 Back
BackChapter 2: The Numerical Side of Chemistry – Precision, Accuracy, and Measurement
Study Guide - Smart Notes

2.1 Numbers in Chemistry—Precision and Accuracy
Exact Numbers vs. Measured Numbers
In chemistry, numbers are used to express quantities and measurements. There are two main types of numerical quantities:
Exact numbers: Values with no uncertainty, such as counted objects (e.g., 12 pennies).
Measured numbers: Values obtained through measurement, which always contain some uncertainty (e.g., a penny is 2.0 cm in diameter).
Precision and Accuracy
Measurements in chemistry are evaluated based on their precision and accuracy:
Precision: How close a set of measurements are to each other. Requires multiple measurements and is often reported as an average.
Accuracy: How close a measurement is to the true or accepted value. Only one measurement is needed to assess accuracy.
The goal in scientific measurement is to achieve both high precision and high accuracy.

Example: Counting Grains of Rice
Given two students' repeated measurements of rice grains, one may be more precise (consistent results), while the other may be more accurate (closer to the true value). Precision and accuracy are independent qualities.
2.2 Uncertainty and Significant Figures
Uncertainty in Measurement
All measured values have an associated uncertainty, typically in the last digit recorded. This uncertainty arises because measurements are limited by the precision of the measuring instrument and human estimation.

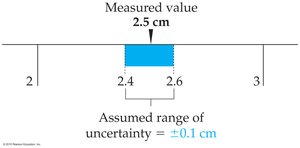
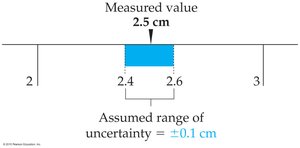
Determining Uncertainty
The uncertainty of a measurement depends on the smallest division of the measuring instrument. For example, if a ruler is marked in tenths of a centimeter, the uncertainty is typically ±0.1 cm.
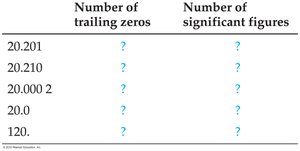
2.3 Zeros and Significant Figures
Rules for Significant Figures
Significant figures (sig figs) reflect the precision of a measured value. Zeros can be tricky:
Leading zeros: Zeros before the first nonzero digit are not significant (e.g., 0.06 has one significant figure).
Trailing zeros: Zeros to the right of the last nonzero digit are significant if they appear after a decimal point (e.g., 0.001020 has four significant figures).
Interpreting Uncertainty in Whole Numbers
Whole numbers without a decimal point can have ambiguous uncertainty. For example, 600 could mean ±100, ±10, or ±1, but 600. (with a decimal) means ±1.
2.4 Scientific Notation
Purpose and Structure
Scientific notation is used to clearly indicate the uncertainty and to conveniently express very large or small numbers. It is written as:
A is a number (the coefficient) with the correct number of significant figures.
x is an integer exponent indicating the power of ten.

Converting Between Standard and Scientific Notation
Move the decimal point to the right of the first nonzero digit. The number of places moved determines the exponent.
If the decimal moves left, the exponent is positive; if right, the exponent is negative.

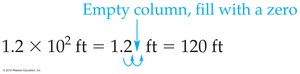
2.5 Significant Figures in Calculations
Rules for Mathematical Operations
Multiplication and Division: The result should have the same number of significant figures as the measurement with the fewest significant figures.
Addition and Subtraction: The result should have the same number of decimal places as the measurement with the fewest decimal places.

Combining Operations
When calculations involve both types of operations, apply the rules stepwise and round only at the end.

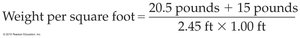


2.6 Units of Measure
SI Units and Prefixes
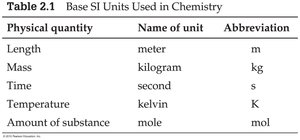
The International System of Units (SI) is the standard in science. Common base units include meter (m), kilogram (kg), second (s), kelvin (K), and mole (mol).
Physical quantity | Name of unit | Abbreviation |
|---|---|---|
Length | meter | m |
Mass | kilogram | kg |
Time | second | s |
Temperature | kelvin | K |
Amount of substance | mole | mol |
Comparing U.S. and Metric Units
Metric units are used internationally, while the U.S. uses customary units. For example, 1 meter is about 3.37 inches longer than a yard.

