 Back
BackChapter 1: The Chemical World – Foundations of Chemistry
Study Guide - Smart Notes

The Chemical World
Introduction to Chemistry
Chemistry is the science that seeks to understand what matter does by studying the behavior of atoms and molecules. Everything in the physical world is composed of chemicals, and chemistry provides the framework for understanding their properties and interactions.

Chemicals are substances that make up everything we see, touch, and use in daily life.
Common examples include water, air, toothpaste, and even the materials in a pen.
Atoms and Molecules in Matter
Basic Building Blocks
All matter is composed of extremely small particles called atoms and molecules. Atoms are the fundamental units, and molecules are groups of atoms bonded together by chemical bonds.
Atoms: The smallest units of elements, retaining the properties of the element.
Molecules: Combinations of two or more atoms held together by chemical bonds.
Chemical bonds: The forces that hold atoms together in molecules.
Water Molecules
A water molecule consists of three atoms—one oxygen and two hydrogen—bonded together in a bent shape. This structure gives water its unique properties, such as being a liquid at room temperature.
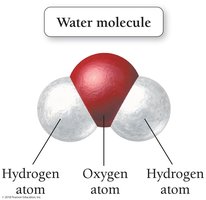
Formula:
The bent shape is crucial for water's physical and chemical properties.
Chemicals Compose Ordinary Things
Chemicals in Everyday Life
Everything around us is made of chemicals, not just dangerous substances or pollutants. Even the air we breathe and the water we drink are chemicals. Chemistry helps us understand the properties and behaviors of these substances by examining the molecules that compose them.

Common misconceptions often limit the definition of chemicals to harmful substances, but chemicals are present in all materials.
Examples: Paint thinner, polluted water, and clean drinking water are all composed of chemicals.



All Things Are Made of Atoms and Molecules
Fundamental Concept
The most important idea in chemistry is that all things are made of atoms. This concept, emphasized by physicist Richard Feynman, forms the foundation of modern science.

The Scientific Method: How Chemists Think
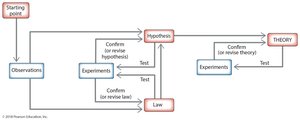
Overview of the Scientific Method
The scientific method is a systematic approach used by chemists to understand the world. It emphasizes observation and experimentation to produce reliable knowledge.
Observation: Gathering data through the senses or instruments.
Hypothesis: A tentative explanation for observations, which must be testable and falsifiable.
Law: A statement that summarizes a large number of observations and predicts future outcomes.
Theory: A well-established model that explains and unifies laws and observations.
Contrast with Ancient Philosophy
The scientific method differs from ancient and medieval approaches, which relied on reasoning and thought rather than empirical evidence.
Experiments and Validation
Hypotheses, laws, and theories must be tested and validated by experiments. If experimental results contradict a hypothesis, it must be revised or replaced.
Observation Example: Combustion
Antoine Lavoisier, a French chemist, observed that the mass of a closed container and its contents does not change during combustion (burning). This led to the formulation of the law of conservation of mass.

Mass: A measure of the quantity of matter in an object.
Combustion: The process of burning a substance in the presence of oxygen.
Law of Conservation of Mass
Lavoisier's experiments led to the law of conservation of mass, which states:
"In a chemical reaction, matter is neither created nor destroyed."
Scientific Theories
One or more well-established hypotheses can form the basis for a scientific theory. Theories provide deep explanations and can predict new phenomena. Well-tested theories are the most reliable knowledge in science.
Scientific Method Flowchart
Example: Dalton's Atomic Theory
John Dalton proposed that all matter is composed of small, indestructible particles called atoms. This theory explained the law of conservation of mass and provided a model for understanding chemical reactions.

Dalton's atomic theory is a foundational scientific theory with strong experimental support.
Modern Evidence for Atomic Theory
Modern technology allows us to visualize individual atoms. For example, the Kanji characters for "atom" have been written using individual iron atoms on a copper surface.

Success as a Beginning Chemist
Skills and Attitudes for Success
To succeed in chemistry, students must be curious, imaginative, and committed. Quantification—making precise measurements—is essential for scientific observation and analysis.
Regular and careful work is necessary for mastering chemistry.
Quantification allows for precise comparison and understanding of chemical phenomena.
Review
Matter and Molecules: Chemistry seeks to understand matter by studying molecules.
The Scientific Method: Chemists use observations, hypotheses, laws, theories, and experiments to build scientific knowledge.
Analyzing Data: Scientific data, often collected as measurements, can be graphed to reveal relationships and trends.
