 Back
BackChapter 5: Ionic and Covalent Compounds – Structured Study Notes
Study Guide - Smart Notes

Compounds
Definition and Examples
A compound is a substance composed of two or more elements combined in a specific ratio and held together by chemical bonds. Compounds are fundamental to chemistry, as they exhibit properties distinct from their constituent elements.
Key Point 1: Compounds are formed from elements in fixed ratios.
Key Point 2: Examples include water (H2O) and sodium chloride (NaCl).
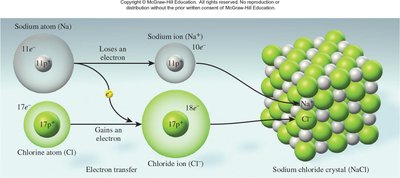
Example: Water is composed of two hydrogen atoms and one oxygen atom, while sodium chloride consists of one sodium atom and one chlorine atom.

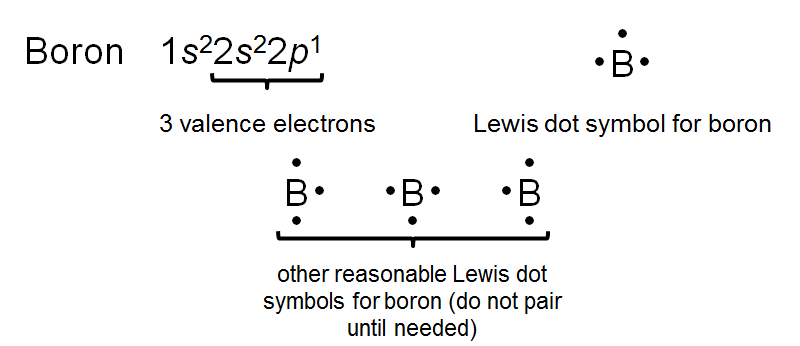
Lewis Dot Symbols
Valence Electrons and Representation
When atoms form compounds, their valence electrons interact. Lewis dot symbols visually represent these electrons as dots around the element's symbol, aiding in understanding bonding and ion formation.
Key Point 1: Each dot represents a valence electron.
Key Point 2: The number of unpaired dots indicates the number of bonds an atom can form.
Example: Sodium (Na) has one dot, indicating one valence electron; oxygen (O) has six dots, with two unpaired, indicating it can form two bonds.
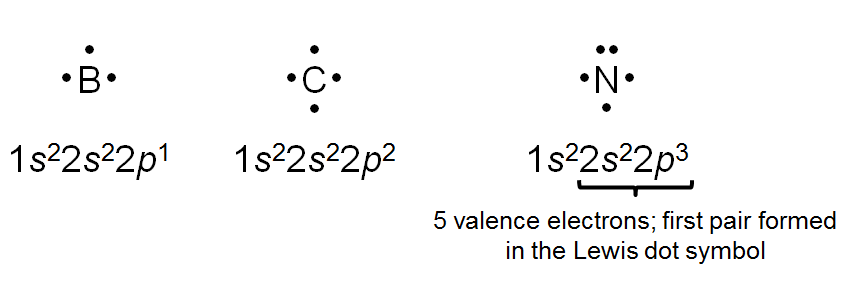
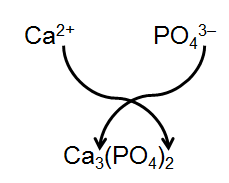
Ionic Compounds and Bonding
Formation and Lattice Energy
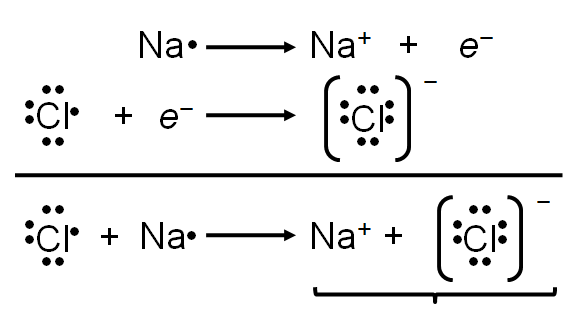
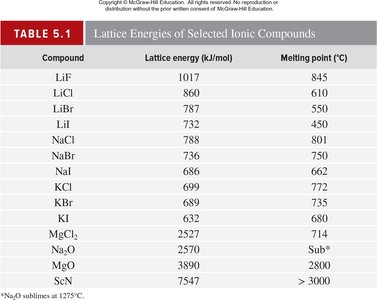
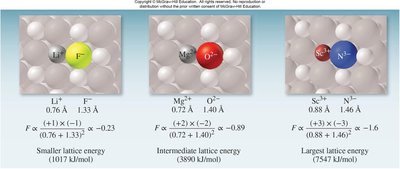
Ionic bonding is the electrostatic attraction between oppositely charged ions, resulting in the formation of ionic compounds. The stability of these compounds is measured by lattice energy, which depends on the charges and distances between ions.
Key Point 1: Ionic compounds are electrically neutral; the sum of charges must be zero.
Key Point 2: Lattice energy increases with higher charges and smaller distances between ions.
Example: Sodium chloride (NaCl) forms from Na+ and Cl− ions, creating a stable lattice.
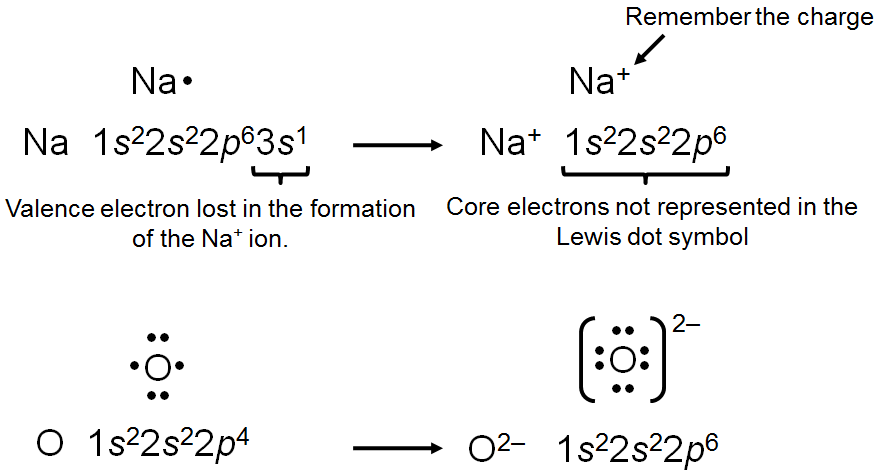
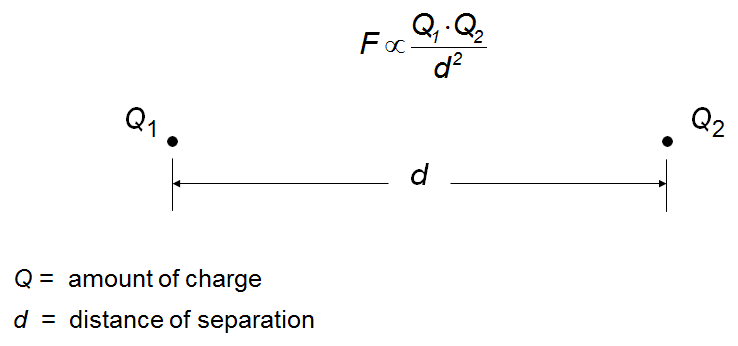


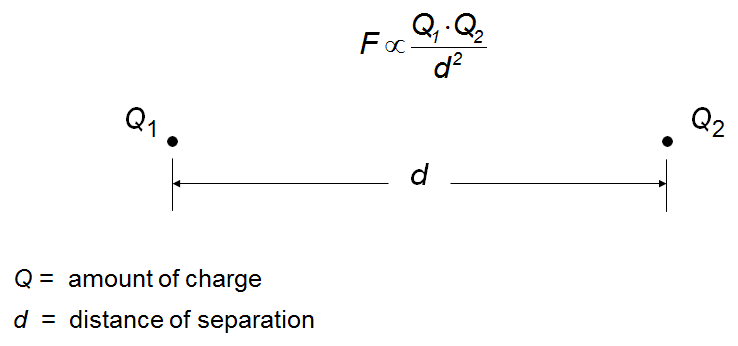
Naming Ions and Ionic Compounds
Monatomic and Polyatomic Ions
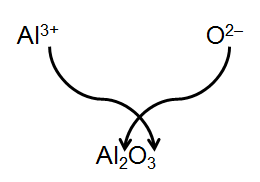
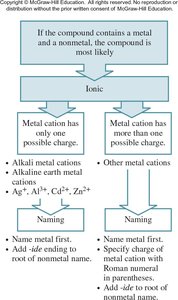
Ions are named systematically. Monatomic cations are named by adding 'ion' to the element name, while monatomic anions change the ending to '-ide'. Some metals form cations with multiple charges, requiring Roman numerals in their names.
