 Back
BackChapter 3: Matter and Energy – Structured Study Notes
Study Guide - Smart Notes

Matter and Energy
Atoms and Molecules in Everyday Life
All visible and invisible objects in our environment are composed of matter, which is fundamentally made up of atoms and molecules. The differences in the types of matter arise from the differences in the molecular and atomic structures that compose them.
Atoms: The basic building blocks of matter.
Molecules: Groups of two or more atoms bonded in specific geometric arrangements.
Example: Water molecules and carbon atoms in graphite illustrate how different substances are composed of distinct atomic and molecular structures.

Defining Matter
Matter is anything that occupies space and has mass. It can be visible (e.g., steel, water, wood, plastic) or invisible without magnification (e.g., air, microscopic dust).
Key Point: Matter may appear smooth and continuous, but it is composed of discrete atoms and molecules.
Matter Is Composed of Atoms and Molecules
Recent advances in microscopy allow us to visualize atoms and molecules, confirming that matter is not continuous but made up of submicroscopic particles.
Atoms: Fundamental particles of matter.
Molecules: Atoms bonded together in specific arrangements.
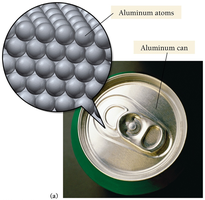
Independent Atomic Particles in Aluminum
In some substances, such as aluminum, atoms exist as independent particles rather than bonded molecules.
Example: Aluminum can is composed of individual aluminum atoms.
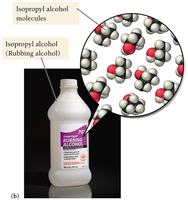
Well-Defined Molecular Particles in Isopropanol
In other substances, such as rubbing alcohol (isopropanol), atoms bond together to form well-defined molecules.
Example: Isopropyl alcohol molecules are composed of several atoms bonded in a specific structure.
Imaging Atoms and Molecules
Scanning tunneling microscopes (STM) can distinguish individual atoms and molecules, providing visual evidence of their existence.
Example: STM images of nickel atoms and DNA molecules show atomic and molecular structures.