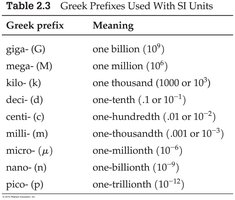
SI Prefixes
Prefixes are used to express very large or small quantities conveniently.
Greek prefix | Meaning |
|---|---|
giga- (G) | one billion (109) |
mega- (M) | one million (106) |
kilo- (k) | one thousand (103) |
deci- (d) | one-tenth (0.1 or 10-1) |
centi- (c) | one-hundredth (0.01 or 10-2) |
milli- (m) | one-thousandth (0.001 or 10-3) |
micro- (μ) | one-millionth (10-6) |
nano- (n) | one-billionth (10-9) |
pico- (p) | one-trillionth (10-12) |
Temperature Scales and Conversions
Temperature can be measured in Celsius (°C), Kelvin (K), or Fahrenheit (°F). The Kelvin scale is the SI unit and is an absolute scale.
To convert between Celsius and Kelvin:
To convert between Celsius and Fahrenheit:
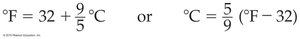

2.7 Density: A Useful Physical Property of Matter
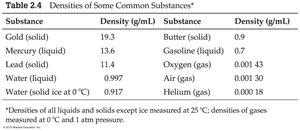
Definition and Calculation
Density is defined as mass per unit volume and is an intensive property (does not depend on the amount of material).
Common units: g/mL or g/cm3

Substance | Density (g/mL) | Substance | Density (g/mL) |
|---|---|---|---|
Gold (solid) | 19.3 | Butter (solid) | 0.9 |
Mercury (liquid) | 13.6 | Gasoline (liquid) | 0.7 |
Lead (solid) | 11.4 | Oxygen (gas) | 0.00143 |
Water (liquid) | 0.997 | Air (gas) | 0.00130 |
Water (ice) | 0.917 | Helium (gas) | 0.00018 |
Measuring Density
Mass is measured with a balance.
Volume can be measured directly (for regular shapes) or by water displacement (for irregular shapes).

2.8 Doing Calculations in Chemistry—Unit Analysis
Unit Analysis (Dimensional Analysis)
Unit analysis is a systematic method for converting between units using conversion factors. The process involves:
Writing the given measurement with its unit.
Multiplying by conversion factors to cancel unwanted units and introduce desired units.
Performing the calculation after all units are converted appropriately.


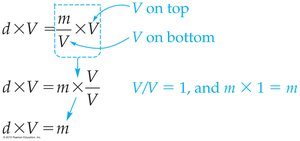
2.9 Rearranging Equations—Algebraic Manipulations with Density
Solving for Different Variables
The density equation can be rearranged to solve for mass or volume:
To solve for mass:
To solve for volume:

2.10 Quantifying Energy
Energy in Physical and Chemical Changes
All changes in matter involve energy, which is the capacity to do work. Energy can be in the form of heat or light.
Units of Energy
Calorie (cal): Not an SI unit. 1 cal is the energy needed to raise 1 g of water by 1°C.
Joule (J): The SI unit of energy.
Food Calories (Cal) are kilocalories:
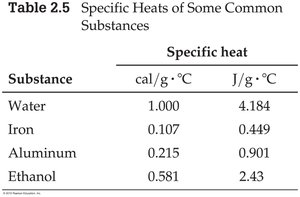
Specific Heat
Specific heat (C) is the amount of heat required to raise the temperature of 1 g of a substance by 1°C.
Substance | cal/g·°C | J/g·°C |
|---|---|---|
Water | 1.000 | 4.184 |
Iron | 0.107 | 0.449 |
Aluminum | 0.215 | 0.901 |
Ethanol | 0.581 | 2.43 |
Calculating Heat
The amount of heat (q) absorbed or released is calculated as:
m = mass (g)
C = specific heat (J/g·°C or cal/g·°C)
= final temperature
= initial temperature
Calorimetry
A calorimeter is an instrument used to measure the energy released or absorbed during a chemical or physical process.
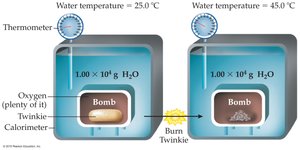
Example: Energy Released from a Burning Twinkie
To find the energy released, use the specific heat equation with the mass of water and the temperature change:

