 Back
BackChapter 10: Chemical Bonding – Study Notes
Study Guide - Smart Notes

Chapter 10: Chemical Bonding
Lewis Dot Symbols (Electron Dot Diagrams)
Lewis Dot Symbols are simplified diagrams that represent the valence electrons of an atom or ion. These diagrams help visualize how atoms bond and interact in chemical compounds.
Valence electrons are the electrons in the outermost shell of an atom and are responsible for chemical bonding.
For Main Group Elements, the number of valence electrons equals the group number (for Groups 1A–8A).
For Transition Metals, valence electrons include both the outermost s and d electrons.
Steps to Draw Lewis Dot Symbols:
Identify the group number of the main group element (ignore transition metals for basic Lewis symbols).
Place one valence electron (dot) at a time on the four sides of the element symbol, starting from the top and moving clockwise.
Pair up electrons as needed until all valence electrons are represented.
Example: Draw the Lewis Dot Symbol for Tellurium (Te): Te is in Group 6A, so it has 6 valence electrons.
Ionic Bonding
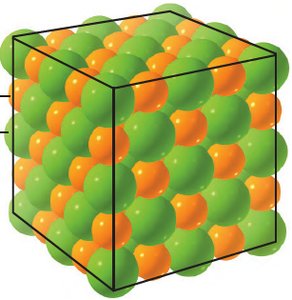
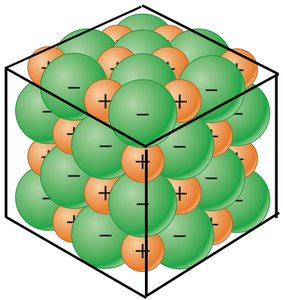
Ionic bonding is the attractive force that holds oppositely charged ions together in a chemical compound. This type of bonding typically occurs between metals and nonmetals.
Metals tend to lose electrons, forming cations (positively charged ions).
Nonmetals tend to gain electrons, forming anions (negatively charged ions).
The resulting electrostatic attraction forms an ionic compound.
Ionic bonding helps lower the potential energy of the ions involved.
Properties of Ionic Compounds:
Usually solid at room temperature.
High melting and boiling points due to strong ionic bonds.
Brittle and hard.
Conduct electricity when dissolved in water (as ions are free to move).