Key Point 1: Cations: potassium ion (K+), iron(III) ion (Fe3+).
Key Point 2: Anions: chloride (Cl−), oxide (O2−).
Example: FeCl2 is iron(II) chloride; FeCl3 is iron(III) chloride.
Covalent Bonding and Molecules
Bond Formation and Laws of Proportion
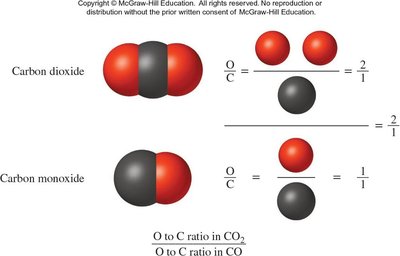
In covalent bonding, electrons are shared between atoms to achieve noble gas configurations. Molecules are combinations of atoms held together by covalent bonds. The law of definite proportions states that compounds always contain the same elements in the same ratio, while the law of multiple proportions describes ratios in compounds formed from the same elements.
Key Point 1: Covalent bonds involve shared electron pairs.
Key Point 2: Molecules can be elements (O2) or compounds (CO2).
Example: Carbon dioxide (CO2) always has a 2:1 ratio of oxygen to carbon.
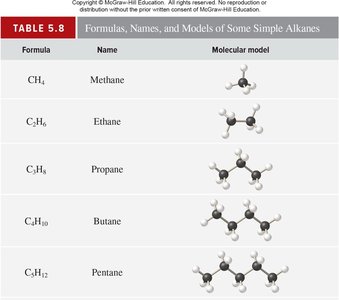
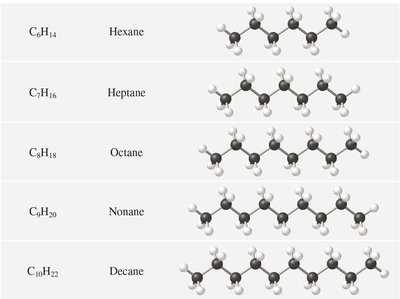
Molecular, Empirical, and Structural Formulas
Types of Chemical Formulas
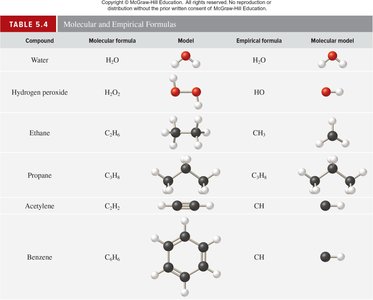
Chemical formulas denote the composition of substances. Molecular formulas show the exact number of atoms, empirical formulas show the simplest ratio, and structural formulas indicate the arrangement of atoms.
Key Point 1: Allotropes are different forms of the same element (e.g., O2 and O3).
Key Point 2: Empirical formulas may be the same as molecular formulas for some compounds.
Example: Ethanol's molecular formula is C2H6O; its empirical formula is C2H6O.




Naming Molecular Compounds
Systematic Nomenclature and Prefixes
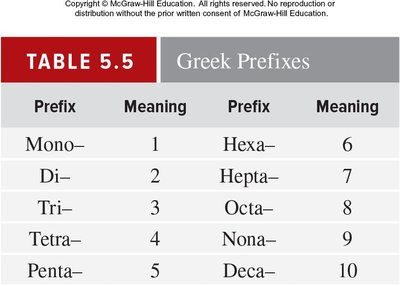
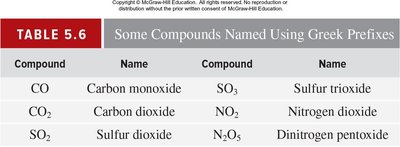
Binary molecular compounds are named using systematic rules. The first element is named as is, the second element's ending is changed to '-ide', and Greek prefixes denote the number of atoms.
Key Point 1: Prefixes: mono-, di-, tri-, tetra-, penta-, etc.
Key Point 2: The prefix 'mono-' is usually omitted for the first element.
Example: N2O5 is dinitrogen pentoxide.
