 Back
BackChapter 10: Chemical Bonding – Study Notes
Study Guide - Smart Notes

Chemical Bonding
Introduction to Chemical Bonding
Chemical bonding explains how atoms combine to form compounds and the resulting properties of these compounds. Understanding bonding is essential for predicting molecular structure, reactivity, and physical properties.
Bonding theories help predict which atoms will bond and the shapes of resulting molecules.
The Lewis model is a foundational bonding theory using electron dot structures to represent valence electrons.
Bonding is crucial in fields such as drug design, as seen in the development of HIV-protease inhibitors.

Lewis Model of Chemical Bonding
Valence Electrons and Lewis Structures
The Lewis model represents valence electrons as dots around the chemical symbol of an element. This visual approach helps predict bonding and molecular stability.
The number of valence electrons for main-group elements equals the group number (except helium).
Lewis structures (dot structures) show these electrons as dots placed singly before pairing.
Atoms with eight valence electrons (an octet) are particularly stable.

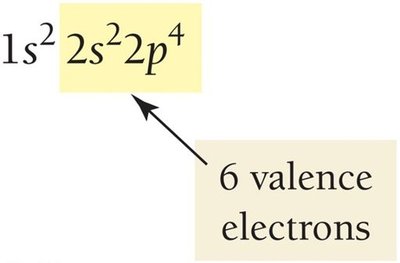

Exception: Helium is stable with two valence electrons (a duet).
Types of Chemical Bonds
Atoms bond to achieve stable electron configurations, usually an octet (eight electrons) in their valence shell.
Ionic bonds: Electrons are transferred from metals to nonmetals, forming cations and anions.
Covalent bonds: Electrons are shared between nonmetals.
Octet rule: Atoms tend to gain, lose, or share electrons to achieve eight valence electrons (except H and He, which achieve a duet).
Lewis Structures for Ionic Compounds
Formation of Ionic Compounds
When metals react with nonmetals, electrons are transferred, resulting in the formation of ions and ionic compounds.
Metals lose electrons to become cations.
Nonmetals gain electrons to become anions.
The resulting electrostatic attraction forms an ionic compound.

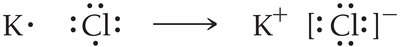
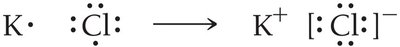
Lewis structures for ions are shown in brackets with the charge indicated.
Lewis Structures for Covalent Compounds
Formation of Covalent Bonds
Nonmetals bond with each other by sharing electrons, forming molecular compounds. The shared electrons allow each atom to achieve an octet (or duet for hydrogen).
Shared electrons are called bonding pairs.
Unshared electrons are called lone pairs or nonbonding pairs.




Bonding pairs are often represented by dashes (lines) between atoms.
Multiple Bonds: Double and Triple Bonds
Some molecules require double or triple bonds to satisfy the octet rule.
Double bond: Two pairs of electrons shared (e.g., O2).
Triple bond: Three pairs of electrons shared (e.g., N2).
Double and triple bonds are shorter and stronger than single bonds.

Steps for Writing Lewis Structures for Covalent Compounds
Follow these steps to construct Lewis structures:
Write the skeletal structure (arrangement of atoms).
Sum the total number of valence electrons (consider charges for polyatomic ions).
Distribute electrons to give octets (or duets for H) to as many atoms as possible.
If any atom lacks an octet, form double or triple bonds as needed.
Exceptions to the Octet Rule
Some molecules do not follow the octet rule:
Molecules with an odd number of electrons (free radicals).
Atoms like boron (B) and beryllium (Be) may have less than an octet.
Expanded octets (more than eight electrons) occur for some elements in period 3 or higher.
Resonance Structures
Some molecules can be represented by more than one valid Lewis structure. These are called resonance structures. The actual molecule is an average (hybrid) of these structures.
Resonance is indicated by double-headed arrows between structures.
Example: SO2 has two resonance structures with different placements of double bonds.
Predicting Molecular Shapes: VSEPR Theory
Valence Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory predicts the three-dimensional shapes of molecules based on the repulsion between electron groups (bonding and lone pairs) around the central atom.
Electron groups arrange themselves as far apart as possible.
The number of electron groups determines the electron geometry.
Lone pairs affect the molecular geometry (the arrangement of atoms).
Common Molecular Geometries
Electron Groups | Bonding Groups | Lone Pairs | Electron Geometry | Bond Angle | Molecular Geometry | Example |
|---|---|---|---|---|---|---|
2 | 2 | 0 | Linear | 180° | Linear | CO2 |
3 | 3 | 0 | Trigonal planar | 120° | Trigonal planar | CH2O |
3 | 2 | 1 | Trigonal planar | ~120° | Bent | SO2 |
4 | 4 | 0 | Tetrahedral | 109.5° | Tetrahedral | CH4 |
4 | 3 | 1 | Tetrahedral | ~109.5° | Trigonal pyramidal | NH3 |
4 | 2 | 2 | Tetrahedral | ~109.5° | Bent | H2O |
Electronegativity and Bond Polarity
Electronegativity
Electronegativity is the ability of an atom to attract electrons in a covalent bond. It is measured on a scale from 0.7 (least) to 4.0 (most, for fluorine).
Electronegativity increases across a period and decreases down a group.
The difference in electronegativity between two atoms determines bond type:
Electronegativity Difference (ΔEN) | Bond Type | Example |
|---|---|---|
0 – 0.4 | Pure covalent (nonpolar) | Cl2 |
0.4 – 2.0 | Polar covalent | HF |
> 2.0 | Ionic | NaCl |
Bond Polarity and Molecular Polarity
A polar covalent bond has an uneven sharing of electrons, resulting in a dipole moment (partial charges at each end of the bond). A polar molecule has a net dipole moment due to the arrangement of polar bonds.
In molecules with more than two atoms, polar bonds may cancel (nonpolar molecule) or add (polar molecule).
Example: CO2 is nonpolar (linear geometry, dipoles cancel); H2O is polar (bent geometry, dipoles add).
Applications and Everyday Chemistry
Chemistry and Health: Drug Design
Understanding molecular shape and bonding is essential in drug design, such as the development of HIV-protease inhibitors, which fit into the active site of the enzyme and block its function.
Why Oil and Water Don’t Mix
Water is polar, oil is nonpolar. Polar molecules attract each other and exclude nonpolar molecules, leading to separation of oil and water.
How Soap Works
Soap molecules have a polar head (attracts water) and a nonpolar tail (attracts grease/oil), allowing water and oil to mix and wash away grease.
Summary
The Lewis model explains ionic and covalent bonding and predicts molecular shapes.
VSEPR theory is used to determine molecular geometry based on electron group repulsion.
Electronegativity differences determine bond polarity and molecular polarity, affecting physical and chemical properties.