Covalent Bonding
Covalent bonding involves the sharing of valence electrons between nonmetal atoms to achieve a stable electron configuration, typically an octet (8 electrons) or duet (2 electrons for hydrogen).
Octet Rule: Atoms tend to form bonds until they are surrounded by 8 valence electrons (except hydrogen, which follows the duet rule).
Duet Rule: Hydrogen forms bonds to achieve 2 valence electrons.
Properties of Covalent Compounds:
Can be solid, liquid, or gas at room temperature.
Lower melting and boiling points compared to ionic compounds.
Poor electrical conductors (do not conduct electricity in solution).
Lewis Dot Structures for Compounds
Lewis Dot Structures are used to represent the arrangement of valence electrons in molecules and ions. They help predict molecular shape, bond order, and reactivity.
Each bond (single, double, triple) is represented by a pair of shared electrons.
Lone pairs (nonbonding electrons) are shown as pairs of dots on individual atoms.
Steps to Draw Lewis Dot Structures:
Count the total number of valence electrons for all atoms in the molecule or ion.
Place the least electronegative atom in the center (except hydrogen).
Connect atoms with single bonds (pairs of electrons).
Distribute remaining electrons to satisfy the octet (or duet) rule for each atom.
Place any extra electrons on the central atom.
If octets are incomplete, form double or triple bonds as needed.
Example: Draw the Lewis Dot Structure for NH3 (ammonia):
Nitrogen (N) is the central atom, with three hydrogens bonded to it and one lone pair on nitrogen.
Bonding Preferences and Multiple Bonds
Atoms form a specific number of bonds based on their group number and the need to achieve a stable electron configuration.
Group 1A–4A: Number of bonds equals the group number.
Group 5A–7A: Number of bonds equals 8 minus the group number.
Lone pairs are pairs of valence electrons not involved in bonding.
Multiple Bonds: Double and triple bonds are formed when single bonds are insufficient to satisfy the octet rule.
Single bond: 1 pair of shared electrons.
Double bond: 2 pairs of shared electrons.
Triple bond: 3 pairs of shared electrons.
Bond strength increases and bond length decreases from single to triple bonds.
Lewis Dot Structures for Ions
Lewis Dot Structures can also represent ions. For cations, electrons are removed; for anions, electrons are added. The structure is placed in brackets with the charge indicated.
For cations, remove electrons from the least electronegative atom.
For anions, add electrons to satisfy the octet rule.
Example: Draw the Lewis Dot Structure for NH4+ (ammonium ion): Four hydrogens bonded to nitrogen, with no lone pairs on nitrogen, and the structure in brackets with a + charge.
Exceptions to the Octet Rule
Some elements can have fewer or more than 8 electrons around them and still be stable.
Incomplete octet: Group 2A (2 electrons), Group 3A (6 electrons).
Expanded octet: Elements in Groups 5A–8A can have more than 8 electrons (e.g., SF6).
Odd electron molecules (free radicals): Molecules with an odd number of electrons, such as NO2.
Resonance Structures
Some molecules have more than one valid Lewis structure, called resonance structures. The actual structure is a resonance hybrid, an average of all possible resonance forms.
Resonance involves the movement of electrons, not atoms.
Double-sided arrows are used to indicate resonance between structures.
Example: The carbonate ion (CO32–) has three resonance structures, each with a double bond to a different oxygen atom.
Valence Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory predicts the geometry of molecules based on the repulsion between electron groups (bonding and lone pairs) around the central atom.
Electron groups include both bonding pairs and lone pairs.
Lone pairs exert more repulsion than bonding pairs, affecting molecular shape.
Electron Geometry
Electron geometry describes the arrangement of all electron groups (bonding and lone pairs) around the central atom.
2 electron groups: linear geometry (180° bond angle).
3 electron groups: trigonal planar geometry (120° bond angle).
4 electron groups: tetrahedral geometry (109.5° bond angle).
Molecular Geometry
Molecular geometry describes the arrangement of only the atoms (not lone pairs) in a molecule.
Linear, trigonal planar, tetrahedral, trigonal pyramidal, and bent are common shapes.
Lone pairs can alter the ideal bond angles and molecular shape.
Bond Angles
Bond angles are the angles between adjacent bonds in a molecule. Ideal bond angles are determined by electron geometry, but lone pairs can decrease these angles.
Linear: 180°
Trigonal planar: 120°
Tetrahedral: 109.5°
Presence of lone pairs decreases bond angles from the ideal values.
Dipole Moment and Electronegativity
Electronegativity (EN) is a measure of an atom's ability to attract electrons in a bond. The difference in electronegativity between two atoms determines the bond type and polarity.
EN increases across a period (left to right) and decreases down a group.
Dipole moment arises when there is unequal sharing of electrons (polar bonds).
ΔEN > 0.4 is considered significant for polarity.
Dipole arrows point toward the more electronegative atom.
Bond Classification by ΔEN:
ΔEN < 0.4: Nonpolar covalent
ΔEN 0.4–2.0: Polar covalent
ΔEN > 2.0: Ionic


Molecular Polarity
Molecular polarity depends on both the polarity of individual bonds and the overall shape of the molecule.
A molecule is nonpolar if it has a symmetrical (perfect) shape and all bond dipoles cancel out.
A molecule is polar if it has an asymmetrical shape or contains polar bonds that do not cancel.
Examples: CO2 is nonpolar (linear, dipoles cancel); H2O is polar (bent, dipoles do not cancel).
Summary Table: Types of Chemical Bonds
Bond Type | ΔEN Range | Electron Sharing | Example |
|---|---|---|---|
Nonpolar Covalent | 0–0.4 | Equal | Cl2 |
Polar Covalent | 0.4–2.0 | Unequal | H2O |
Ionic | >2.0 | Transfer | NaCl |
Additional info: These notes cover the essential concepts of chemical bonding, including Lewis structures, ionic and covalent bonding, resonance, VSEPR theory, molecular geometry, bond angles, electronegativity, dipole moments, and molecular polarity, as outlined in a typical introductory chemistry curriculum.