Compounds Containing Hydrogen and Acids
Common Names and Acid Nomenclature
Molecular compounds containing hydrogen often have nonsystematic names. Acids are named by modifying the names of their constituent elements, especially when dissolved in water.
Key Point 1: Common names: NH3 (ammonia), H2O (water).
Key Point 2: Binary acids: hydrogen chloride becomes hydrochloric acid.
Example: HCl in water is called hydrochloric acid.

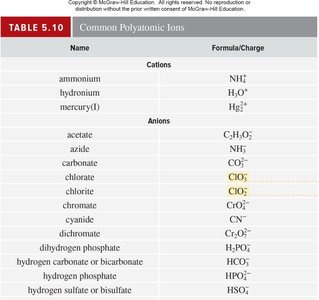
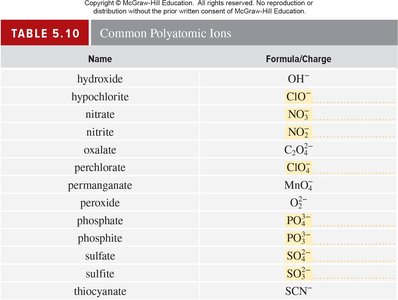
Polyatomic Ions, Oxoanions, and Oxoacids
Structure and Nomenclature
Polyatomic ions are combinations of two or more atoms with a net charge. Oxoanions contain oxygen and another element, and their names depend on the number of oxygen atoms. Oxoacids produce hydrogen ions and corresponding oxoanions in water.
Key Point 1: Naming oxoanions: 'per-' and 'hypo-' prefixes indicate more or fewer oxygen atoms.
Key Point 2: Oxoacids: '-ic' for acids from '-ate' ions, '-ous' for acids from '-ite' ions.
Example: H2SO4 is sulfuric acid; H2SO3 is sulfurous acid.
Hydrates
Water of Crystallization
Hydrates are compounds with a specific number of water molecules incorporated into their solid structure. The water can be removed by heating, resulting in an anhydrous compound.
Key Point 1: Systematic names include the number of water molecules (e.g., pentahydrate).
Key Point 2: Anhydrous compounds differ in appearance from their hydrated forms.
Example: CuSO4·5H2O is blue; anhydrous CuSO4 is white.

Molecular and Formula Mass
Calculating Masses
The molecular mass is the sum of atomic masses in a molecule, measured in atomic mass units (amu). Formula mass applies to ionic compounds, calculated similarly using the empirical formula.
Key Point 1: Use atomic masses from the periodic table.
Key Point 2: Formula mass is used for ionic compounds; molecular mass for covalent compounds.
Example: Molecular mass of H2O: amu.
Percent Composition of Compounds
Determining Elemental Percentages
Percent composition by mass is the percentage of each element in a compound. It is calculated using the formula:
Formula:
Key Point 1: The sum of percent compositions should be approximately 100%.
Example: In Li2CO3, %Li = 18.79%, %C = 16.25%, %O = 64.96%.
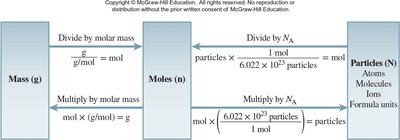
Molar Mass and Interconversions
Mole Concept and Calculations
Molar mass is the mass in grams of one mole of a substance. It is numerically equal to the atomic or formula mass. The mole concept allows conversion between mass, moles, and number of particles using Avogadro's number ().
Key Point 1: particles.
Key Point 2: Use molar mass as a conversion factor between mass and moles.
Example:
Empirical and Molecular Formulas from Percent Composition
Determination Process
Empirical formulas show the simplest ratio of elements, while molecular formulas show the actual number of atoms. Percent composition and molar mass are used to determine these formulas.
Key Point 1: Assume a 100 g sample for ease of calculation.
Key Point 2: Convert grams to moles, divide by the smallest value, and adjust to whole numbers.
Example: A compound with 30.45% N and 69.55% O yields an empirical formula of NO2 and a molecular formula of N2O4 if the molar mass is 92 g/mol.
Chapter Summary: Key Points
Compounds and hydrates
Lewis dot symbols
Ionic and covalent bonding
Chemical formulas and nomenclature
Lattice energy and stability
Molecular, empirical, and structural formulas
Polyatomic ions, oxoanions, and oxoacids
Percent composition and molar mass
Interconverting mass, moles, and particles
