 Back
BackChapter 1: The Chemical World – Foundations of Chemistry
Study Guide - Smart Notes

Chemistry & Matter
Definition and Scope of Chemistry
Chemistry is the scientific study of matter, its properties, composition, and the changes it undergoes. Matter is defined as anything that occupies space and has mass. Understanding chemistry requires examining the particles that compose matter and how these particles interact and behave.
Chemistry: The study of the composition, structure, properties, and changes of matter.
Matter: Anything that has mass and occupies space.
To understand matter, we must understand the particles (atoms, molecules, ions) that compose it.
Examples of matter include solids, liquids, gases, and plasma.

Classification of Matter
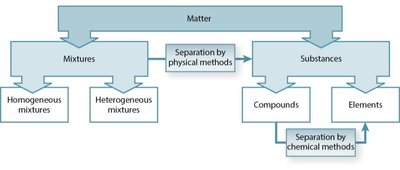
Matter can be classified based on its physical and chemical properties. The main categories are mixtures and pure substances, which can be further subdivided based on their characteristics and methods of separation.
Mixtures: Physical combinations of two or more substances. Can be separated by physical methods.
Homogeneous mixtures: Uniform composition throughout (e.g., saltwater).
Heterogeneous mixtures: Non-uniform composition (e.g., salad, sand in water).
Substances: Matter with a fixed composition; can be elements or compounds.
Elements: Pure substances that cannot be broken down by chemical means (e.g., oxygen, gold).
Compounds: Substances composed of two or more elements chemically combined (e.g., water, carbon dioxide).
Separation Methods:
Mixtures can be separated by physical methods (e.g., filtration, distillation).
Compounds can be separated into elements by chemical methods (e.g., electrolysis).
The Scientific Method
Overview of the Scientific Method
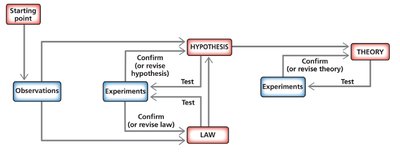
The scientific method is a systematic, repetitive process used to learn about the natural world through observation and experimentation. It is the foundation of scientific inquiry and leads to the development of scientific laws and theories.
Observation: Gathering information about phenomena.
Hypothesis: A tentative explanation or prediction that can be tested by experiments.
Experiment: A controlled procedure to test the hypothesis.
Law: A statement that summarizes past observations and predicts future ones (e.g., Law of Conservation of Mass).
Theory (Model): An explanation for the observed behavior of matter, supported by extensive evidence (e.g., Atomic Theory).
Scientific Laws vs. Theories
Scientific laws and theories are both important outcomes of the scientific method, but they serve different purposes:
Scientific Law: Describes what happens; summarizes observations (e.g., Newton's Laws of Motion).
Theory: Explains why something happens; provides a model for understanding observations (e.g., Kinetic Molecular Theory).
Both laws and theories are subject to revision based on new evidence. As famously stated by Albert Einstein, "Many experiments may prove me right, but it takes only one to prove me wrong." This highlights the tentative nature of scientific knowledge.

Analyzing & Interpreting Data
Patterns and Graphing
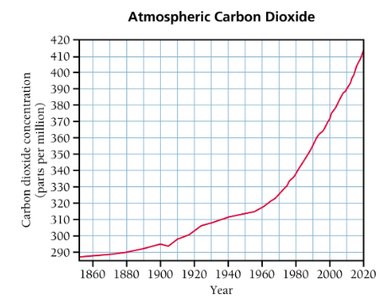
Data obtained from experiments may not always provide an obvious answer. Scientists look for patterns in the data, and graphing is a useful tool for visualizing trends and relationships.
Graphical representation helps in identifying trends, correlations, and anomalies.
Example: Graphing atmospheric carbon dioxide concentration over time reveals trends in climate data.
Tabular Data and Ratios
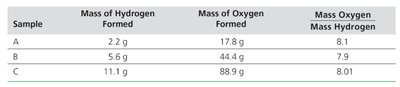
Tables are used to organize experimental data for analysis. Calculating ratios, such as the mass of oxygen to the mass of hydrogen formed in a reaction, helps in identifying consistent relationships and supports the development of laws (e.g., Law of Definite Proportions).
Sample | Mass of Hydrogen Formed | Mass of Oxygen Formed | Mass Oxygen / Mass Hydrogen |
|---|---|---|---|
A | 2.2 g | 17.8 g | 8.1 |
B | 5.6 g | 44.4 g | 7.9 |
C | 11.1 g | 88.9 g | 8.01 |
Chapter 1 Study Guide
Key Terms and Concepts
Be familiar with the definitions of chemistry, matter, hypothesis, scientific law, and theory.
Understand the basics of the scientific method: making observations, formulating hypotheses, performing experiments, analyzing results, and drawing conclusions.
Recognize that the scientific method is a repetitive process used to learn through observation and experimentation, leading to the development of theories and laws.