States of Matter
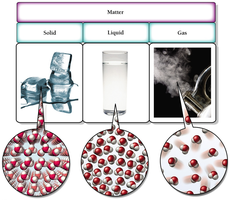
The Common States of Matter
Matter exists in three common states: solid, liquid, and gas. Each state is characterized by the arrangement and movement of its atoms or molecules.
Solid: Molecules are closely spaced and vibrate about fixed points.
Liquid: Molecules are closely spaced but free to move around each other.
Gas: Molecules are separated by large distances and move freely.
Example: Water exists as ice (solid), liquid water, and steam (gas).
States of Matter: Solid
In solids, atoms or molecules are packed closely in fixed locations. Solids have a fixed volume and rigid shape.
Examples: Ice, diamond, quartz, iron.
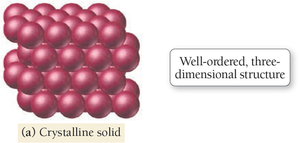
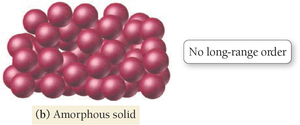
Types of Solids: Crystalline vs. Amorphous
Solids can be classified as crystalline or amorphous based on the arrangement of their atoms or molecules.
Crystalline Solid: Atoms/molecules arranged in geometric patterns with long-range order (e.g., salt, diamond).
Amorphous Solid: Atoms/molecules lack long-range order (e.g., glass, rubber, plastic).
States of Matter: Liquid
Liquids have a fixed volume but assume the shape of their container. Atoms or molecules are close together but can move past each other.
Examples: Water, gasoline, alcohol, mercury.
States of Matter: Gas
Gases are compressible and always assume the shape and volume of their container. Atoms or molecules are separated by large distances and move freely.
Examples: Oxygen, helium, carbon dioxide.
Classification of Matter
Pure Substances and Mixtures
Matter can be classified according to its composition as either a pure substance or a mixture.
Pure Substance: Composed of only one type of atom or molecule.
Mixture: Composed of two or more different types of atoms or molecules combined in variable proportions.
Elements and Compounds
Pure substances are further classified as elements or compounds.
Element: Cannot be broken down into simpler substances; listed in the periodic table.
Compound: Composed of two or more elements in fixed, definite proportions; can be decomposed into simpler substances.
Example: Helium (element), water (compound).
Mixtures: Homogeneous and Heterogeneous
Mixtures can be homogeneous (uniform composition) or heterogeneous (composition varies from region to region).
Homogeneous Mixture: Same composition throughout (e.g., sweetened tea).
Heterogeneous Mixture: Composition varies (e.g., hydrocarbon and water).
Classification Table of Matter
The classification of matter can be summarized in the following table:
Type | Examples |
|---|---|
Element | Copper, Helium |
Compound | Water, Sugar |
Homogeneous Mixture | Sweetened tea, Air |
Heterogeneous Mixture | Hydrocarbon and water, Seawater |
Properties and Changes of Matter
Physical vs. Chemical Properties
Properties of matter are classified as physical or chemical.
Physical Property: Displayed without changing composition (e.g., odor, boiling point).
Chemical Property: Displayed only through changing composition (e.g., flammability, rusting).
Physical and Chemical Changes
Changes in matter can be physical or chemical.
Physical Change: Appearance changes, composition does not (e.g., melting, boiling).
Chemical Change: Composition changes, new substances are formed (e.g., rusting, burning).
Conservation Laws
Law of Conservation of Mass
Matter is neither created nor destroyed in a chemical reaction. The total mass remains constant during physical and chemical changes.
Example: Burning butane produces carbon dioxide and water, but the total mass of reactants equals the total mass of products.
Law of Conservation of Energy
Energy is neither created nor destroyed. The total amount of energy is constant, though it can be transferred or converted between forms.
Key Point: Energy can be changed from one form to another and transferred between objects.
Energy in Chemistry
Forms of Energy
The total energy of a sample is the sum of its kinetic and potential energy.
Kinetic Energy: Energy associated with motion.
Potential Energy: Energy associated with position or composition.
Electrical Energy: Energy from the flow of electrical charge.
Thermal Energy: Energy from random motion of atoms and molecules.
Chemical Energy: Potential energy in chemical systems.
Units of Energy
Energy is measured in several units:
Joule (J): SI unit of energy.
Calorie (cal): Energy required to raise 1 g of water by 1°C.
Calorie (Cal): Nutritional unit, 1 Cal = 1000 cal.
Kilowatt-hour (kWh): Used for electrical energy.
Energy Conversion Factors
Common conversion factors:
Unit | Conversion |
|---|---|
1 Cal | 1000 cal |
1 cal | 4.184 J |
1 kWh | 3.60 × 106 J |
Exothermic and Endothermic Reactions
Energy changes in chemical reactions:
Exothermic Reaction: Releases energy.
Endothermic Reaction: Absorbs energy.
Temperature and Heat
Temperature Scales
Temperature is measured in Fahrenheit, Celsius, and Kelvin scales.
Fahrenheit (°F): Water freezes at 32°F, boils at 212°F.
Celsius (°C): Water freezes at 0°C, boils at 100°C.
Kelvin (K): Water freezes at 273 K, boils at 373 K; absolute zero is 0 K.
Converting Between Temperature Scales
Conversion formulas:
Heat Capacity and Specific Heat
Heat capacity is the quantity of heat required to change the temperature of a substance by 1°C. Specific heat capacity is the heat required per gram per degree Celsius.
Equation:
q: Heat (Joules)
m: Mass (grams)
C: Specific heat capacity (J/g°C)
ΔT: Temperature change (°C)
Summary Table: Properties of Solids, Liquids, and Gases
State | Shape | Volume | Compressibility |
|---|---|---|---|
Solid | Rigid | Fixed | No |
Liquid | Variable (container) | Fixed | No |
Gas | Variable (container) | Variable | Yes |
Review and Learning Objectives
Classify matter as element, compound, or mixture.
Distinguish between physical and chemical properties.
Distinguish between physical and chemical changes.
Apply the law of conservation of mass.
Identify and convert among energy units.
Convert between Fahrenheit, Celsius, and Kelvin temperature scales.
Perform calculations involving transfer of heat and changes in temperature.
