 Back
BackChapter 13: Solutions – Study Notes for Introductory Chemistry
Study Guide - Smart Notes

Solutions
Concept of Solutions
Solutions are homogeneous mixtures composed of two or more substances that form a uniform composition throughout. The main components of a solution are the solute and the solvent.
Solute: The substance present in a lesser amount, which is dissolved in the solvent.
Solvent: The substance present in the greater amount, which dissolves the solute.
Concentration: A measure of the amount of solute in a given quantity of solvent or solution.
Example: If 10.0 g NaCl is dissolved in 500.0 mL of water, NaCl is the solute and water is the solvent.
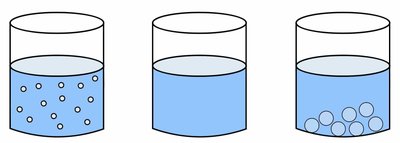
Types of Mixtures: Solutions, Suspensions, and Colloids
Mixtures can be classified based on the size of the solute particles and their behavior in the solvent.
Solutions: Particle size < 1 nm; particles do not settle; homogeneous (e.g., salt water).
Colloids: Particle size between 1 nm and 500 nm; particles do not settle; appear cloudy (e.g., milk).
Suspensions: Particle size > 500 nm; particles settle out upon standing; heterogeneous (e.g., salad dressing).
Solubility and Intermolecular Forces
Like Dissolves Like Principle
Solubility depends on the nature of the solute and solvent. According to the principle of like dissolves like, substances with similar intermolecular forces or polarity will dissolve in each other.
Polar solutes dissolve in polar solvents (e.g., NaCl in water).
Nonpolar solutes dissolve in nonpolar solvents (e.g., oil in hexane).
Example: Methanol (CH3OH, polar) will dissolve in water (polar), but not in hexane (nonpolar).
Concentration Units
Mass Percent
Mass percent expresses the mass of solute as a percentage of the total mass of the solution.
Formula:
Example: A solution with 23.0 g NaOH in 100 g solution is 23.0% NaOH by mass.
Molarity (M)
Molarity is the number of moles of solute per liter of solution. It is a common unit for expressing solution concentration.
Formula:
Example: To prepare a 0.550 M Na3PO4 solution, dissolve the appropriate mass in enough water to make 300.0 mL of solution.
Osmolarity
Definition and Calculation
Osmolarity is the total concentration of all ions in solution, expressed as moles of ions per liter of solution.
Formula:
Example: For AlCl3, which dissociates into 4 ions (1 Al3+ and 3 Cl-), a 0.10 M solution has an osmolarity of 0.40 osmol/L.
Solubility and Temperature
Saturated, Unsaturated, and Supersaturated Solutions
The amount of solute that can dissolve in a solvent depends on temperature.
Saturated solution: Contains the maximum amount of dissolved solute at a given temperature.
Unsaturated solution: Can dissolve more solute at the same temperature.
Supersaturated solution: Contains more solute than is stable at that temperature.
Temperature Effects
As temperature increases, the solubility of most solids in liquids increases, while the solubility of gases in liquids decreases.

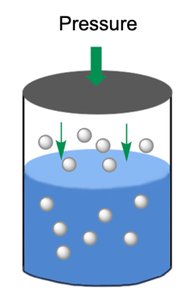
Henry’s Law
Gas Solubility and Pressure
Henry’s Law states that the solubility of a gas in a liquid is directly proportional to the partial pressure of the gas above the liquid.
Formula: (where C is concentration, k is Henry’s constant, and P is pressure)
Dilutions
Preparation of Dilute Solutions
Dilution involves adding solvent to a solution to decrease its concentration. The relationship is given by:
Formula:
and are the initial molarity and volume; and are after dilution.

Solution Stoichiometry
Stoichiometric Calculations in Solution
Solution stoichiometry involves using molarity and volume to determine the amounts of reactants and products in chemical reactions.
Steps: Convert given quantity to moles, use mole ratios, convert to desired units.
Molality (m)
Definition and Calculation
Molality is the number of moles of solute per kilogram of solvent. It is used when temperature changes are involved, as it does not depend on volume.
Formula:
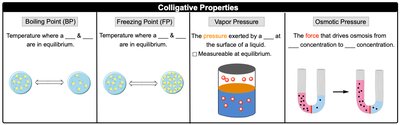
Colligative Properties
Overview
Colligative properties depend on the number of solute particles in solution, not their identity. The four main colligative properties are:
Boiling Point Elevation
Freezing Point Depression
Vapor Pressure Lowering
Osmotic Pressure
Boiling Point Elevation
Adding a solute to a solvent raises the boiling point of the solvent.
Formula:
= van’t Hoff factor (number of particles formed per formula unit)
= boiling point elevation constant
= molality
Freezing Point Depression
Adding a solute to a solvent lowers the freezing point of the solvent.
Formula:
= freezing point depression constant
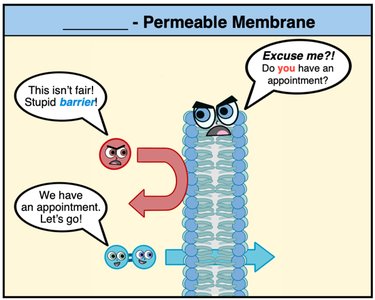
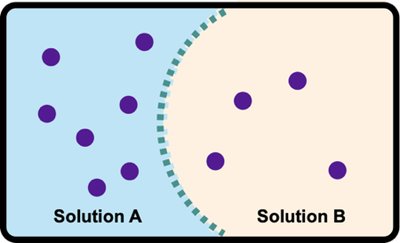
Osmosis and Osmotic Pressure
Osmosis
Osmosis is the movement of solvent molecules (usually water) across a semipermeable membrane from a region of lower solute concentration to higher solute concentration.
Semipermeable membrane: Allows solvent but not solute to pass through.
Osmotic pressure: The pressure required to stop the net flow of solvent.
Tonicity and Red Blood Cells
Tonicity describes the relative concentration of solutes in two solutions separated by a semipermeable membrane.
Hypotonic: Lower solute concentration outside the cell; water enters the cell (cell swells).
Isotonic: Equal solute concentration; no net water movement.
Hypertonic: Higher solute concentration outside the cell; water leaves the cell (cell shrinks).
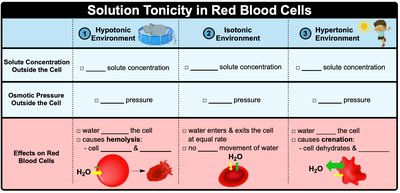
Example: A red blood cell placed in pure water (hypotonic) will swell and may burst (hemolysis).
Additional info: Colligative properties are important in biological and industrial processes, such as intravenous fluid preparation and antifreeze formulation.
