 Back
BackChapter 3: Matter and Energy – Structured Study Notes for Introductory Chemistry
Study Guide - Smart Notes

Matter and Energy
Atoms and Molecules in Matter
All visible and invisible substances in our environment are composed of matter. The differences between types of matter arise from the distinct arrangements and types of atoms and molecules that constitute them. Atoms are the fundamental building blocks, and molecules are groups of atoms bonded in specific geometric arrangements.
Atoms: Smallest unit of an element, indivisible by chemical means.
Molecules: Two or more atoms bonded together, forming distinct structures.
Example: Water molecules (H2O) and carbon atoms in graphite.

Additional info: Advances in microscopy, such as Scanning Tunneling Microscopy (STM), allow visualization of individual atoms and molecules.
Defining Matter
Matter is anything that occupies space and has mass. It can be visible (steel, water, wood) or invisible (air, microscopic dust).
Key Point: Matter appears continuous but is composed of discrete atoms and molecules.
The Common States of Matter
Matter exists in three primary states: solid, liquid, and gas. These states differ in the arrangement, spacing, and motion of their constituent particles.
Solid: Atoms/molecules are closely packed in fixed positions; definite shape and volume.
Liquid: Atoms/molecules are close but can move past each other; definite volume, variable shape.
Gas: Atoms/molecules are far apart and move freely; variable shape and volume, compressible.
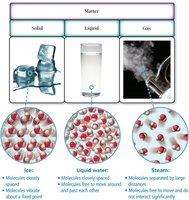
Properties of Solids, Liquids, and Gases
Solids, liquids, and gases exhibit distinct physical properties, such as compressibility and rigidity.
Solids: Not compressible, rigid structure.
Gases: Compressible, particles far apart.

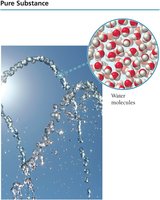
Classifying Matter: Pure Substances and Mixtures
Matter is classified as either pure substances or mixtures based on its composition.
Pure Substance: Composed of only one kind of atom or molecule.
Mixture: Composed of two or more kinds of atoms or molecules in variable proportions.
Elements
An element is a pure substance that cannot be broken down into simpler substances. The smallest particle is an atom.
Compounds
A compound is a pure substance composed of two or more elements in fixed, definite proportions. Compounds can be decomposed into simpler substances.
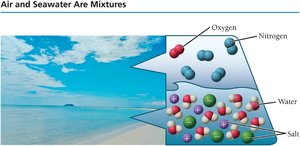
Mixtures
Mixtures can be heterogeneous (variable composition, e.g., oil and water) or homogeneous (uniform composition, e.g., sweetened tea).
Differences in Matter: Chemical and Physical Properties
Properties of matter are classified as physical or chemical.
Physical Property: Observed without changing composition (e.g., odor, boiling point, color).
Chemical Property: Observed only by changing composition (e.g., rusting, flammability).
Changes in Matter: Physical and Chemical Changes
Matter undergoes physical changes (appearance changes, composition unchanged) and chemical changes (composition changes, new substances formed).
Physical Change: State changes (melting, boiling), cutting, crushing.
Chemical Change: Chemical reactions, color or temperature change, formation of new substances.


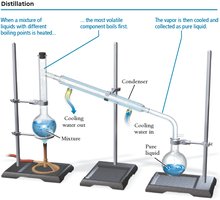
Separating Mixtures: Physical Methods
Mixtures can be separated by physical changes, such as distillation and filtration.
Distillation: Separates based on differences in boiling points.
Filtration: Separates solids from liquids using filter paper.

Law of Conservation of Mass
The law of conservation of mass states that matter is neither created nor destroyed in a chemical reaction. The total mass of reactants equals the total mass of products.
Example: Burning butane: 58 g butane + 208 g oxygen → 176 g carbon dioxide + 90 g water; total mass before and after is 266 g.
Energy in Chemistry
Energy is the capacity to do work. It is conserved and can be transferred or transformed but not created or destroyed.
Kinetic Energy: Energy of motion.
Potential Energy: Energy due to position or composition.


Forms and Units of Energy
Energy exists in various forms: electrical, thermal, and chemical. The SI unit is the joule (J). Other units include calorie (cal), Calorie (Cal), and kilowatt-hour (kWh).
1 cal = 4.184 J
1 Cal = 1000 cal
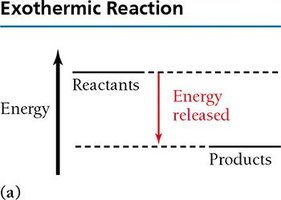
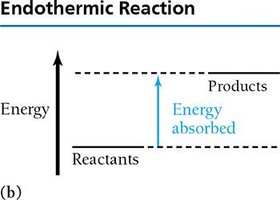
Exothermic and Endothermic Reactions
Chemical reactions can release (exothermic) or absorb (endothermic) energy.
Exothermic: Energy released, products have lower energy than reactants.
Endothermic: Energy absorbed, products have higher energy than reactants.
Temperature and Heat
Temperature measures the thermal energy of matter, while heat is the transfer of thermal energy due to temperature difference.
Temperature Scales: Fahrenheit (°F), Celsius (°C), Kelvin (K).
Absolute zero: 0 K, the point where molecular motion virtually stops.
Converting Between Temperature Scales
Temperature can be converted between scales using the following formulas:
Fahrenheit to Celsius:
Celsius to Fahrenheit:
Celsius to Kelvin:
Specific Heat Capacity
Specific heat capacity is the amount of heat required to raise the temperature of 1 gram of a substance by 1°C. It is measured in J/g·°C.
Water: Has a high specific heat capacity, meaning it absorbs a lot of heat with little temperature change.
The relationship between heat, mass, specific heat, and temperature change is given by:
q: Heat (Joules)
m: Mass (grams)
C: Specific heat capacity (J/g·°C)
ΔT: Change in temperature (°C)
Summary Table: Classification of Matter
Type | Definition | Examples |
|---|---|---|
Element | Pure substance, cannot be decomposed | Helium, Iron |
Compound | Pure substance, two or more elements in fixed proportions | Water (H2O), Salt (NaCl) |
Homogeneous Mixture | Uniform composition throughout | Air, Sweetened tea |
Heterogeneous Mixture | Variable composition | Oil and water, Salad |
Summary Table: Properties of States of Matter
State | Shape | Volume | Compressibility |
|---|---|---|---|
Solid | Fixed | Fixed | No |
Liquid | Variable (container) | Fixed | No |
Gas | Variable (container) | Variable | Yes |
Learning Objectives
Define matter, atoms, and molecules.
Classify matter as solid, liquid, or gas.
Classify matter as element, compound, or mixture.
Distinguish between physical and chemical properties and changes.
Apply the law of conservation of mass.
Recognize different forms of energy and convert between energy units.
Distinguish between exothermic and endothermic reactions.
Convert between Fahrenheit, Celsius, and Kelvin temperature scales.
Relate energy, temperature change, and heat capacity.
Perform calculations involving transfer of heat and changes in temperature.
