 Back
BackChapter 11: Gases – Properties, Laws, and Applications
Study Guide - Smart Notes

Gases
Kinetic Molecular Theory
The kinetic molecular theory provides a model for understanding the behavior of gases. It explains the properties of gases based on the motion and interactions of their particles. This theory is generally accurate under many conditions but breaks down at low temperatures and high pressures.
Postulate 1: A gas is a collection of particles in constant, random motion.
Postulate 2: Gas particles do not attract or repel each other; they interact only through elastic collisions with each other and with surfaces.
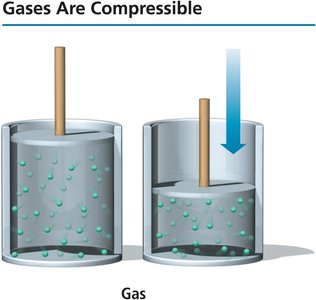
Postulate 3: There is a lot of empty space between gas particles compared to their size.
Postulate 4: The average kinetic energy of gas particles is proportional to the temperature (in kelvin). As temperature increases, particle speed and energy increase.
Properties of Gases Explained by Kinetic Molecular Theory:
Gases are compressible because there is a lot of empty space between particles.
Gases assume the shape and volume of their container.
Gases have low densities compared to liquids and solids.
Pressure
Pressure is the result of constant collisions between gas particles and the surfaces around them. It is defined as force per unit area:
The pressure exerted by a gas depends on the number of particles in a given volume, their speed, and the frequency of collisions with surfaces.
Units of Pressure
Pressure can be measured in several units:
Atmosphere (atm): Average pressure at sea level.
Pascal (Pa): SI unit; .
Millimeter of mercury (mm Hg): Based on the height of mercury in a barometer; .
Torr: .
Pounds per square inch (psi): .
Inches of mercury (in. Hg): .
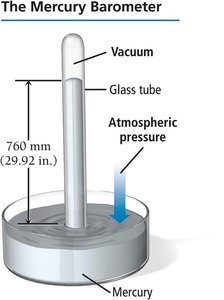
Table: Common Units of Pressure
Unit | Average Air Pressure at Sea Level |
|---|---|
Pascal (Pa) | 101,325 Pa |
Atmosphere (atm) | 1 atm |
Millimeter of mercury (mm Hg) | 760 mm Hg |
Torr (torr) | 760 torr |
Pounds per square inch (psi) | 14.7 psi |
Inches of mercury (in. Hg) | 29.92 in. Hg |
Gas Laws
Boyle’s Law (Pressure–Volume Relationship)
At constant temperature and amount of gas, the pressure of a gas is inversely proportional to its volume:
For two sets of conditions:
If pressure increases, volume decreases, and vice versa.
Charles’s Law (Volume–Temperature Relationship)
At constant pressure and amount of gas, the volume of a gas is directly proportional to its temperature (in kelvin):
All temperatures must be in kelvin.
Avogadro’s Law (Volume–Amount Relationship)
At constant temperature and pressure, the volume of a gas is directly proportional to the number of moles (n):
The Combined Gas Law
When pressure, volume, and temperature all change (amount of gas constant):
The Ideal Gas Law
Combines Boyle’s, Charles’s, and Avogadro’s laws into one equation:
P = pressure (atm)
V = volume (L)
n = moles of gas
R = ideal gas constant ()
T = temperature (K)
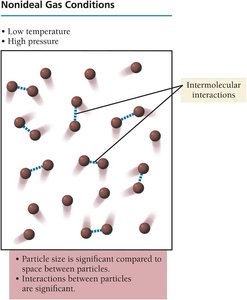
Ideal vs. Non-Ideal Gas Behavior
Gases behave ideally at high temperature and low pressure, where particle size and intermolecular forces are negligible. At low temperature and high pressure, real gases deviate from ideal behavior due to significant intermolecular interactions and particle volume.

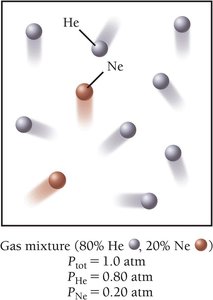
Partial Pressures and Gas Mixtures
In a mixture, each gas exerts its own pressure, called partial pressure. The total pressure is the sum of the partial pressures (Dalton’s Law):
The partial pressure of a component is its fractional composition times the total pressure:
Vapor Pressure and Collecting Gases Over Water
When collecting a gas over water, the total pressure is the sum of the gas pressure and the vapor pressure of water at that temperature. The vapor pressure of water depends on temperature and must be subtracted from the total pressure to find the pressure of the collected gas.
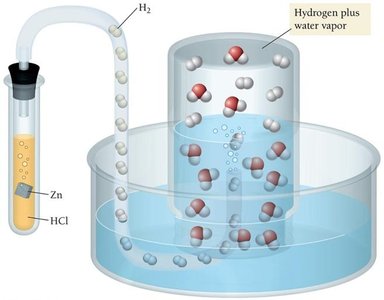
Table: Vapor Pressure of Water vs. Temperature
Temperature (°C) | Vapor Pressure (mm Hg) |
|---|---|
10 | 9.2 |
20 | 17.5 |
25 | 23.8 |
30 | 31.8 |
40 | 55.3 |
50 | 92.5 |
60 | 149.4 |
70 | 233.7 |
80 | 355.1 |
Stoichiometry Involving Gases
For reactions involving gases, use the ideal gas law to relate volume, pressure, and temperature to moles. Then use stoichiometric coefficients to convert between reactants and products.
Molar Volume at Standard Temperature and Pressure (STP)
At STP (0°C or 273.15 K and 1 atm), 1 mole of any ideal gas occupies 22.4 L. This is called the molar volume of a gas at STP and is useful for conversions between moles and volume.
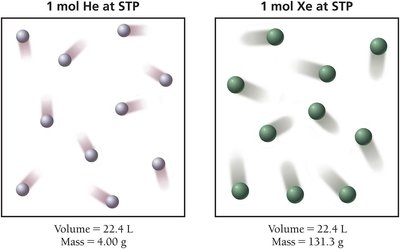
1 mol gas at STP = 22.4 L
Summary Table: Simple Gas Laws and the Ideal Gas Law
Variables Changing | Variables Constant | Simple Gas Law | Equation |
|---|---|---|---|
V and P | n and T | Boyle's Law | |
V and T | n and P | Charles's Law | |
P and T | n and V | Gay-Lussac's Law | |
V and n | T and P | Avogadro's Law |
