and a power of 10. SI prefixes are used to simplify these expressions.
Prefix | Symbol | Multiplier |
|---|---|---|
giga | G | 109 |
mega | M | 106 |
kilo | k | 103 |
centi | c | 10-2 |
milli | m | 10-3 |
micro | μ | 10-6 |
nano | n | 10-9 |
Measurement Units
There are several systems of units, but the SI (International System) is standard in science. Key base units include:
Quantity | English Unit | Metric Unit | SI Unit | Relationships |
|---|---|---|---|---|
Mass | Pound (lb) | Gram (g) | Kilogram (kg) | 1 kg = 2.205 lb; 1 kg = 1000 g |
Length | Foot (ft) | Meter (m) | Meter (m) | 1 m = 3.281 ft |
Volume | Quart (qt) | Liter (L) | Cubic meter (m3) | 0.946 L = 1 qt; 1 m3 = 1000 L |
Energy | calorie (cal) | calorie (cal) | Joule (J) | 4.184 J = 1 cal |
Temperature | Degree Fahrenheit (°F) | Degree Celsius (°C) | Kelvin (K) | °F = (1.8 × °C) + 32; K = °C + 273.15 |
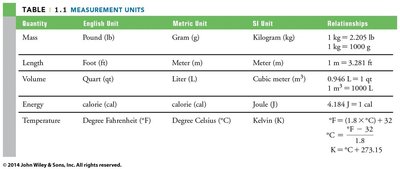
Quality of Measurements
Accuracy, Precision, and Significant Figures
Measurement quality is determined by:
Accuracy: How close a measurement is to the true value.
Precision: How close repeated measurements are to each other.
Significant figures: The digits in a measurement that are known with certainty plus one estimated digit.
Formula for percent error:
Reading Measurements and Significant Figures
When using measuring devices:
For digital devices, record all displayed digits.
For analog devices, record all known digits plus one estimated digit.
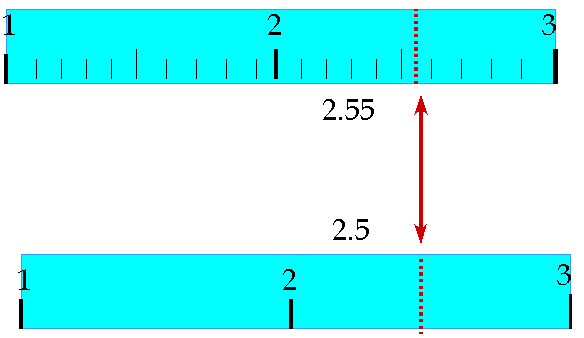
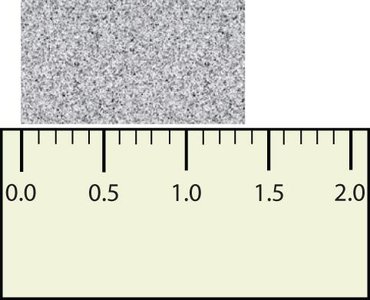


Rules for Significant Figures
Rules for determining significant figures:
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Leading zeros are not significant.
Trailing zeros are significant only if there is a decimal point.

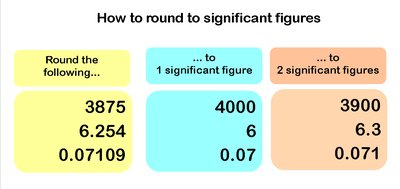
Rounding and Calculations with Significant Figures
When multiplying or dividing, the answer should have the same number of significant figures as the value with the fewest significant figures.
When adding or subtracting, the answer should have the same number of decimal places as the value with the fewest decimal places.

Conversion Factors and Dimensional Analysis
Using Conversion Factors
The factor label method (dimensional analysis) uses conversion factors to convert between units. A conversion factor is a ratio that expresses how many of one unit are equal to another unit.
Example: To convert 2 dozen eggs to the number of eggs:
Complex and Derived Units
Density:
Specific heat:
Specific gravity:

Applications in Medicine and Everyday Life
Dimensional analysis is crucial in fields such as medicine for calculating dosages and converting between units.

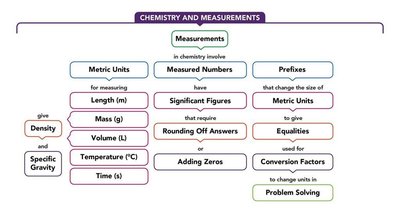
Concept Map
The following concept map summarizes the relationships between measurements, units, significant figures, and problem-solving in chemistry: