 Back
BackChapter 13: Solutions – Properties, Preparation, and Calculations
Study Guide - Smart Notes

Solutions and Mixtures
Types of Mixtures
Mixtures are combinations of two or more substances that are not chemically bonded. They can be classified as either homogeneous or heterogeneous based on their uniformity.
Homogeneous Mixture (Solution): A mixture with a uniform composition throughout. Every portion is identical. Examples: Salt water, sweetened tea.
Heterogeneous Mixture: A mixture with a non-uniform composition; different regions have different properties. Example: Oil and water mixture.
Examples of Solutions
Solutions can involve combinations of gases, liquids, and solids. For example, the ocean is a solution of salts in water, and blood plasma is a solution of various solids and gases in water.
Solutions are found in nature and in biological systems, such as dissolved gases in water for aquatic life and gases dissolved in blood.
Formation of Solutions
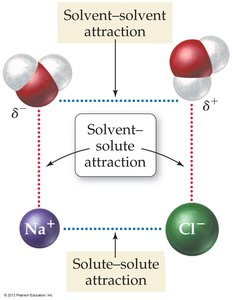
Solute-Solvent Interactions
When a solid dissolves in water, the process depends on the interactions between solute and solvent particles. The attractions between solvent and solute must overcome the attractions among solute particles and among solvent particles.
Solvent-solvent attraction: Forces holding solvent molecules together.
Solute-solute attraction: Forces holding solute particles together.
Solvent-solute attraction: Forces between solvent and solute particles; these must be strong enough for dissolution to occur.
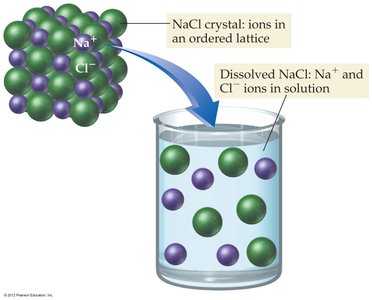
Dissolution of Ionic Compounds
When ionic compounds like NaCl dissolve in water, the ions are separated and surrounded by water molecules, a process called hydration.
The positive ends of water dipoles are attracted to anions, and the negative ends are attracted to cations.
This disperses the ions throughout the solution.
Solubility and Saturation
Definitions
Solubility: The maximum amount of solute (in grams) that can dissolve in a given amount of solvent at a specific temperature.
Saturated Solution: Contains the maximum amount of dissolved solute at a given temperature; additional solute will not dissolve.
Unsaturated Solution: Contains less than the maximum amount of solute; more solute can dissolve.
Supersaturated Solution: Contains more than the normal maximum amount of solute; excess solute will precipitate out.
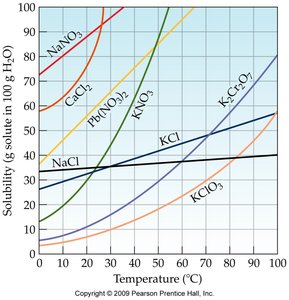
Effect of Temperature on Solubility
For most solid solutes, solubility increases with temperature. However, the degree of increase varies by compound.
Solutions of Gases in Water
Gas Solubility and Temperature
The solubility of gases in liquids generally decreases as temperature increases. This is why warm soda fizzes more than cold soda—dissolved CO2 escapes more readily at higher temperatures.
Gases like O2, CO2, and N2 are found dissolved in water and are essential for aquatic life and biological processes.
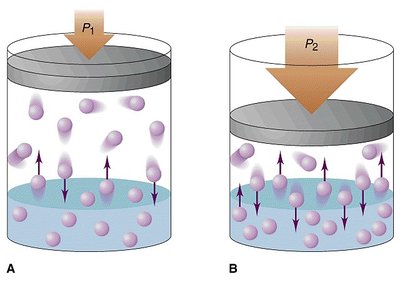
Henry's Law
The solubility of a gas in a liquid is directly proportional to the pressure of the gas above the liquid. Higher pressure increases gas solubility.
Specifying Solution Concentration
Molarity (M)
Molarity is a common unit of concentration, defined as the number of moles of solute per liter of solution.
Formula:
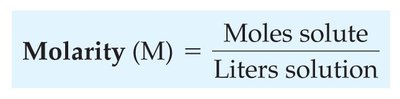
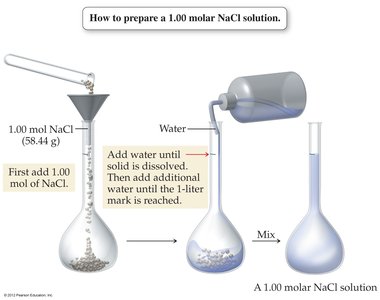
Preparing a Solution of Specific Molarity
To prepare a solution of known molarity, dissolve the calculated amount of solute in water and dilute to the desired final volume.
Example Calculation: Molarity
Calculate the molarity of a solution made by dissolving 15.5 g NaCl in enough water to make 1.50 L of solution.
Step 1: Convert grams NaCl to moles.
Step 2: Divide moles by liters of solution.

Example Calculation: Mass of Solute from Molarity and Volume
To find the mass of sucrose in a given volume and molarity, use the relationships between volume, molarity, and molar mass.
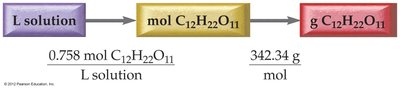

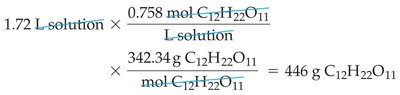
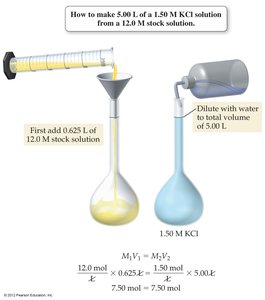
Solution Dilution
Dilution Equation
To prepare a less concentrated solution from a stock solution, use the dilution equation:
Where and are the molarity and volume of the stock solution, and and are those of the diluted solution.
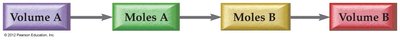
Solution Stoichiometry
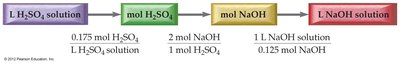
Using Concentration and Volume in Reactions
In aqueous reactions, the amount of reactant or product can be calculated using solution volume and concentration, then applying stoichiometry from the balanced equation.
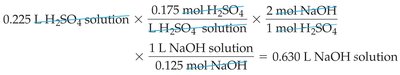
Other Ways to Express Concentration
Mass Percent
Mass percent expresses the concentration as grams of solute per 100 grams of solution.
Formula:
Colligative Properties
Definition and Types
Colligative properties depend on the number of solute particles in a solution, not their identity. These include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.
Freezing Point Depression: The freezing point of a solution is lower than that of the pure solvent.
Boiling Point Elevation: The boiling point of a solution is higher than that of the pure solvent.
Osmotic Pressure: The pressure required to stop the net flow of solvent through a semipermeable membrane into a solution.
Freezing Point Depression
The decrease in freezing point is proportional to the molality of the solution:
Where is the change in freezing point, is molality, and is the freezing point depression constant.

Boiling Point Elevation
The increase in boiling point is proportional to the molality of the solution:
Where is the change in boiling point, is molality, and is the boiling point elevation constant.

Osmosis and Osmotic Pressure
Osmosis is the net flow of solvent (usually water) across a semipermeable membrane from a region of lower solute concentration to higher solute concentration. Osmotic pressure is the pressure required to stop this flow.
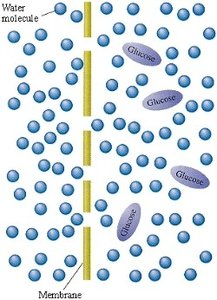
Additional info: Colligative properties are important in biological and environmental systems, such as antifreeze in car engines and the regulation of water balance in cells.
